Abstract
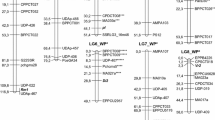
Nine different F2 families of peach [Prunus persica (L.) Batsch] were analyzed for linkage relationships between 14 morphological and two isozyme loci. Linkage was detected between weeping (We) and white flower (W), 33 cM; double flower (Dl) and pillar (Br), 10 cM; and flesh color (Y) and malate dehydrogenase (Mdh1), 26 cM. A leaf variant phenotypically distinct from the previously reported wavy-leaf (Wa) mutant in peach was found in progeny of ‘Davie II’. The new willow-leaf character (designated Wa2) was closely linked (0.4 cM) to a new dwarf phenotype (designated Dw3). Two families derived from the pollen-fertile cultivar ‘White Glory’ segregated for pollen sterility, but segregation did not follow a 3∶1 ratio. Evidence is presented suggesting that ‘White Glory’ possesses a pollen-sterility gene (designated Ps2) that is non-allelic to the previously reported pollen-sterility gene (Ps) in peach. Ps2 was linked to both weeping (We-Ps2, 15.5 cM) and white flower (Ps2-W, 25.3 cM). A genomic map of peach containing 83 RAPD, one isozyme, and four morphological markers was generated using an F2 family obtained by selfing an NC174RL x ‘Pillar’ F1. A total of 83 RAPD markers were assigned to 15 linkage groups. Various RAPD markers were linked to morphological traits. Bulked segregant analysis was used to identify RAPD markers flanking the red-leaf (Gr) and Mdh1 loci in the NC174RL x ‘Pillar’ and ‘Marsun’ x ‘White Glory’ F2 families, respectively. Three markers flanking Mdh1 and ten markers flanking Gr were identified. The combination of RAPD markers and bulked segregant analysis provides an efficient method of identifying markers flanking traits of interest. Markers linked to traits that can only be scored late in development are potentially useful for marker-aided selection in trees. Alternatives for obtaining additional map order information for repulsion-phase markers in large F2 populations are proposed.
Similar content being viewed by others
References
Allard R (1956) Formulae and tables to facilitate the calculation of recombination values in heredity. Hilgardia 24:235–278
Arulsekar S, Parfitt D, Beres W, Hansche P (1986) Genetics of malate dehydrogenase isozymes in peach. J Hered 77:49–51
Arumuganathan K, Earle E (1991) Nuclear DNA content of some important plant species. Plant Mol Biol Rep 9:208–218
Bailey J, French A (1949) The inheritance of certain fruit and foliage characters in the peach. Mass Agr Exp Sta Bul 452
Bousquet J, Simon L, LaLonde M (1990) DNA amplification from vegetative and sexual tissues of trees using polymerase chain reaction. Can J For Res 20:254–257
Gebhardt C, Ritter E, Barone A, Debener T, Walkemeier B, Schachtschabel U, Kaufmann H, Thompson R, Bonierbale M, Ganal M, Tanksley S, Salamini F (1991) RFLP maps of potato and their alignment with the homoeologous tomato genome. Theor Appl Genet 83:49–57
Giovannoni J, Wing R, Ganal M, Tanksley S (1991) Isolation of molecular markers from specific chromosomal intervals using DNA pools from existing mapping populations. Nucleic Acids Res 19:6553–6558
Grattapaglia D, Wilcox P, Chaparro J, O'Malley D, McCord S, Whetten R, McIntyre L, Sederoff R (1991) A RAPD map in 60 days. Proc 3rd Int Congress of Plant Mol Biol 2224
Hansche P (1988) Two genes that induce brachytic dwarfism in peach. HortScience 23:604–606
Hauagge R, Kester D, Asay R (1987) Isozyme variation among California almond cultivars: I. Inheritance. J Am Soc Hort Sci 112:687–693
Hesse C (1975) Peaches 285–335 In: Janick J, Moore J (eds) Advances in fruit breeding. Purdue University Press West Lafayette, Indiana, pp 623
Junghans H, Metzlaff M (1990) A simple and rapid method for the preparation of total plant DNA. BioTechniques 8:176
Kosambi D (1944) The estimation of map distances from recombination values. Ann Eugen 12:172–175
Lander E, Green P, Abrahamson J, Barlow A, Daly M, Lincoln S, Newberg L (1987) MMAKER: An interactive computer program for constructing primary linkage maps of experimental and natural populations. Genomics 1:174–181
Mather K (1936) Types of linkage data and their value. Ann Eugen 7:251–264
Michelmore R, Paran I, Kesseli RV (1991) Identification of markers linked to disease-resistance genes by bulked segregant analysis: a rapid method to detect markers in specific genomic regions by using segregating populations. Proc Natl Acad Sci USA 88:8928–9832
Monet R (1989) Peach genetics: past present and future. In: Coston D (ed) 2nd Int peach symposium. Acta Hort 254: 49–57
Monet R, Gibault B (1991) Polymorphisme de l'a amylase chez le pecher. Etude genetique. Agronomie 11:353–358
Monet R, Bastard Y, Gibault B (1985) Etude genetique et amelioration des peches plates. Agronomie 5:727–731
Monet R, Bastard Y, Gibault B (1988) Etude genetique du caractere “port pleureur” chez le pecher. Agronomie 8:127–132
Mowrey B, Werner D, Byrne D (1990) Inheritance of isocitrate dehydrogenase, malate dehydrogenase, and shikimate dehydrogenase in peach and peach x almond hybrids. J Am Soc Hort Sci 115:312–319
Myers S, Okie W, Lightner G (1989) The ‘Elberta’ peach. Fruit Var J 43:130–138
Paran I, Kesseli R, Michelmore R (1991) Identification of restriction fragment length polymorphic DNA markers linked to downy mildew resistance genes in lettuce using near-isogenic lines. Genome 34:1021–1027
Ritter E, Gebhardt C, Salamini F (1990) Estimation of recombination frequencies and construction of RFLP linkage maps in plants from crosses between heterozygous parents. Genetics 125:645–654
Robinson R (1971) Gene mapping in laboratory mammals. Part A. Plenum Publishing Co., London
Santi F, Lemoine M (1990) Genetic markers for Prunus avium L: inheritance and linkage of isozyme loci. Ann Sci For 47:131–139
Scott D, Cullinan F (1942) The inheritance of wavy-leaf character in the peach. J Hered 33:293–295
Suzuki D, Griffiths A, Miller J, Lewontin R (1989) An introduction to genetic analysis. W.H. Freeman and Co, New York
Tanksley S, Young N, Paterson A, Bonierbale M (1989) RFLP mapping in plant breeding: new tools for an old science. Bio/technol 7:257–264
Weeden N, Timmerman G, Hemmat M, Loaiza-Figueroa F, Lu J (1991) Evaluation and characterization of RAPD markers for genome mapping. Proc 3rd Int Congress of Plant Mol Biol 1721
Werner D (1985) ‘White Glory’ weeping nectarine. HortScience 20:308–309
Werner D (1992) Catalase polymorphism and inheritance in peach. HortScience 27:41–43
Werner D, Moxley D (1991) Relationship between malate dehydrogenase isozyme genotype and plant vigor in peach. J Am Soc Hort Sci 116:327–329
Whitkus R, Doebley J, Lee M (1992) Comparative genome mapping of sorghum and maize. Genetics 132:1119–1130
Williams JGK, Kubelik AR, Livak KJ, Rafalski JA, Tingey SV (1990) DNA polymorphisms amplified by arbitrary primers are useful as genetic markers. Nucleic Acids Res 18:6531–6535
Womack J, Dietz A, Gallagher D, Li L, Zhang N, Neibergs H, Moll Y, Ryan A (1992) Addition of 43 informative comparative anchor loci to the bovine synteny map. Proc 1992 Miami Bio/Technology Winter Symposium 2:8
Yamazaki K, Okabe M, Takahashi E (1987) Inheritance of some characteristics and breeding of new hybrids in flowering peaches. Bull Kanagawa Hort Exp Sta 34:46–53
Author information
Authors and Affiliations
Additional information
Communicated by H. K. Dooner
This work was supported in part by the McKnight Foundation, North Carolina Biotechnology Center, North Carolina State University Forest Biotechnology Research Consortium, and the North Carolina Agricultural Research Service, Raleigh, North Carolina
Rights and permissions
About this article
Cite this article
Chaparro, J.X., Werner, D.J., O'Malley, D. et al. Targeted mapping and linkage analysis of morphological isozyme, and RAPD markers in peach. Theoret. Appl. Genetics 87, 805–815 (1994). https://doi.org/10.1007/BF00221132
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00221132