Abstract
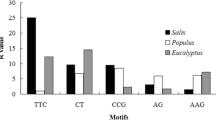
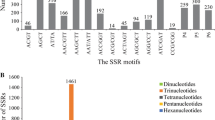
Traditionally, simple sequence repeat (SSR) markers have been developed from libraries of genomic DNA. However, the large, repetitive nature of conifer genomes makes development of robust, single-copy SSR markers from genomic DNA difficult. Expressed sequence tags (ESTs), or sequences of messenger RNA, offer the opportunity to exploit single, low-copy, conserved sequence motifs for SSR development. From a 20,275-unigene spruce EST set, we identified 44 candidate EST-SSR markers. Of these, 25 amplified and were polymorphic in white, Sitka, and black spruce; 20 amplified in all 23 spruce species tested; the remaining five amplified in all except one species. In addition, 101 previously described spruce SSRs (mostly developed from genomic DNA), were tested. Of these, 17 amplified across white, Sitka, and black spruce. The 25 EST-SSRs had approximately 9% less heterozygosity than the 17 genomic-derived SSRs (mean H=0.65 vs 0.72), but appeared to have less null alleles, as evidenced by much lower apparent inbreeding (mean F=0.046 vs 0.126). These robust SSRs are of particular use in comparative studies, and as the EST-SSRs are within the expressed portion of the genome, they are more likely to be associated with a particular gene of interest, improving their utility for quantitative trait loci mapping and allowing detection of selective sweeps at specific genes.




Similar content being viewed by others
References
Arnold C, Rossetto M, McNally J, Henry RJ (2002) The application of SSRs characterized for grape (Vitis vinifera) to conservation studies in Vitaceae. Am J Bot 89:22–28
Bérubé Y, Ritland CE, Ritland K (2003) Isolation, characterization, and cross-species utility of microsatellites in yellow cedar (Chamaecyparis nootkatensis). Genome 46:353–361
Besnard G, Achere V, Faivre Rampant P, Favre JM, Jeandroz S (2003) A set of cross-species amplifying microsatellite markers developed from DNA sequence databanks in Picea (Pinaceae). Mol Ecol Notes 3:380–383
Bowcock AM, Ruiz-Linares A, Tomfohrde J, Minch E, Kidd JR, Cavalli-Sforza LL (1994) High resolution of human evolutionary trees with polymorphic microsatellites. Nature 368:455–457
Bruford MW, Wayne RK (1993) Microsatellites and their application to population genetic studies. Curr Opin Genet Dev 3:939–943
Cardle L, Ramsay L, Milbourne D, Macaulay M, Marshall D, Waugh R (2000) Computational and experimental characterization of physically clustered simple sequence repeats in plants. Genetics 156:847–854
Dib C, Faure S, Fizames C, Samson D, Drouot N, Vignal A, Millasseau P, Marc S, Hazan J, Seboun E, Lathrop M, Gyapay G, Morissette J, Weissenbach J (1996) A comprehensive genetic map of the human genome based on 5,264 microsatellites. Nature 380:152–154
Doyle JJ, Doyle JL (1990) Isolation of plant DNA from fresh tissue. Focus 23:13–15
Ewing B, Green P (1998) Base-calling of automated sequencer traces using PHRED II. Error probabilities. Genome Res 8:186–194
Ewing B, Hillier L, Wendl MC, Green P (1998) Base-calling of automated sequencer traces using PHRED. I. Accuracy assessment. Genome Res 8:175–185
Felsenstein J (1995) PHYLIP (Phylogeny Inference Package), ver 3.57c, University of Washington
Goldstein DB, Ruiz Linares A, Cavalli-Sforza LL, Feldman MW (1995) An evaluation of genetic distances for use with microsatellite loci. Genetics 139:463–471
Hodgetts RB, Aleksiuk MA, Brown A, Clarke C, Macdonald E, Nadeem S, Khasa D (2001) Development of microsatellite markers for white spruce (Picea glauca) and related species. Theor Appl Genet 102:1252–1258
Huang X, Madan A (1999) CAP3: A DNA sequence assembly program. Genome Res 9:868–877
Jones CJ, Edwards KJ, Castaglione S, Winfield MO, Sala F, van de Wiel C, Bredemeijer G, Vosman B, Matthes M, Daly A, Brettschneider R, Bettini P, Buiatti M, Maestri E, Malcevschi A, Marmiroli N, Aert R, Volckaert G, Rueda J, Linacero R, Vazquez A, Karp A (1997) Reproducibility testing of RAPD, AFLP and SSR markers in plants by a network of European laboratories. Mol Breed 3:381–390
Morgante M, Hanafey M, Powell W (2002) Microsatellites are preferentially associated with nonrepetitive DNA in plant genomes. Nat Genet 30:194–200
Neff BD (2004) Mean d2 and divergence time: transformations and standardizations. J Hered 95:165–171
Oetting WS, Lee HK, Flanders DJ, Wiesner GL, Sellers TA, King RA (1995) Linkage analysis with multiplexed short tandem repeat polymorphisms using infrared fluorescence and M13 tailed primers. Genomics 30:450–458
Paglia G, Morgante M (1998) PCR-based multiplex DNA fingerprinting techniques for the analysis of conifer genomes. Mol Breed 4:173–177
Pfeiffer A, Olivieri AM, Morgante M (1997) Identification and characterization of microsatellites in Norway spruce (Picea abies K.). Genome 40:411–419
Queller DC, Strassmann JE, Hughes CR (1993) Microsatellites and kinship. Trends Ecol Evol 8:285–288
Rafalski A (2002) Applications of single nucleotide polymorphisms in crop genetics. Curr Opin Plant Biol 5:94–100
Rajora OP, Rahman MH, Dayanandan S, Mosseler A (2000) Isolation, characterization, inheritance and linkage of microsatellite DNA markers in white spruce (Picea glauca) and their usefulness in other spruce species. Mol Gen Genet 264:871–882
Ritland C, Ritland K (2000) DNA fragment markers in plants. In: Baker AJ (ed) Molecular methods in ecology. Blackwell, Oxford, pp 208–234
Rozen S, Skaletsky HJ (2000) Primer3 on the WWW for general users and for biologist programmers. In: Krawetz S, Misener S (eds) Bioinformatics methods and protocols: methods in molecular biology. Humana Press, Totowa, pp 365–386
Saha S, Karaca M, Jenkins JN, Zipf AE, Reddy OUK, Kantety RV (2003) Simple sequence repeats as useful resources to study transcribed genes of cotton. Euphytica 130:355–364
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Schlötterer C (2001) Genealogical inference of closely related species based on microsatellites. Genet Res 78:209–212
Scotti I, Magni F, Fink R, Powell W, Binelli G, Hedley PE (2000) Microsatellite repeats are not randomly distributed within Norway spruce (Picea abies K.) expressed sequences. Genome 43:41–46
Scotti I, Magni F, Paglia G, Morgante M (2002a) Trinucleotide microsatellites in Norway spruce (Picea abies): their features and the development of molecular markers. Theor Appl Genet 106:40–50
Scotti I, Paglia G, Magni F, Morgante M (2002b) Efficient development of dinucleotide microsatellite markers in Norway spruce (Picea abies Karst.) through dot-blot selection. Theor Appl Genet 104:1035–1041
Sigurgeirsson A, Szmidt AE (1993) Phylogenetic and biogeographic implications of chloroplast DNA variation in Picea. Nord J Bot 13:233–246
Soares MB, Bonaldo MF, Jelene, P, Su L, Lawton L, Efstratiadis A (1994) Construction and characterization of a normalized cDNA library. Proc Natl Acad Sci USA 91:9228–9232
Squirrell J, Hollingsworth PM, Woodhead M, Russell J, Lowe AJ, Gibby M, Powell W (2003) How much effort is required to isolate nuclear microsatellites from plants? Mol Ecol 12:1339–1348
Ven WTG van de, McNicol RJ (1996) Microsatellites as DNA markers in Sitka spruce. Theor Appl Genet 93:613–617
Vigouroux Y, McMullen M, Hittinger CT, Houchins K, Schulz L, Kresovich S, Matsuoka Y, Doebley J (2002) Identifying genes of agronomic importance in maize by screening microsatellites for evidence of selection during domestication Proc Natl Acad Sci USA 99:9650–9655
Acknowledgements
Genome Canada and the Province of British Columbia, through the Genome BC Forestry Genome Project, funded this research. We acknowledge the support of the Vancouver Genome Sciences Centre for EST sequencing and database development. We thank Dr. Sally Aitken (University of British Columbia) for the white spruce collections, Washington Gapare (University of British Columbia) for the Sitka spruce collections, and Dr. Om Rajora (Dalhousie University) for the black spruce collections. Dr. Barry Jaquish of the B.C. Ministry of Forests Kalamalka research station provided the spruce species collection.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by O. Savolainen
Rights and permissions
About this article
Cite this article
Rungis, D., Bérubé, Y., Zhang, J. et al. Robust simple sequence repeat markers for spruce (Picea spp.) from expressed sequence tags. Theor Appl Genet 109, 1283–1294 (2004). https://doi.org/10.1007/s00122-004-1742-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-004-1742-5