Abstract
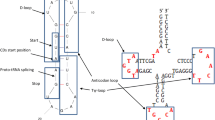
The evolutionary history of the two structural and functional domains of tRNA is controversial but harbors the secrets of early translation and the genetic code. To explore the origin and evolution of tRNA, we reconstructed phylogenetic trees directly from molecular structure. Forty-two structural characters describing the geometry of 571 tRNAs and three statistical parameters describing thermodynamic and mechanical features of molecules quantitatively were used to derive phylogenetic trees of molecules and molecular substructures. Trees of molecules failed to group tRNA according to amino acid specificity and did not reveal the tripartite nature of life, probably due to loss of phylogenetic signal or because tRNA diversification predated organismal diversification. Trees of substructures derived from both structural and statistical characters support the origin of tRNA in the acceptor arm and the hypothesis that the top half domain composed of acceptor and pseudouridine (TΨC) arms is more ancient than the bottom half domain composed of dihydrouridine (DHU) and anticodon arms. This constitutes the cornerstone of the genomic tag hypothesis that postulates tRNAs were ancient telomeres in the RNA world. The trees of substructures suggest a model for the evolution of the major functional and structural components of tRNA. In this model, short RNA hairpins with stems homologous to the acceptor arm of present day tRNAs were extended with regions homologous to TΨC and anticodon arms. The DHU arm was then incorporated into the resulting three-stemmed structure to form a proto-cloverleaf structure. The variable region was the last structural addition to the molecular repertoire of evolving tRNA substructures.






Similar content being viewed by others
References
Ancel LW, Fontana W (2000) Plasticity, evolvability, and modularity in RNA. J Exp Zool (Mol Dev Evol) 288:242–283
Billoud B, Guerrucci MA, Masselot M, Deutsch JS (2000) Cirripede phylogeny using a novel approach: molecular morphometrics. Mol Biol Evol 17:1435–1445
Bloch DP, McArthur B, Mirrop S (1985) tRNA-rRNA sequence homologies: evidence for an ancient modular format shared by tRNAs and rRNAs. Biosystems 17:209–225
Bremer K (1988) The limits of amino acid sequence data in angiosperm phylogenetic reconstruction. Evolution 42:795–803
Bull AT, Goodfellow M, Slater JH (1992) Biodiversity as a source of innovation in biotechnology. Annu Rev Microbiol 46:219–252
Caetano-Anollés G (2001) Novel strategies to study the role of mutation and nucleic acid structure in evolution. Plant Cell Tissue Org Cult 67:115–132
Caetano-Anollés G (2002a) Evolved RNA secondary structure and the rooting of the universal tree of life. J Mol Evol 54:333–345
Caetano-Anollés G (2002b) Tracing the evolution of RNA structure in ribosomes. Nucleic Acids Res 30:2575–2587
Caetano-Anollés G (2005) Grass evolution inferred from chromosomal rearrangements and geometrical and statistical features in RNA structure. J Mol Evol 60:635–652
Collins LJ, Moulton V, Penny D (2000) Use of RNA secondary structure for studying the evolution of RNase P and RNase MRP. J Mol Evol 51:194–2004
Dick TP, Schamel WWA (1995) Molecular evolution of transfer RNA from two precursor hairpins: implications for the origin of protein synthesis. J Mol Evol 41:1–9
Di Giulio M (1992) On the origin of the transfer RNA molecule. J Theor Biol 159:199–214
Di Giulio M (1999) The non-monophyletic origin of the tRNA molecule. J Theor Biol 197:403–414
Di Giulio M (2000) The RNA world, the genetic code and the tRNA molecule. Trends Genet 16:17–18
Eigen M, Winkler-Oswatitsch R (1981) Transfer-RNA, an early gene? Naturwissenschaften 68:282–292
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39:783–791
Felsenstein J (1988) Phylogenies from molecular sequences: inference and reliability. Annu Rev Genet 22:521–565
Fontana W (2002) Modelling ‘evo-devo’ with RNA. BioEssays 24:1164–1177
Gladyshev GP, Ershov YA (1982) Principles of the thermodynamics of biological systems. J Theor Biol 94:301–343
Goodenbour JM, Pan T (2006) Diversity of tRNA genes in eukaryotes. Nucleic Acids Res 34:6137–6146
Gultyaev PA, van Batenburg FHD, Pleij CWA (2002) Selective pressures on RNA hairpins in vivo and in vitro. J Mol Evol 54: 1–8
Higgs PG (1993) RNA secondary structure: a comparison of real and random sequences. J Phys I France 3:43–59
Higgs PG (1995) Thermodynamic properties of transfer RNA: a computational study. J Chem Soc Faraday Trans 91:2531–2540
Higgs PG (2000) RNA secondary structure: physical and computational aspects. Quarterly Rev Biophys 33:199–253
Hillis DM, Huelsenbeck JP (1992) Signal, noise, and reliability in molecular phylogenetic analyses. J Hered 83:189–195
Hopfield JJ (1978) Origin of the genetic code: a testable hypothesis based on tRNA structure, sequence, and kinetic proofreading. Proc Natl Acad Sci USA 75:4334–4338
Maddison WP, Maddison DR (2003) MacClade 4: analysis of phylogeny and character evolution, version 4.06. Sinauer Associates, Sunderland, MA
Maizels N, Weiner AM (1994) Phylogeny from function: evidence from the molecular fossil record that tRNA originated in replication, not translation. Proc Natl Acad Sci USA 91:6729–6734
Marck C, Kachouri-Lafond R, Lafontaine I, Westhof E, Dujon B, Grosjean H (2006) The RNA polymerase III-dependent family of genes in hemiascomycetes: comparative RNomics, decoding strategies, transcription and evolutionary implications. Nucleic Acids Res 34:1816–1835
Marlière P (1983) Computer building and folding of fictitious transfer-RNA sequences. Biochimie 65:267–273
Martinis SA, Schimmel P (1995) Small RNA oligonucleotide substrate for specific aminoacylations. In: Söll D, RajBandary V (eds) tRNA: structure, biosynthesis, and function. ASM Press, Washington, DC, pp 349–370
Muller AWJ (2005) Thermosynthesis as energy source for the RNA world: a model for the bioenergetics of the origin of life. Biosystems 82:93–102
Nagaswamy U, Fox G (2003) RNA ligation and the origin of tRNA. Origins Life Evol B 33:199–209
Pollock D (2003) The Zuckerkandl Prize: structure and evolution. J Mol Evol 56:375–376
Randau L, Münch R, Hohn MJ, Jahn D, Söll D (2005) Nanoarchaeum equitans creates functional tRNAs from separate genes for their 5′- and 3′-halves. Nature 433:537–541
Rodin S, Ohno S, Rodin A (1993) Transfer RNAs with complementary anticodons: Could they reflect early evolution of discriminative genetic code adaptors? Proc Natl Acad Sci USA 90:4723–4727
Rodin S, Rodin A, Ohno S (1996) The presence of codon-anticodon pairs in the acceptor stem of tRNAs. Proc Natl Acad Sci USA 93:4537–4542
Rodin SN, Rodin AS (2006a) Origin of the genetic code: first aminoacyl-tRNA systhetases could replace isofunctional ribozymes when only the second base of codons was established. DNA Cell Biol 25:365–375
Rodin SN, Rodin AS (2006b) Partitioning of aminoacyl-tRNA synthetases in two classes could have been coded in a strand-symmetric RNA world. DNA Cell Biol 25:617–626
Schimmel P, Giegé R, Moras D, Yokoyama S (1993) An operational RNA code for amino acids and possible relationship to genetic code. Proc Natl Acad Sci USA 90:8763–8768
Schimmel P, Ribas de Pouplana L (1995) Transfer RNA: from minihelix to genetic code. Cell 81:983–986
Schultes EA, Bartel DP (2000) One sequence, two ribozymes: implications for the emergence of new ribozyme folds. Science 289:448–452
Schultes EA, Hraber PT, LaBean TH (1999) Estimating the contributions of selection and self-organization in RNA secondary structure. J Mol Evol 49:76–83
Selmer M, Dunham CM, Murphy FV IV, Weixlbaumer A, Petry S, Kelley AC, Weir JR, Ramakrishnan V (2006) Structure of the 70S ribosome complexed with mRNA and tRNA. Science 313:1935–1942
Shi H, Moore PB (2000) The crystal structure of yeast phenylalanine tRNA at 1.93 Å resolution: a classic structure revisited. RNA 6:1091–1105
Sprinzl M, Vassilenko KS (2005) Compilation of tRNA sequences and sequences of tRNA genes. Nucleic Acids Res 33:D139–D140
Steel M, Penny D (2000) Parsimony, likelihood, and the role of models in molecular phylogenetics. Mol Biol Evol 17:839–850
Steffens W, Digby D (1999) mRNA have greater negative folding free energies than shuffled or codon choice randomized sequences. Nucleic Acids Res 27:1578–1584
Stegger G, Hofman H, Fortsch J, Gross HJ, Randles JW, Sanger HL, Riesner D (1984) Conformational transitions in viroids and virusoids: comparison of results from energy minimization algorithm and from experimental data. J Biomol Struct Dynam 2:543–571
Steinberg S, Misch A, Sprinzl M (1993) Compilation of tRNA sequences and sequences of tRNA genes. Nucleic Acids Res 21:3011–3015
Sun F-J, Fleurdépine S, Bousquet-Antonelli C, Caetano-Anollés G, Deragon J-M (2007) Common evolutionary trends for tRNA-derived SINE RNA structures. Trends Genet 23:26–33
Swain TD, Taylor DJ (2003) Structural rRNA characters support monophyly of raptorial limbs and paraphyly of limb specialization in water fleas. Proc R Soc London B 270:887–896
Swofford DL (2002) PAUP*: Phylogenetic Analysis Using Parsimony (*and other methods), version 4.0b10. Sinauer Associates, Sunderland, MA
Szathmáry E (1999) The origin of the genetic code: amino acids as cofactors in an RNA world. Trends Genet 15:223–229
Tanaka T, Kikuchi Y (2001) Origin of the cloverleaf shape of transfer RNA - the double-hairpin model: implication for the role of tRNA intron and the long extra loop. Viva Origino 29:134–142
Wang M, Caetano-Anollés G (2006) Global phylogeny determined by the combination of protein domains in proteomes. Mol Biol Evol 23:2444–2454
Wang M, Yafremava LS, Caetano-Anollés D, Mittenthal JE, Caetano-Anollés G (2007) Reductive evolution of architectural repertoires in proteomes and the birth of the tripartite world. Genome Res 17:1572–1585
Weiner AM, Maizels N (1987) tRNA-like structures tag the 3′ ends of genomic RNA moleculesfor replication: implications for the origin of protein synthesis. Proc Natl Acad Sci USA 84:7383–7387
Weiner AM, Maizels N (1999) The genomic tag hypothesis: modern viruses as molecular fossils of ancient strategies for genomic replication, and clues regarding the origin of protein synthesis. Biol Bull 196:327–330
Widmann J, Di Giulio M, Yarus M, Knight R (2005) tRNA creation by hairpin duplication. J Mol Evol 61:524–535
Woese CR (1969) The biological significance of the genetic code. Prog Mol Subcell Biol 1:5–46
Yusupov MM, Yusupov GZ, Baucom A, Lieberman K, Earnest TN, Cate JHD, Noller HF (2001) Crystal structure of the ribosome at 5.5 Å resolution. Science 292:883–895
Zhu W, Freeland S (2006) The standard genetic code enhances adaptive evolution of proteins. J Theor Biol 239:63–70
Acknowledgments
We thank Hee Shin Kim, Ajith Harish, Minglei Wang, and Jay Mittenthal for helpful discussions, Joe Felsenstein for comments on phylogenetic reconstruction, Paul Schimmel for suggestions, Sergei Rodin for many critical comments an suggestions that improved the manuscript, and Vegeir Knudsen for writing stoat, a program that codes statistical characters. Both authors designed and performed research, analyzed data, and wrote the paper. This work was supported by National Science Foundation Grant MCB-0343126 (to G.C.A.) and the Critical Research Initiative of the University of Illinois. Any opinions, findings, and conclusions and recommendations expressed in this material are those of the authors and do not necessarily reflect the views of the funding agencies.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Sun, FJ., Caetano-Anollés, G. The Origin and Evolution of tRNA Inferred from Phylogenetic Analysis of Structure. J Mol Evol 66, 21–35 (2008). https://doi.org/10.1007/s00239-007-9050-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00239-007-9050-8