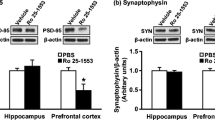
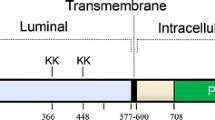
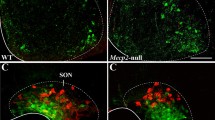
Abstract
The molecular mechanisms responsible for behavior are largely unknown. A state of the art model, paving the path from genes to behavior, is offered by transgenic animals. Candidate molecules are classic neuropeptides, such as vasoactive intestinal peptide (VIP). Transgenic mice harboring a chimeric VIP gene driven by the polyoma promoter were produced. Behavioral studies revealed learning impairment and prolonged retardation in memory acquisition in the genetically altered animals. Furthermore, reduced performance was observed when the male transgenic mice were tested for sexual activity in the presence of receptive females. Surprisingly, radioimmunoassays showed an approx 20% decrease in the VIP content of the transgenic mice brains. To directly assess genetically reduced VIP content as a cause for learning impairment, transgenic mice carrying diphtheria toxia-encoding sequences driven by the rat VIP promoter were created. These animals had reduced brain VIP and exhibited deficiencies in learning abilities, strongly supporting an important neurobiological function for VIP in vivo.
Similar content being viewed by others
References
Agoston D. V., Eiden L. E., Brenneman D. E., and Gozes I. (1991) Spontaneous electrical activity regulates vasoactive intestinal peptide expression in dissociated spinal cord cell cultures.Mol. Brain Res. 10, 235–240.
Arimura A. (1992) Pituitary adenylate cyclase activating polypeptide (PACAP): discovery and current status of research.Regulatory Peptides 37, 287.
Bodner M., Fridkin M., and Gozes I. (1985) VIP and PHM-27 sequences are located on two adjacent exons in the human genome.Proc. Natl. Acad. Sci. USA 82, 3548–3551.
Bohnlein E., Chowdhury K., and Gruss P. (1985) Functional analysis of the regulatory region of polyoma mutant F9-1 DNA.Nucleic Acids Res. 13, 4789–4809.
Brenneman D. E. and Eiden L. E. (1986) Vasoactive intestinal peptide and electrical activity influence neuronal survival.Proc. Natl. Acad. Sci. USA 83, 1159–1162.
Brenneman D. E., Nicol T., Warren D., and Bowers L. M. (1990) Vasoactive intestinal peptide: a neurotrophic releasing agent and astroglia mitogen.J. Neurosci. Res. 25, 386–394.
Csillag A., Zilles K., Schleicher A., and Hajos F. (1992) Matching localization of immunoreactive VIP and VIP receptor in the visual cortex.Neurosci. Soc. Abs. 18, 1451.
Dixson A. F., Kendrick K. M., Blank M. A., and Bloom S. R. (1984) Effects of tactile and electrical stimuli upon the release of vasoactive intestinal polypeptide in the mammalian penis.J. Endocrinol. 100, 249–252.
Emson P. C., Fahrenkrug J., Schaffalitzky de Muckadell O. B., Jessel T. M., and Iverson, L. L. (1978) Vasoactive intestinal peptide (VIP) vesicular localization and potassium evoked release from rat hypothalamus.Brain Res. 143, 174–178.
Fink J. S., Verhave M., Walton K., Mandel G., and Goodman R. H. (1991) Cyclic-AMP and phorbol ester-induced transcriptional activation are mediated by the same enhancer element in the human vasoactive intestinal peptide gene.J. Biol. Chem. 266, 3882–3887.
Giladi E., Shani Y., and Gozes I. (1990) The complete structure of the rat VIP gene.Mol. Brain Res. 7, 261–267.
Glowa J. R., Panlilio L. V., Brenneman D. E., Gozes I., Fridkin M., and Hill J. M. (1992) Learning impairment following intracerebral administration of the HIV envelope protein gp120 or a VIP antagonist.Brain Res. 570, 49–53.
Gozes I., Bodner M., Shani Y., and Fridkin M. (1986) Structure and expression of the vasoactive intestinal peptide (VIP) gene in a human tumor.Peptides 7, 1–6.
Gozes I. and Brenneman D. E. (1989) VIP molecular biology and neurobiological function.Mol. Neurobiol. 3, 1–36.
Gozes I. and Brenneman D. E. (1993) Neuropeptides as growth and differentiation factors in general and VIP in particular.J. Mol. Neurosci. 4, 1–9.
Gozes I. and Fridkin M. (1992) A fatty neuropeptide: potential drug for noninvasive impotence treatment in a rat model.J. Clin. Invest. 90, 810–814.
Gozes I., Hill J. M., Mervis R. F., Fridkin M., and Brenneman D. E. (1990) VIP antagonist produces neuronal damage and retardation of behavioral development in neonatal rats.Neurosci. Soc. Abs. 16, 1292.
Gozes I., Meltzer E., Rubinraut S., Brenneman D. E., and Fridkin M. (1989) Vasoactive intestinal peptide potentiate sexual behavior: inhibition by novel antagonist.Endocrinology 125, 2945–2949.
Gozes I., McCune S. K., Jacobson L., Warren D., Moody T. W., Fridkin M., and Brenneman D. E. (1991a) An antagonist to vasoactive intestinal peptide: effects on cellular functions in the central nervous system.J. Pharmacol. Exp. Therap. 257, 959–966.
Gozes Y., Brenneman D. E., Fridkin M., Asofsky R., and Gozes I. (1991b) A VIP antagonist distinguishes VIP receptors on spinal cord cells and lymphocytesBrain Res. 540, 319–321.
Gozes I., Shani Y., and Rostene W. H. (1987) Developmental expression of the VIP-gene in brain and intestine.Mol. Brain Res. 2, 137–148.
Griep A. E. and Westphal H. (1990) Differentiation versus proliferation of transgenic mouse lens cells expressing polyoma large T antigen: evidence for regulation by an endogenous growth factor.New Biol. 2, 727–738.
Heinrich G., Gros P., and Habener J. F. (1984) Glucagon gene sequence.J. Biol. Chem.,259, 14,082–14,087.
Hill J. M., Gozes I., Hill J. L., Fridkin M., and Brenneman D. E. (1991) Vasoactive intestinal peptide antagonist retards the development of neonatal behaviors in the rat.Peptides 12, 187–192.
Lamperti E. D., Rosen K. M., and Villa-Komaroff L. (1991) Characterization of the gene and messages for vasoactive intestinal polypeptide (VIP) in rat and mouse.Molecular Brain Res. 9, 217–231.
Loren I., Emson P. C., Fahrenkrug J., Bjorklund A., Alumets J., Hakanson R., and Sundler F. (1979) Distribution of vasoactive intestinal polypeptide in the rat mouse brain.Neuroscience 4, 1953–1976.
Magistretti P. J., Morrison J. H., Shoemaker W. J., Sapin V., and Bloom F. E. (1981) Vasoactive intestinal polypeptide induces glycogenolysis in mouse cortical slices: a possible regulatory mechanism for the local control of energy metabolism.Proc. Natl. Acad. Sci. USA 78, 6535–6539.
Mahon K. A., Chepelinsky A. B., Khillan J. S., Overbeek P. A., Piatigorsky J., and Westphal H. (1987) Oncogenesis of the lens in transgenic mice.Science 235, 1622–1628.
Maxwell I. H., Maxwell F., and Glode L. M. (1986) Regulated expression of a diphtheria toxin A-chain gene transfected into human cells: possible strategy for inducing cancer cell suicide.Cancer Res. 46, 4660–4664.
Maxwell F., Maxwell I. H., and Glode L. M. (1987) Cloning, sequence determination and expression in transfected cells of the coding sequence for the tox 176 attenuated diphtheria toxin A chain.Molec. Cell. Biol. 7, 1576–1579.
Moody, T. W., Zia, F., Dravi, M., Brenneman, D. E., Fridkin, M., Davidson, A., and Gozes, I. (1993) A vasoactive intestinal peptide antagonist inhibits non-small cell cancer growth.Proc. Natl. Acad. Sci. USA 90, 4345–4349.
Morris R. G. M., Anderson E., Lynch G. S., and Baudry M. (1986) Selective impairment of learning and blockade of long-term potentiation by anN-methyl-d-aspartate receptor antagonist, AP5.Nature 319, 774–776.
Morris R. G. M., Garrud P., Rawlins J. N. P., and O’Keefe J. (1982) Place navigation impaired in rats with hippocampal lesions.Nature 297, 681–683.
Nakamura T., Mahon K. A., Miskin R., Dey A., Kuwabara T., and Westphal H. (1989) Differentiation and oncogenesis: phenotypically distinct lens tumors in transgenic mice.New Biol. 1, 193–204.
Ottesen B., Wagener G., Virag R., and Fahrenkrug J. (1984) Penile erection: possible role for vasoactive intestinal polypeptide.Br. Med. J. 288, 9–11.
Rosselin G., Anteunis A., Astesano A., Boissard C., Gali P., Hejblum G., and Marie J. C. (1988) Regulation of vasoactive intestinal peptide receptor.Ann. NY Acad. Sci. (Said S. I. and Mutt, V., eds.),527, 220–237.
Said S. I. and Mutt V. (1970) Polypeptide with broad biological activity: isolation from small intestine.Science 169, 1217–1218.
Southern E. (1975) Detection of specific sequences among DNA fragments separated by gel electrophoresis.J. Mol. Biol. 98, 503–517.
Sweeney J. E., Hohmann C. F., Moran T. H., and Coyle J. T. (1988) A long acting cholinesterase inhibitor reverse spatial memory deficits in mice.Pharmacol. Biochem. Behav. 31, 141–147.
Tybulewicz V. L. J., Tremblay M. L., Lamarca M. E., Willemsen R., Stubblefield B. K., Winfield S., Zablocka B., Sidransky E., Martin B. M., Huang S. P., Mintzer K. A., Westphal H., Mulligan R. C., and Ginns E. I. (1992) Animal model of Gaucher’s diseae from targeted disruption of the mouse glucocerebrosidase gene.Nature 357, 407–410.
Upchurch M. and Wehner J. M. (1988) Differences between inbred strains of mice in Morris Water Maze Performance.Behav. Genet. 18, 55–68.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gozes, I., Glowa, J., Brenneman, D.E. et al. Learning and sexual deficiencies in transgenic mice carrying a chimeric vasoactive intestinal peptide gene. J Mol Neurosci 4, 185–193 (1993). https://doi.org/10.1007/BF02782501
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02782501