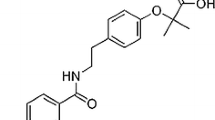
Abstract
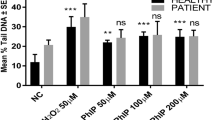
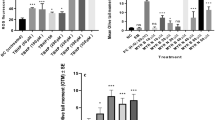
Hemodialysis patients have an elevated genomic damage in peripheral blood lymphocytes (PBLs) and an increased cancer incidence, possibly due to accumulation of uremic toxins like advanced glycation end products (AGEs). Because the vitamin B1 prodrug benfotiamine reduces AGE levels in experimental diabetes, and dialysis patients often suffer from vitamin B1 deficiency, we conducted two consecutive studies supplementing hemodialysis patients with benfotiamine. In both studies, genomic damage was measured as micronucleus frequency of PBLs before and at three time-points after initiation of benfotiamine supplementation. AGE-associated fluorescence in plasma, and in the second study additionally, the antioxidative capacity of plasma was analyzed. Benfotiamine significantly lowered the genomic damage of PBLs in hemodialysis patients of both studies independent of changes in plasma AGE levels. The second study gave a hint to the mechanism, as the antioxidative capacity of the plasma of the treated patients clearly increased, which might ameliorate the DNA damage.


Similar content being viewed by others
References
Agalou S, Ahmed N, Babaei-Jadidi R et al (2005) Profound mishandling of protein glycation degradation products in uremia and dialysis. J Am Soc Nephrol 16:1471–1485
Babaei-Jadidi R, Karachalias N, Ahmed N et al (2003) Prevention of incipient diabetic nephropathy by high-dose thiamine and benfotiamine. Diabetes 52:2110–2120
Basile C (2003) The effect of convection on the nutritional status of haemodialysis patients. Nephrol Dial Transplant 18(Suppl 7):vii46–49 discussion vii58–49
Benzie IF, Strain JJ (1996) The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: the FRAP assay. Anal Biochem 239:70–76
Bierhaus A, Hofmann MA, Ziegler R et al (1998) AGEs and their interaction with AGE-receptors in vascular disease and diabetes mellitus. I. The AGE concept. Cardiovasc Res 37:586–600
Bonassi S, Znaor A, Ceppi M et al (2007) An increased micronucleus frequency in peripheral blood lymphocytes predicts the risk of cancer in humans. Carcinogenesis 28:625–631
Boure T, Vanholder R (2004) Biochemical and clinical evidence for uremic toxicity. Artif Organs 28:248–253
Buemi M, Floccari F, Costa C et al (2006) Dialysis-related genotoxicity: sister chromatid exchanges and DNA lesions in T and B lymphocytes of uremic patients. Genomic damage in patients on hemodiafiltration. Blood Purif 24:569–574
Cengiz K, Block AM, Hossfeld DK et al (1988) Sister chromatid exchange and chromosome abnormalities in uremic patients. Cancer Genet Cytogenet 36:55–67
Ceriello A, Mercuri F, Quagliaro L et al (2001) Detection of nitrotyrosine in the diabetic plasma: evidence of oxidative stress. Diabetologia 44:834–838
Ceylan-Isik AF, Wu S, Li Q et al (2006) High-dose benfotiamine rescues cardiomyocyte contractile dysfunction in streptozotocin-induced diabetes mellitus. J Appl Physiol 100:150–156
Croteau DL, Bohr VA (1997) Repair of oxidative damage to nuclear and mitochondrial DNA in mammalian cells. J Biol Chem 272:25409–25412
Fenech M (2002) Micronutrients and genomic stability: a new paradigm for recommended dietary allowances (RDAs). Food Chem Toxicol 40:1113–1117
Fenech M, Holland N, Chang WP et al (1999) The HUman MicroNucleus Project—an international collaborative study on the use of the micronucleus technique for measuring DNA damage in humans. Mutat Res 428:271–283
Fenech M, Chang WP, Kirsch-Volders M et al (2003) HUMN project: detailed description of the scoring criteria for the cytokinesis-block micronucleus assay using isolated human lymphocyte cultures. Mutat Res 534:65–75
Fenech M, Baghurst P, Luderer W et al (2005) Low intake of calcium, folate, nicotinic acid, vitamin E, retinol, beta-carotene and high intake of pantothenic acid, biotin and riboflavin are significantly associated with increased genome instability–results from a dietary intake and micronucleus index survey in South Australia. Carcinogenesis 26:991–999
Fragedaki E, Nebel M, Schupp N et al (2005) Genomic damage and circulating AGE levels in patients undergoing daily versus standard haemodialysis. Nephrol Dial Transplant 20:1936–1943
Frank T, Bitsch R, Maiwald J et al (2000) High thiamine diphosphate concentrations in erythrocytes can be achieved in dialysis patients by oral administration of benfontiamine. Eur J Clin Pharmacol 56:251–257
Gadau S, Emanueli C, Van Linthout S et al (2006) Benfotiamine accelerates the healing of ischaemic diabetic limbs in mice through protein kinase B/Akt-mediated potentiation of angiogenesis and inhibition of apoptosis. Diabetologia 49:405–420
Glorieux G, Helling R, Henle T et al (2004) In vitro evidence for immune activating effect of specific AGE structures retained in uremia. Kidney Int 66:1873–1880
Hammes HP, Du X, Edelstein D et al (2003) Benfotiamine blocks three major pathways of hyperglycemic damage and prevents experimental diabetic retinopathy. Nat Med 9:294–299
Haupt E, Ledermann H, Kopcke W (2005) Benfotiamine in the treatment of diabetic polyneuropathy–a three-week randomized, controlled pilot study (BEDIP study). Int J Clin Pharmacol Ther 43:71–77
Himmelfarb J, Stenvinkel P, Ikizler TA et al (2002) The elephant in uremia: oxidant stress as a unifying concept of cardiovascular disease in uremia. Kidney Int 62:1524–1538
Hung SC, Hung SH, Tarng DC et al (2001a) Thiamine deficiency and unexplained encephalopathy in hemodialysis and peritoneal dialysis patients. Am J Kidney Dis 38:941–947
Hung SC, Hung SH, Tarng DC et al (2001b) Chorea induced by thiamine deficiency in hemodialysis patients. Am J Kidney Dis 37:427–430
Kan E, Undeger U, Bali M et al (2002) Assessment of DNA strand breakage by the alkaline COMET assay in dialysis patients and the role of Vitamin E supplementation. Mutat Res 520:151–159
Lim PS, Ma YS, Cheng YM et al (2002) Mitochondrial DNA mutations and oxidative damage in skeletal muscle of patients with chronic uremia. J Biomed Sci 9:549–560
Liu CS, Ko LY, Lim PS et al (2001) Biomarkers of DNA damage in patients with end-stage renal disease: mitochondrial DNA mutation in hair follicles. Nephrol Dial Transplant 16:561–565
Maisonneuve P, Agodoa L, Gellert R et al (1999) Cancer in patients on dialysis for end-stage renal disease: an international collaborative study. Lancet 354:93–99
Morena M, Cristol JP, Senecal L et al (2002) Oxidative stress in hemodialysis patients: is NADPH oxidase complex the culprit? Kidney Int Suppl 80:109–114
Morena M, Delbosc S, Dupuy AM et al (2005) Overproduction of reactive oxygen species in end-stage renal disease patients: a potential component of hemodialysis-associated inflammation. Hemodial Int 9:37–46
Munch G, Keis R, Wessels A et al (1997) Determination of advanced glycation end products in serum by fluorescence spectroscopy and competitive ELISA. Eur J Clin Chem Clin Biochem 35:669–677
Nascimento MM, Suliman ME, Murayama Y et al (2006) Effect of high-dose thiamine and pyridoxine on advanced glycation end products and other oxidative stress markers in hemodialysis patients: a randomized placebo-controlled study. J Ren Nutr 16:119–124
Pietrzak I, Baczyk K (1997) Comparison of the thiamine level in blood and erythrocyte transketolase activity in hemodialyzed and nondialyzed patients during recombinant human erythropoietin therapy. Miner Electrolyte Metab 23:277–282
Pomero F, Molinar Min A, La Selva M et al (2001) Benfotiamine is similar to thiamine in correcting endothelial cell defects induced by high glucose. Acta Diabetol 38:135–138
Raj DS, Choudhury D, Welbourne TC et al (2000) Advanced glycation end products: a Nephrologist’s perspective. Am J Kidney Dis 35:365–380
Schinzel R, Munch G, Heidland A et al (2001) Advanced glycation end products in end-stage renal disease and their removal. Nephron 87:295–303
Schleicher ED, Bierhaus A, Haring HU et al (2001) Chemistry and pathobiology of advanced glycation end products. Contrib Nephrol 131:1–9
Schmid U, Stopper H, Heidland A et al (2008) Benfotiamine exhibits direct antioxidative capacity and prevents induction of DNA damage in vitro. Diabetes Metab Res Rev (in press)
Schwenger V, Zeier M, Henle T et al (2001) Advanced glycation endproducts (AGEs) as uremic toxins. Nahrung 45:172–176
Siems WG, Sommerburg O, Grune T (2000) Erythrocyte free radical and energy metabolism. Clin Nephrol 53:S9–17
Stamatas GN, Estanislao RB, Suero M et al (2006) Facial skin fluorescence as a marker of the skin’s response to chronic environmental insults and its dependence on age. Br J Dermatol 154:125–132
Stewart JH, Buccianti G, Agodoa L et al (2003) Cancers of the kidney and urinary tract in patients on dialysis for end-stage renal disease: analysis of data from the United States, Europe, and Australia and New Zealand. J Am Soc Nephrol 14:197–207
Stirban A, Negrean M, Stratmann B et al (2006) Benfotiamine prevents macro- and microvascular endothelial dysfunction and oxidative stress following a meal rich in advanced glycation end products in individuals with type 2 diabetes. Diabetes Care 29:2064–2071
Stopper H, Müller SO (1997) Micronuclei. Biological end point for genotoxicity. Vitro 11:661–667
Stopper H, Meysen T, Bockenforde A et al (1999) Increased genomic damage in lymphocytes of patients before and after long-term maintenance hemodialysis therapy. Am J Kidney Dis 34:433–437
Stopper H, Boullay F, Heidland A et al (2001) Comet-assay analysis identifies genomic damage in lymphocytes of uremic patients. Am J Kidney Dis 38:296–301
Stopper H, Schinzel R, Sebekova K et al (2003) Genotoxicity of advanced glycation end products in mammalian cells. Cancer Lett 190:151–156
Tarng DC, Huang TP, Wei YH et al (2000) 8-hydroxy-2′-deoxyguanosine of leukocyte DNA as a marker of oxidative stress in chronic hemodialysis patients. Am J Kidney Dis 36:934–944
Teschner M, Garte C, Ruckle-Lanz H et al (2002) Incidence and spectrum of malignant disease among dialysis patients in North Bavaria. Dtsch Med Wochenschr 127:2497–2502
Thijssen S, Wystrychowski G, Usvyat L et al (2007) Determinants of serum albumin concentration analyzed in a large cohort of patients on maintenance hemodialysis. J Ren Nutr 17:70–74
Thornalley PJ, Babaei-Jadidi R, Al Ali H et al (2007) High prevalence of low plasma thiamine concentration in diabetes linked to a marker of vascular disease. Diabetologia 50:2164–2170
Vamvakas S, Bahner U, Heidland A (1998) Cancer in end-stage renal disease: potential factors involved -editorial. Am J Nephrol 18:89–95
Vanholder R, De Smet R, Glorieux G et al (2003) Review on uremic toxins: classification, concentration, and interindividual variability. Kidney Int 63:1934–1943
Volkovova K, Barancokova M, Kazimirova A et al (2005) Antioxidant supplementation reduces inter-individual variation in markers of oxidative damage. Free Radic Res 39:659–666
Winkler G, Pal B, Nagybeganyi E et al (1999) Effectiveness of different benfotiamine dosage regimens in the treatment of painful diabetic neuropathy. Arzneim-Forsch 49:220–224
Wu S, Ren J (2006) Benfotiamine alleviates diabetes-induced cerebral oxidative damage independent of advanced glycation end-product, tissue factor and TNF-alpha. Neurosci Lett 394:158–162
Yamagishi S, Takeuchi M, Inagaki Y et al (2003) Role of advanced glycation end products (AGEs) and their receptor (RAGE) in the pathogenesis of diabetic microangiopathy. Int J Clin Pharmacol Res 23:129–134
Zevin D, Malachi T, Gafter U et al (1991) Impaired DNA repair in patients with end-stage renal disease and its improvement with hemodialysis. Miner Electrolyte Metab 17:303–306
Acknowledgement
This study was supported by a grant from the Else Kröner-Fresenius Stiftung. Benfotiamine was donated by Wörwag Pharma GmbH & Co. KG (Böblingen, Germany). We appreciate the technical assistance of Maria Scheurich and Michael Kessler.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schupp, N., Dette, E.M., Schmid, U. et al. Benfotiamine reduces genomic damage in peripheral lymphocytes of hemodialysis patients. Naunyn-Schmied Arch Pharmacol 378, 283–291 (2008). https://doi.org/10.1007/s00210-008-0310-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-008-0310-y