Abstract
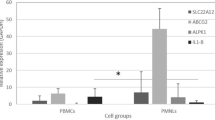
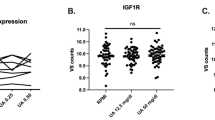
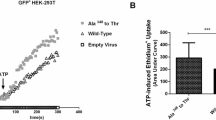
The molecular functions and pathophysiologic role of the lymphocyte α-kinase gene (ALPK1) in gout are unknown. We aimed to examine ALPK1 expression in patients with gout and investigate its role in monosodium urate monohydrate (MSU)-induced inflammatory responses. Microarray data mining was performed with six datasets containing three clinical gout and three volunteer samples. Real-time quantitative polymerase chain reaction (qPCR) assay was used to profile ALPK1 mRNA expression in 62 independent samples. RNA interference for ALPK1 suppression in THP1 cells (human monocytic cell line) was used to scrutinize the functional role of ALPK1 in MSU-mediated inflammatory responses, and ALPK1 expression in MSU-treated THP1 cells was determined by qPCR and Western blot analysis. Cytokine mRNA expression in HEK293 cells after incubation with different concentrations of MSU crystals in the presence or absence of ALPK1 was also detected by qPCR, and ERK1/2, p38, and JNK expressions were investigated by Western blot analysis. ALPK1 mRNA was overexpressed in the clinical gout samples. MSU treatment promoted ALPK1 expression at the mRNA and protein levels. Furthermore, ALPK1 knockdown in THP1 cells resulted in a markedly decreased IL-1β, TNF-α, and IL-8 mRNA expression; plasmid ALPK1 transfection and MSU stimulation synergistically increased the mRNA expression of these cytokines in a concentration-dependent manner. The synergistic effect also led to ERK1/2 activation. ALPK1 is a gout-susceptible gene involved in MSU-induced inflammatory responses. It may contribute to the development of gout by enhancing the inflammatory responses via the mitogen-activated protein kinase pathway.






Similar content being viewed by others
References
Wang WH, Chang SJ, Wang TN, Cheng LS, Feng YP, Chen CJ, Huang CH, Ko YC (2004) Complex segregation and linkage analysis of familial gout in Taiwanese aborigines. Arthritis Rheum 50:242–246. doi:10.1002/art.11441
Cheng LS, Chiang SL, Tu HP, Chang SJ, Wang TN, Ko AM, Chakraborty R, Ko YC (2004) Genomewide scan for gout in Taiwanese aborigines reveals linkage to chromosome 4q25. Am J Hum Genet 75:498–503. doi:10.1086/423429
Ryazanov AG, Pavur KS, Dorovkov MV (1999) Alpha-kinases: a new class of protein kinases with a novel catalytic domain. Curr Biol 9:R43–R45
Heine M, Cramm-Behrens CI, Ansari A, Chu HP, Ryazanov AG, Naim HY, Jacob R (2005) Alpha-kinase 1, a new component in apical protein transport. J Biol Chem 280:25637–25643. doi:10.1074/jbc.M502265200
Schumacher HR (1996) Crystal-induced arthritis: an overview. Am J Med 100:46S–52S
Bieber JD, Terkeltaub RA (2004) Gout: on the brink of novel therapeutic options for an ancient disease. Arthritis Rheum 50:2400–2414. doi:10.1002/art.20438
di Giovine FS, Malawista SE, Thornton E, Duff GW (1991) Urate crystals stimulate production of tumor necrosis factor alpha from human blood monocytes and synovial cells. Cytokine mRNA and protein kinetics, and cellular distribution. J Clin Invest 87:1375–1381. doi:10.1172/JCI115142
Liu R, O’Connell M, Johnson K, Pritzker K, Mackman N, Terkeltaub R (2000) Extracellular signal-regulated kinase 1/extracellular signal-regulated kinase 2 mitogen-activated protein kinase signaling and activation of activator protein 1 and nuclear factor kappaB transcription factors play central roles in interleukin-8 expression stimulated by monosodium urate monohydrate and calcium pyrophosphate crystals in monocytic cells. Arthritis Rheum 43:1145–1155. doi:10.1002/1529-0131(200005)43:5<1145::AID-ANR25>3.0.CO;2-T
Jaramillo M, Godbout M, Naccache PH, Olivier M (2004) Signaling events involved in macrophage chemokine expression in response to monosodium urate crystals. J Biol Chem 279:52797–52805. doi:10.1074/jbc.M403823200
Wallace SL, Robinson H, Masi AT, Decker JL, McCarty DJ, Yu TF (1977) Preliminary criteria for the classification of the acute arthritis of primary gout. Arthritis Rheum 20:895–900
Choi HK, Atkinson K, Karlson EW, Willett W, Curhan G (2004) Purine-rich foods, dairy and protein intake, and the risk of gout in men. N Engl J Med 350:1093–1103. doi:10.1056/NEJMoa035700
Jiang SS, Fang WT, Hou YH, Huang SF, Yen BL, Chang JL, Li SM, Liu HP, Liu YL, Huang CT et al (2010) Upregulation of SOX9 in lung adenocarcinoma and its involvement in the regulation of cell growth and tumorigenicity. Clin Cancer Res 16:4363–4373. doi:10.1158/1078-0432.CCR-10-0138
Ding LH, Xie Y, Park S, Xiao G, Story MD (2008) Enhanced identification and biological validation of differential gene expression via Illumina whole-genome expression arrays through the use of the model-based background correction methodology. Nucleic Acids Res 36:e58. doi:10.1093/nar/gkn234
Schiltz C, Liote F, Prudhommeaux F, Meunier A, Champy R, Callebert J, Bardin T (2002) Monosodium urate monohydrate crystal-induced inflammation in vivo: quantitative histomorphometric analysis of cellular events. Arthritis Rheum 46:1643–1650. doi:10.1002/art.10326
Martinon F, Petrilli V, Mayor A, Tardivel A, Tschopp J (2006) Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature 440:237–241. doi:10.1038/nature04516
Choi HK, Mount DB, Reginato AM (2005) Pathogenesis of gout. Ann Intern Med 143:499–516
Liu-Bryan R, Liote F (2005) Monosodium urate and calcium pyrophosphate dihydrate (CPPD) crystals, inflammation, and cellular signaling. Joint Bone Spine 72:295–302. doi:10.1016/j.jbspin.2004.12.010
Yagnik DR, Hillyer P, Marshall D, Smythe CD, Krausz T, Haskard DO, Landis RC (2000) Noninflammatory phagocytosis of monosodium urate monohydrate crystals by mouse macrophages. Implications for the control of joint inflammation in gout. Arthritis Rheum 43:1779–1789. doi:10.1002/1529-0131(200008)43:8<1779::AID-ANR14>3.0.CO;2-2
Landis RC, Yagnik DR, Florey O, Philippidis P, Emons V, Mason JC, Haskard DO (2002) Safe disposal of inflammatory monosodium urate monohydrate crystals by differentiated macrophages. Arthritis Rheum 46:3026–3033. doi:10.1002/art.10614
Scott P, Ma H, Viriyakosol S, Terkeltaub R, Liu-Bryan R (2006) Engagement of CD14 mediates the inflammatory potential of monosodium urate crystals. J Immunol 177:6370–6378
Pope RM, Tschopp J (2007) The role of interleukin-1 and the inflammasome in gout: implications for therapy. Arthritis Rheum 56:3183–3188. doi:10.1002/art.22938
Martin WJ, Walton M, Harper J (2009) Resident macrophages initiating and driving inflammation in a monosodium urate monohydrate crystal-induced murine peritoneal model of acute gout. Arthritis Rheum 60:281–289. doi:10.1002/art.24185
Martin WJ, Shaw O, Liu X, Steiger S, Harper JL (2011) Monosodium urate monohydrate crystal-recruited noninflammatory monocytes differentiate into M1-like proinflammatory macrophages in a peritoneal murine model of gout. Arthritis Rheum 63:1322–1332. doi:10.1002/art.30249
Dorovkov MV, Ryazanov AG (2004) Phosphorylation of annexin I by TRPM7 channel-kinase. J Biol Chem 279:50643–50646. doi:10.1074/jbc.C400441200
Riazanova LV, Pavur KS, Petrov AN, Dorovkov MV, Riazanov AG (2001) Novel type of signaling molecules: protein kinases covalently linked to ion channels. Mol Biol (Mosk) 35:321–332
Ryazanov AG (2002) Elongation factor-2 kinase and its newly discovered relatives. FEBS Lett 514:26–29
Liu R, Aupperle K, Terkeltaub R (2001) Src family protein tyrosine kinase signaling mediates monosodium urate crystal-induced IL-8 expression by monocytic THP-1 cells. J Leukoc Biol 70:961–968
Bomalaski JS, Baker DG, Brophy LM, Clark MA (1990) Monosodium urate crystals stimulate phospholipase A2 enzyme activities and the synthesis of a phospholipase A2-activating protein. J Immunol 145:3391–3397
Gaudry M, Gilbert C, Barabe F, Poubelle PE, Naccache PH (1995) Activation of Lyn is a common element of the stimulation of human neutrophils by soluble and particulate agonists. Blood 86:3567–3574
Chen M, Xu R (2011) Motor coordination deficits in Alpk1 mutant mice with the inserted piggyBac transposon. BMC Neurosci 12:1. doi:10.1186/1471-2202-12-1
Enomoto A, Kimura H, Chairoungdua A, Shigeta Y, Jutabha P, Cha SH, Hosoyamada M, Takeda M, Sekine T, Igarashi T et al (2002) Molecular identification of a renal urate anion exchanger that regulates blood urate levels. Nature 417:447–452. doi:10.1038/nature742
McConnell RE, Tyska MJ (2007) Myosin-1a powers the sliding of apical membrane along microvillar actin bundles. J Cell Biol 177:671–681. doi:10.1083/jcb.200701144
Brone B, Eggermont J (2005) PDZ proteins retain and regulate membrane transporters in polarized epithelial cell membranes. Am J Physiol Cell Physiol 288:C20–C29. doi:10.1152/ajpcell.00368.2004
Anzai N, Miyazaki H, Noshiro R, Khamdang S, Chairoungdua A, Shin HJ, Enomoto A, Sakamoto S, Hirata T, Tomita K et al (2004) The multivalent PDZ domain-containing protein PDZK1 regulates transport activity of renal urate-anion exchanger URAT1 via its C terminus. J Biol Chem 279:45942–45950. doi:10.1074/jbc.M406724200
Cunningham R, Brazie M, Kanumuru S, Xiaofei E, Biswas R, Wang F, Steplock D, Wade JB, Anzai N, Endou H et al (2007) Sodium-hydrogen exchanger regulatory factor-1 interacts with mouse urate transporter 1 to regulate renal proximal tubule uric acid transport. J Am Soc Nephrol 18:1419–1425. doi:10.1681/ASN.2006090980
Acknowledgments
This study was supported by grants from Center of Excellence for Environmental Medicine, Kaohsiung Medical University, Ministry of Education, Taiwan (KMU-EM-98-1-1 and KMU-EM-99-1-1), and the National Science Council, Taiwan (NSC97-2314-B-037-007-MY3 and NSC99-2628-B-037-039-MY3).
Disclosure statement
The authors have declared that no conflict of interest exists.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, SJ., Tu, HP., Ko, A.MS. et al. Lymphocyte α-kinase is a gout-susceptible gene involved in monosodium urate monohydrate-induced inflammatory responses. J Mol Med 89, 1241–1251 (2011). https://doi.org/10.1007/s00109-011-0796-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-011-0796-5