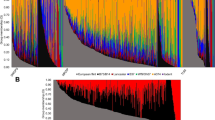
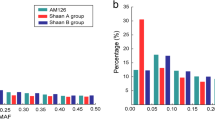
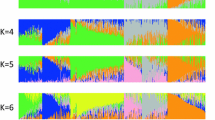
Abstract
Recent progress in genotyping and resequencing techniques have opened new opportunities for deciphering quantitative trait variation by looking for associations between traits of interest and polymorphisms in panels of diverse inbred lines. Association mapping raises specific issues related to the choice of appropriate (i) panels and marker-densities and (ii) statistical methods to capture associations. In this study, we used a panel of 314 maize inbred lines from the dent pool, composed of inbred material from public institutes (113 inbred lines) and a private company (201 inbred lines). We showed that local LD was higher and genetic diversity lower in the material of private origin than in the public material. We compared the results obtained by different software for identifying population structure and computing relatedness among lines, and ran association tests for earliness related traits. Our results confirmed the importance of the mite polymorphism of Vgt1 on flowering time, but also showed that its effect can be captured by zmRap2.7 polymorphisms located 70 kb apart. We also highlighted associations with polymorphisms within genes putatively involved in lignin biosynthesis pathway, which deserve further investigations.




Similar content being viewed by others
References
Andersen J, Zein I, Wenzel G, Darnhofer B, Eder J, Ouzunova M, Luebberstedt T (2008) Characterization of phenylpropanoid pathway genes within European Maize (Zea mays L.) inbreds. BMC Plant Biol 8(1):2
Aranzana MJ, Kim S, Zhao K, Bakker E, Horton M, Jakob K (2005) Genome-wide association mapping in Arabidopsis identifies previously known flowering time and pathogen resistance genes. PLoS Genet 1(5):e60
Atwell S, Huang YS, Vilhjalmsson BJ, Willems G, Horton M, Li Y, Meng D, Platt A, Tarone AM, Hu TT, Jiang R, Muliyati NW, Zhang X, Amer MA, Baxter I, Brachi B, Chory J, Dean C, Debieu M, de Meaux J, Ecker JR, Faure N, Kniskern JM, Jones JDG, Michael T, Nemri A, Roux F, Salt DE, Tang C, Todesco M, Traw MB, Weigel D, Marjoram P, Borevitz JO, Bergelson J, Nordborg M (2010) Genome-wide association study of 107 phenotypes in Arabidopsis thaliana inbred lines. Nature 465(7298):627–631
Bar-Hen A, Charcosset A, Bourgoin M, Guiard J (1995) Relationship between genetic markers and morphological traits in a Maize inbred lines collection. Euphytica 84(2):145–154
Barrière Y, Riboulet C, Méchin V, Maltese S, Pichon M, Cardinal A, Lapierre C, Martinant JP (2007) Genetics and genomics of lignification in grass cell walls based on Maize as model species. Genes Genom Genomics 1(2):133–156
Beló A, Zheng P, Luck S, Shen B, Meyer DJ, Li B, Tingey S, Rafalski A (2007) Whole genome scan detects an allelic variant of fad2 associated with increased oleic acid levels in Maize. Mol Genet Genomics 279(1):1–10
Bernardo R, Yu J (2007) Prospects for genome-wide selection for quantitative traits in Maize. Crop Sci 47(3):1082–1090
Bradbury PJ, Zhang Z, Kroon DE, Casstevens TM, Ram-doss Y, Buckler ES (2007) TASSEL: software for association mapping of complex traits in diverse samples. Bioinformatics 23(19):2633–2635
Buckler ES, Holland JB, Bradbury PJ, Acharya CB, Brown PJ, Browne C, Ersoz E, Flint-Garcia S, Garcia A, Glaubitz JC, Goodman MM, Harjes C, Guill K, Kroon DE, Larsson S, Lepak NK, Li H, Mitchell SE, Pressoir G, Peiffer JA, Rosas MO, Rocheford TR, Cinta Romay M, Romero S, Salvo S, Sanchez Villeda H, da Silva HS, Sun Q, Tian F, Upadyayula N, Ware D, Yates H, Yu J, Zhang Z, Kresovich S, McMullen MD (2009) The genetic architecture of Maize flowering time. Science 325(5941):714–718
Butler D, Cullis BR, Gilmour AR, Gogel BJ (2007) ASReml-R estimates variance components under a general linear mixed model by residual maximum likelihood (REML). Analysis of mixed models for S language environments. DPI and F Publications, Queensland
Camus-Kulandaivelu L, Veyrieras JB, Madur D, Combes V, Fourmann M, Barraud S, Dubreuil P, Gouesnard B, Manicacci D, Charcosset A (2006) Maize adaptation to temperate climate: relationship between population structure and polymorphism in the dwarf 8 gene. Genetics 172(4):2449–2463
Chardon F, Virlon B, Moreau L, Falque M, Joets J, Decousset L, Murigneux A, Charcosset A (2004) Genetic architecture of flowering time in Maize as inferred from quantitative trait loci meta-analysis and synteny conservation with the rice genome. Genetics 168(4):2169–2185
Chen Y, Zein I, Brenner EA, Andersen JR, Landbeck M, Ouzunova M, Lübberstedt T (2010) Polymorphisms in monolignol biosynthetic genes are associated with biomass yield and agronomic traits in European Maize (Zea mays L.). BMC Plant Biol 10(1):12
Ching A, Caldwell KS, Jung M, Dolan M, Smith OSH, Tingey S, Morgante M, Rafalski A (2002) SNP frequency, haplotype structure and linkage disequilibrium in elite Maize inbred lines. BMC Genet 3(1):19
Churchill GA, Doerge RW (1994) Empirical threshold values for quantitative trait mapping. Genetics 138(3):963–971
Dray S, Dufour AB (2007) The ade4 package: implementing the duality diagram for ecologists. J Stat Softw 22(4):1–20
Ducrocq S, Madur D, Veyrieras JB, Camus-Kulandaivelu L, Kloiber-Maitz M, Presterl T, Ouzunova M, Manicacci D, Charcosset A (2008) Key impact of Vgt1 on flowering time adaptation in Maize: evidence from association mapping and ecogeographical information. Genetics 178(4):2433–2437
Eckert AJ, van Heerwaarden J, Wegrzyn JL, Nelson CD, Ross-Ibarra J, Gonzalez-Martinez SC, Neale DB (2010) Patterns of population structure and environmental associations to aridity across the range of loblolly pine (Pinus taeda L., Pinaceae). Genetics 185(3):969–982
Elshire RJ, Glaubitz JC, Sun Q, Poland JA, Kawamoto K, Buckler ES, Mitchell SE (2011) A robust, simple genotyping-by-sequencing (GBS) approach for high diversity species. PLoS ONE 6(5):e19379
Ersoz ES, Yu JM, Buckler ES (2009) Applications of linkage disequilibrium and association mapping in Maize. In: A.L.Kriz BAL (ed) Biotechnology in agriculture and forestry-molecular genetic approaches to Maize improvement, vol 63. Springer-Verlag, Berlin Heidelberg
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol Ecol 14(8):2611–2620
Evoldir community (2008) Evoldir-month in review. http://evol.mcmaster.ca/evoldir.html
Flint-Garcia SA, Thornsberry JM, Buckler ES (2003) Structure of linkage disequilibrium in plants. Annu Rev Plant Biol 54(1):357–374
Guillaumie S, San-Clemente H, Deswarte C, Martinez Y, Lapierre C, Murigneux A, Barrière Y, Pichon M, Goffner D (2007) MAIZEWALL database and developmental gene expression profiling of cell wall biosynthesis and assembly in Maize. Plant Physiol 143(1):339–363
Gunderson KL, Kruglyak S, Graige MS, Garcia F, Kermani BG, Zhao C, Che D, Dickinson T, Wickham E, Bierle J, Doucet D, Milewski M, Yang R, Siegmund C, Haas J, Zhou L, Oliphant A, Fan J-B, Barnard S, Chee MS (2004) Decoding randomly ordered DNA arrays. Genome Res 14(5):870–877
Hamblin MT, Warburton ML, Buckler ES (2007) Empirical comparison of simple sequence repeats and single nucleotide polymorphisms in assessment of Maize diversity and relatedness. PLoS ONE 2(12):e1367
Hardy OJ, Vekemans X (2002) SPAGeDi: a versatile computer program to analyse spatial genetic structure at the individual or population levels. Mol Ecol Notes 2(4):618–620
Jombart T, Devillard S, Balloux F (2010) Discriminant analysis of principal components: a new method for the analysis of genetically structured populations. BMC genomics 11(1):94
Jung M, Ching A, Bhattramakki D, Dolan M, Tingey S, Morgante M, Rafalski A (2004) Linkage disequilibrium and sequence diversity in a 500-kbp region around the adh1 locus in elite Maize germplasm. Theor Appl Genet 109(4):681–689
Kang HM, Zaitlen NA, Wade CM, Kirby A, Heckerman D, Daly MJ, Eskin E (2008) Efficient control of population structure in model organism association mapping. Genetics 178(3):1709–1723
Librado P, Rozas J (2009) DnaSP v5: a software for comprehensive analysis of DNA polymorphism data. Bioinformatics 25:1451–1452
Liu K, Muse SV (2005) PowerMarker: an integrated analysis environment for genetic marker analysis. Bioinformatics 21(9):2128–2129
Loiselle BA, Sork VL, Nason J, Graham C (1995) Spatial genetic structure of a tropical understory shrub, Psychotria officinalis (Rubiaceae). Am J Bot 82(11):1420–1425
Lorenz AJ, Coors JG, Hansey CN, Kaeppler SM, De Leon N (2010) Genetic analysis of cell wall traits relevant to cellulosic ethanol production in Maize (Zea mays L.). Crop Sci 50(3):842–852
Lu Y, Yan J, Guimarães C, Taba S, Hao Z, Gao S, Chen S, Li J, Zhang S, Vivek B, Magorokosho C, Mugo S, Makumbi D, Parentoni S, Shah T, Rong T, Crouch J, Xu Y (2009) Molecular characterization of global Maize breeding germplasm based on genome-wide single nucleotide polymorphisms. Theor Appl Genet 120(1):93–115
Lübberstedt T, Zein I, Andersen J, Wenzel G, Krützfeldt B, Eder J, Ouzunova M, Chun S (2005) Development and application of functional markers in Maize. Euphytica 146(1):101–108
Mantel N (1967) The detection of disease clustering and a generalized regression approach. Cancer Res 27(2):209–220
Meuwissen THE, Hayes BJ, Goddard ME (2001) Prediction of total genetic value using genome-wide dense marker maps. Genetics 157(4):1819–1829
Mezmouk S, Dubreuil P, Bosio M, Décousset L, Charcosset A, Praud S, Mangin B (2011) Effect of population structure corrections on the results of association mapping tests in complex Maize diversity panels. Theor Appl Genet 122(6):1149–1160
Mikel MA (2008) Genetic diversity and improvement of contemporary proprietary North American dent corn. Crop Sci 48(5):1686–1695
Mikel MA, Dudley JW (2006) Evolution of North American dent corn from public to proprietary germplasm. Crop Sci 46(3):1193–1205
Müller BU, Stich B, Piepho HP (2011) A general method for controlling the genome-wide type I error rate in linkage and association mapping experiments in plants. Heredity 106(5):825–831
Myles S, Peiffer JA, Brown JB, Ersoz ES, Zhang Z, Costich DE, Buckler ES (2009) Association mapping: critical considerations shift from genotyping to experimental design. Plant Cell 21(8):2194–2202
Patterson N, Price AL, Reich D (2006) Population structure and Eigen analysis. PLoS Genet 2(12):e190
Porto Neto LR, Bunch RJ, Harrison BE, Barendse W (2010) DNA variation in the gene ELTD1 is associated with tick burden in cattle. Anim Genet 42(1):50–55
Price AL, Patterson NJ, Plenge RM, Weinblatt ME, Shadick NA, Reich D (2006) Principal components analysis corrects for stratification in genome-wide association studies. Nat Genet 38(8):904–909
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155(2):945–959
R Development Core Team (2011) R: a language and environment for statistical computing. 3 R Foundation for Statistical Computing
Rafalski JA (2002) Novel genetic mapping tools in plants: SNPs and LD-based approaches. Plant Sci 162(3):329–333
Remington DL, Thornsberry JM, Matsuoka Y, Wilson LM, Whitt SR, Doebley J, Kresovich S, Goodman MM, Buckler ES (2001) Structure of linkage disequilibrium and phenotypic associations in the Maize genome. PNAS 98(20):11479–11484
Ritland K (1996) Estimators for pairwise relatedness and individual inbreeding coefficients. Genet Res (Camb) 67:175–185
Rousset F (2002) Inbreeding and relatedness coefficients: what do they measure? Heredity 88(5):371–380
Sakamato Y, Kitagawa G (1987) Akaike information criterion statistics. Kluwer Academic Publishers, Norwell
Salvi S, Sponza G, Morgante M, Tomes D, Niu X, Fengler KA, Meeley R, Ananiev EV, Svitashev S, Bruggemann E, Li B, Hainey CF, Radovic S, Zaina G, Rafalski JA, Tingey SV, Miao GH, Phillips RL, Tuberosa R (2007) Conserved noncoding genomic sequences associated with a flowering-time quantitative trait locus in Maize. PNAS 104(27):11376–11381
Salvi S, Castelletti S, Tuberosa R (2009) An updated consensus map for flowering time QTLs in Maize. Maydica 54(4):501–512
SAS (1989) SAS/STAT User’s guide. Version 6, vol 2, 4th edn. SAS Institute Inc., Cary
Sibout R, Plantegenet S, Hardtke CS (2008) Flowering as a condition for xylem expansion in Arabidopsis hypocotyl and root. Curr Biol 18(6):458–463
Staden R, Beal KF, Bonfield JK (1998) The Staden Package. In: Krawetz SMaSA (ed) Computer methods in molecular biology, bioinformatics methods and protocols. vol 132. The Humana Press Inc., Totowa, 115–130
Stich B, Melchinger AE, Frisch M, Maurer HP, Heckenberger M, Reif JC (2005) Linkage disequilibrium in European elite Maize germplasm investigated with SSRs. Theor Appl Genet 111(4):723–730
Storey JD, Tibshirani R (2003) Statistical significance for genome-wide experiments. PNAS 100:9440–9445
Stracke S, Haseneyer G, Veyrieras JB, Geiger HH, Sauer S, Graner A, Piepho HP (2009) Association mapping reveals gene action and interactions in the determination of Flowering time in barley. Theor Appl Genet 118(2):259–273
Tajima F (1989) Statistical method for testing the neutral mutation hypothesis by DNA polymorphism. Genetics 123:585–595
Tenaillon MI, Sawkins MC, Long AD, Gaut RL, Doebley JF, Gaut BS (2001) Patterns of DNA sequence polymorphism along chromosome 1 of Maize (Zea mays ssp. mays L.). PNAS 98(16):9161–9166
Thornsberry JM, Goodman MM, Doebley J, Kresovich S, Nielsen D, Buckler IE (2001) Dwarf8 polymorphisms associate with variation in flowering time. Nat Genet 28:286–289
Truntzler M, Barrière Y, Sawkins M, Lespinasse D, Betran J, Charcosset A, Moreau L (2010) Meta-analysis of QTL involved in silage quality of Maize and comparison with the position of candidate genes. Theor Appl Genet 121(8):1465–1482
Van Inghelandt D, Melchinger AE, Lebreton C, Stich B (2010) Population structure and genetic diversity in a commercial Maize breeding program assessed with SSR and SNP markers. Theor Appl Genet 120(7):1289–1299
Van Inghelandt D, Reif J, Dhillon B, Flament P, Melchinger A (2011) Extent and genome-wide distribution of linkage disequilibrium in commercial Maize germplasm. Theor Appl Genet 123(1):11–20
Vekemans X, Hardy OJ (2004) New insights from fine-scale spatial genetic structure analyses in plant populations. Mol Ecol 13(4):921–935
Veyrieras JB, Camus-Kulandaivelu L, Charcosset A (2006) Etude du déterminisme génétique de caractères quantitatifs chez les végétaux: Méta-analyse de QTL et études d’association. PhD thesis, Agroparistech, Paris
Wilson LM, Whitt SR, Ibanez AM, Rocheford TR, Goodman MM, Buckler IE (2004) Dissection of Maize kernel composition and starch production by candidate gene association. Plant Cell 16(10):2719–2733
Yan J, Shah T, Warburton ML, Buckler ES, McMullen MD, Crouch J (2009) Genetic characterization and linkage disequilibrium estimation of a global Maize collection using SNP markers. PLoS ONE 4(12):e8451
Yang X, Gao S, Xu S, Zhang Z, Prasanna B, Li L, Li J, Yan J (2010) Characterization of a global germplasm collection and its potential utilization for analysis of complex quantitative traits in Maize. Mol Breed. doi:10.1007/s11032-010-9500-7
Yu JM, Buckler ES (2006) Genetic association mapping and genome organization of Maize. Curr Opin Biotechnol 17(2):155–160
Yu JM, Pressoir G, Briggs WH, Bi IV, Yamasaki M, Doebley J, McMullen MD, Gaut BS, Nielsen D, Holland JB, Kresovich S, Buckler ES (2006) A unified mixed-model method for association mapping that accounts for multiple levels of relatedness. Nat Genet 38(2):203–208
Yu J, Holland JB, McMullen MD, Buckler ES (2008) Genetic design and statistical power of nested association mapping in Maize. Genetics 178(1):539–551
Yu J, Zhang J, Zhu C, Tabanao D, Pressoir G, Tuinstra MR, Kresovich S, Todhunter RJ, Buckler ES (2009) Simulation appraisal of the adequacy of number of background markers for relationship estimation in association mapping. The Plant Genome 2:63–77
Zhao K, Aranzana MJ, Kim S, Lister C, Shindo C, Tang C (2007) An Arabidopsis example of association mapping in structured samples. PLoS Genet 3(1):e4
Zhu C, Yu JM (2009) Nonmetric multidimensional scaling corrects for population structure in association mapping with different sample types. Genetics 182(3):875–888
Acknowledgments
We thank Pascal Delage, Philippe Jamin, Denis Coubriche, Sophie Pin, Dominique Denoué and Christoph Mainka for the set up of the trials, traits measurements and harvest as well as Jean-Paul Muller for insightful discussions on maize germplasm and phenotypic analyses. Part of this work was financed by Syngenta Seeds; we thank them as well for the phenotypic and genetic material. We thank the University of Oslo Bioportal for providing CPU time. Part of this work was carried out by using the resources of the Computational Biology Service Unit from Cornell University, which is partially funded by Microsoft Corporation.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M. Bohn.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Truntzler, M., Ranc, N., Sawkins, M.C. et al. Diversity and linkage disequilibrium features in a composite public/private dent maize panel: consequences for association genetics as evaluated from a case study using flowering time. Theor Appl Genet 125, 731–747 (2012). https://doi.org/10.1007/s00122-012-1866-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-012-1866-y