Summary
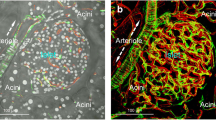
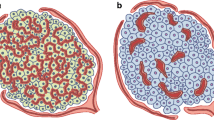
We have suggested that the order of cellular vascular perfusion within the islet is important in the regulation of islet hormone secretion. Anatomically, the A and D cells appear to be randomly dispersed throughout the mantle. Although islet capillary blood flow is known to be from the B-cell core to the A- and D-cell mantle, it has not yet been established whether the cells of the mantle may influence one another vascularly. Rat pancreata were perfused in vitro anterogradely and retrogradely with or without glucagon antibody in order to determine the order of cellular perfusion and interaction between the A and D cells in the islet mantle. Anterograde infusion of glucagon antibody did not affect insulin secretion, but rapidly decreased somatostatin secretion −46±8%, (p<0.005). Retrograde infusion of glucagon anti body decreased insulin secretion (−27±8%, p<0.005) but had no effect upon somatostatin secretion. This study not only confirms a core to mantle islet perfusion but also establishes that the A cell precedes the D cell in the terms of vascular perfusion. Thus within the islet, vascular borne insulin regulates the release of glucagon, which in turn, regulates the release of somatostatin. Somatostatin is vascularly neutral owing to its downstream position in the sequence (B to A to D) of cellular perfusion.
Article PDF
Similar content being viewed by others
References
Orci L, Unger RH (1975) Functional subdivision of islets of Langerhans and possible role of D cells. Lancet II: 1243
Orci L, Malaisse-Lagae F, Rouiller D, Renold AE, Perelet A, Unger RH (1975) A morphological basis for intercellular communication between alpha and beta cells in the endocrine pancreas. J Clin Invest 56: 1066–1070
Haist RE (1965) Effects of changes in stimulation on the structure and function of islet cells. In: Leibel BS, Wrenshall GA (eds) On the nature and treatment of diabetes. Excerpta Medica, Amsterdam, pp 12–130
Samols E, Weir GC, Bonner-Weir S (1983) Intraislet insulin-glucagon somatostatin relationships. In: Lefebvre PJ (ed) Handbook of experimental pharmacology, Vol 66/II. Springer, Berlin Heidelberg New York, pp 133–173
Samols E, Tyler J, Marks V (1972) Glucagon-insulin interrelationships. In: Lefebvre PJ, Unger RH (eds) Glucagon. Molecular physiology, clinical and therapeutic implications. Pergamon Press, Oxford, pp 151–173
Unger RH, Orci L (1977) Hypothesis: the possible role of the pancreatic D cell in the normal and diabetic states. Diabetes 26: 241–244
Fujita T, Yanatori Y, Murakomi T (1976) Insulo-acinar axis, its vascular basis and its functional and morphological changes caused by CCK-PZ and caerulein. In: Fujita T (ed) Endocrine gut and pancreas. Elsevier, Amsterdam, pp 347–358
Fujita T (1985) Morphology of islet blood supple. In: Lainer J, Pohl SL (eds) Methods in diabetes research, Vol.I, Part C. Wiley-Interscience, New York, pp 325–332
Bonner-Weir S, Orci L (1982) New perspectives on the microvasculature of the islets of Langerhans in the rat. Diabetes 31: 883–889
Stagner JI, Samols E (1986) Retrograde perfusion as a model for testing the relative effects of glucose versus insulin on the A cell. J Clin Invest 77: 1034–1037
Stagner JI, Samols E, Bonner-Weir S (1988) β→α→δ Pancreatic islet cellular perfusion in dogs. Diabetes 37: 1715–1721
Samols E, Stagner JI, Ewart RBL, Marks V (1988) The order of islet microvascular cellular perfusion is B→A→D in the perfused rat pancreas. J Clin Invest 82: 350–353
Maruyama H, Hisatomi A, Orci L, Grodsky GM, Unger RH (1984) Insulin within islets is a physiologic glucagon release inhibitor. J Clin Invest 74: 2296–2299
Grodsky GM, Heldt H (1984) Method for the in vitro perfusion of the pancreas. In: Larner J, Pohl SL (eds) Methods in diabetes research, Vol I, part B. Wiley, New York, pp 137–146
Weir GC, Samols E, Loo S, Patel YC, Gabbay KH (1979) Somatostatin and pancreatic polypeptide secretion: effects of glucagon, insulin and arginine. Diabetes 28: 35–40
Kvietys PR, Perrey A, Grange DN (1983) Permeability of pancreatic capillaries to small molecules. Am J Physiol 245: G519-G524
Maruyama H, Tominaga M, Bolli G, Orci L, Unger RH (1985) The alpha cell response to glucose change during perfusion of anti-insulin serum in pancreas isolated from normal rats. Diabetologia 28: 836–840
Unger RH (1981) The milieu interieur and the islet of Langerhans. Diabetologia 20: 1–11
Kawai K, Ipp E, Orci L, Perellet A, Unger RH (1982) Circulating somatostatin acts on the islets of Langerhans by way of a somato-statin-poor compartment. Science 218: 477–478
Samols E, Harrison J (1976) Remarkable potency of somatostatin as a glucagon suppressant. Metabolism 25: 1495–1497
Taborsky GJ Jr (1983) Evidence of a paracrine role for pancreatic somatostatin in vivo. Am J Physiol 245: E 598-E 603
Stagner JI, Samols E, Taborsky GJ (1987) Mediation of B-cell secretion by intra-islet somatostatin. Diabetes 36: 7 (Abstract)
Larsson LS, Goltermann N, de Magistris L, Rehfeld IF, Schwarte TW (1979) Somatostatin cell processes as pathways for paracrine secretion. Science 205: 1393–1395
Meda P, Perrelet A, Orci L (1982) Endocrine cell interactions within the islets of Langerhans. In: Pritts JD (ed) Functional integration of cells in animal tissues. Cambridge University Press, Cambridge, pp 113–131
Samols E, Bonner-Weir S, Weir GC (1986) Intra-islet-insulin-glucagon-somatostatin relationships. Clin Endocrinol Metab 15: 33–58
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Stagner, J.I., Samols, E. & Marks, V. The anterograde and retrograde infusion of glucagon antibodies suggests that A cells are vascularly perfused before D cells within the rat islet. Diabetologia 32, 203–206 (1989). https://doi.org/10.1007/BF00265095
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00265095