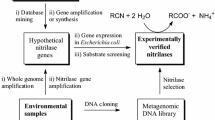
Abstract
Biocatalytic transformations converting aromatic and arylaliphatic nitriles into the analogous related amide or acid were investigated. These studies included synthesis of the β-substituted nitrile 3-hydroxy-3-phenylpropionitrile, subsequent enrichment and isolation on this substrate of nitrile-degrading microorganisms from the environment, and a comparative study of enzymatic reactions of nitriles by resting cell cultures and enzymes. Each biocatalyst exhibited a distinctive substrate selectivity profile, generally related to the length of the aliphatic chain of the arylaliphatic nitrile and the position of substituents on the aromatic ring or aliphatic chain. Cell-free nitrilases generally exhibited a narrower substrate range than resting whole cells of Rhodococcus strains. The Rhodococcus strains all exhibited nitrile hydratase activity and converted β-hydroxy nitriles (but did not demonstrate enantioselectivity on this substrate). The biocatalysts also mediated the synthesis of a range of α-hydroxy carboxylic acids or amides from aldehydes in the presence of cyanide. The use of an amidase inhibitor permits halting the nitrile hydratase/amidase reaction at the amide intermediate.






Similar content being viewed by others
References
Almatawah QA, Cramp R, Cowan DA (1999) Characterisation of an inducible nitrilase from a thermophilic bacillus. Extremophiles 3:283–291
Banerjee A, Sharma R, Banerjee UC (2002) The nitrile-degrading enzymes: current status and future prospects. Appl Microbiol Biotechnol 60: 33–44
Bauer R, Knackmuss H, Stolz A (1998) Enantioselective hydration of 2-arylpropionitriles by a nitrile hydratase from Agrobacterium tumefaciens strain d3. Appl Microbiol Biotechnol 49: 89–95
Bradford MM (1976) Rapid and sensitive method for quantification of microgram quantities of protein utilizing the principle dye binding. Anal Biochem 72:248–254
Beard T, Cohen MA, Parratt JS, Turner NJ, Crosby J, Moillet J (1993) Stereoselective hydrolysis of nitriles and amides under mild conditions using whole cell catalyst. Tetrahedron Asym 4 1085–1104
Blakey A, Colby J, Williams E, O’Reilly C (1995) Regio- and stereo-specific nitrile hydratase from Rhodococcus AJ270. FEMS Microbiol Lett 129: 47–62
Bunch A (1998) Biotransformation of nitriles by rhodococci. Antonie van Leeuwenhoek 74:89–97
Cohen M, Sawden J, Turner NJ (1990) Selective hydrolysis of nitriles under mild conditions by an enzyme. Tetrahedron Lett 31:7223–7226
Colby J, Snell D, Black GW (2000) Immobilization of Rhodococcus AJ270 and use of entrapped biocatalyst for the production of acrylic acid. Chemical Monthly (Monatshefte für Chemie) 131:655–666
Dadd MR, Claridge TDW, Pettman AJ, Knowles CJ (2001a) Biotransformation of benzonitrile to benzohydroxamic acid by Rhodococcus rhodochrous in the presence of hydroxylamine. Biotechnol Lett 23: 221–225
Dadd MR, Claridge TDW, Walton R, Pettman AJ, Knowles CJ (2001b) Regioselective biotransformation of the dinitrile compounds 2-, 3- and 4-(cyanomethyl) benzonitrile by the soil bacterium Rhodococcus rhodochrous LL100–21. Enz Microbial Technol 29:20–27
DeSantis G, Zhu Z, Greenberg WA, Wong K, Chaplin J, Hanson SR, Farwell B, Nicholson LW, Rand CL, Weiner DP, Robertson DE, Burk MJ (2002) An enzyme library approach to biocatalysis: development of nitrilases for enantioselective production of carboxylic acid derivatives. J Am Chem Soc 124:9024–9025
Effenberger F, Graef BW (1998) Chemo- and enantioselective hydrolysis of nitriles and acid amides, respectively, with resting cells of Rhodococcus sp C3II and Rhodococcus erythropolis MP50. J Biotechnol 60:165–174
Effenberger F, Osswald S (2001a) Enantioselective hydrolysis of (R,S)-2-fluoroacetonitriles using nitrilase from Arabidopsis thaliana. Tetrahedron Asym 12:279–285
Effenberger F, Osswald S (2001b) (E)-Selective hydrolysis of (E,Z)-α,β-unsaturated nitriles by the recombinant nitrilase AtNIT1 from Arabidopsis thaliana. Tetrahed Asym 12:2581–2587
Faber K (1992) Biotransformations in organic chemistry. Springer, Berlin Heidelberg New York, pp 113–134
Fallon RD, Stieglitz B, Turner I (1997) A Pseudomonas putida capable of enantioselective hydrolysis of nitriles. Appl Microbiol Biotechnol 47:156–161
Gavagan JE, Fager SK, Fallon RD, Folsom PW, Herkes FE, Eisenberg A, Hann EC, DiCosimo R (1998) Chemoenzymatic production of lactams from aliphatic α,ω-dinitriles. J Org Chem 63:4792–4801
Hirrlinger B, Stolz A, Knackmuss H-J (1997) Formation of a chiral hydroxamic acid with an amidase from Rhodococcus erythropolis MP50 and subsequent chemical Lossen rearrangement to a chiral amine. Appl Environ Microbiol 63:3390–3393
Kamal A, Khanna GBR (2001) A facile preparation of (±)-β-hydroxy nitriles and their enzymatic resolution with lipases. Tetrahed Asym 12:405–410
Klempier N, de Raadt A, Faber K, Griengl H (1991) Selective transformation of nitriles into amides and carboxylic acids by an immobilized nitrilase. Tetrahedron Lett 32:341–344
Kobayashi M, Goda M, Shimizu S (1999) Hydrazide synthesis: novel substrate specificity of amidase. Biochem Biophys Res Comm 256:415–418
Layh N, Hirrlinger B, Stolz A, Knackmuss H-J (1997) Enrichment strategies for nitrile-hydrolysing bacteria. Appl Microbiol Biotechnol 47:668–674
Layh N, Parratt J, Willetts A (1998) Characterization and partial purification of an enantioselective arylacetonitrilase from Pseudomonas fluorescens DSM 7155. J Mol Catalysis B: Enzymatic 5:467–474
Martínková L, Krěn V (2002) Nitrile-and amide-converting microbial enzymes: stereo-, regio- and chemoselectivity. Biocat Biotrans 20:73–93
Martínková L, Stolz A, Knackmuss HJ (1996) Enantioselectivity of the nitrile-hydratase from Rhodococcus equi A4 towards substituted (R,S)-2-arylpropionitriles. Biotechnol Lett 18:1073–76
Osswald S, Wajant H, Effenberger F (2002) Characterization and synthetic applications of recombinant AtNIT1 from Arabidopsis thaliana. Eur J Biochem 269:680–687
Payne MS, Wu S, Fallon RD, Tudor G, Stieglitz B, Turner IM and Nelson MJ (1997) A stereoselective cobalt-containing nitrile hydratase. Biochemistry 36:5447–5454
Prepechalová I, Martínková L, Stolz A, Ovesna M, Bezouska K, Kopecky J, Krěn V (2001) Purification and characterization of the enantioselective nitrile hydratase from Rhodococcus equi A4. Appl Microbiol Biotechnol 55:150–156
Sugai T, Yamakazi T, Yokoyama M, Ohta H (1997) Biocatalysis in organic synthesis: the use of nitrile- and amide-hydrolyzing microorganisms. Biosci Biotech Biochem 61:1419–1427
Taylor SK, Chmiel NH, Simons LJ, Vyvyan JR (1996) Conversion of hydroxy nitriles to lactones using Rhodococcus rhodochrous whole cells. J Org Chem. 61:9084–9085
Vogel AI (1989) In: Furniss BS, Hannaford AJ, Smith PWG, Tatchell AR (eds) Vogel’s textbook of practical organic chemistry, 5th edn. Longman Scientific and Technical, New York pp 747, 753
Wegman MA, Heinemann U, Stolz A, van Rantwijk F, Sheldon RA (2000) Stereoretentive nitrile hydratase-catalysed hydration of D-phenylglycine nitrile. Org Process Res Dev 4:318–322
Wegman MA, Heinemann U, van Rantwijk F, Stolz A, Sheldon RA (2001) Hydrolysis of d,l-phenylglycine nitrile by new bacterial cultures. J Mol Catal B: Enzymatic 11: 249–253
Weiner DP, Chaplin JA (2000) Methods for producing enantiomerically pure α-substituted carboxylic acids. Patent WO 01/48175
Wieser M, Nagasawa T (2000) Stereoselective nitrile-converting enzymes. In: Patel RN (ed) Stereoselective biocatalysis. Marcel Dekker, New York, pp 461–486
Wieser M, Takeuchi K, Wada Y, Yamada H, Nagasawa T (1998) Low-molecular-mass nitrile hydratase from Rhodococcus rhodochrous J1: purification, substrate specificity and comparison with analogous high-molecular-mass enzyme. FEMS Microbiol Lett 169:17–22
Yamamoto K, Oishi K, Fujimatsu I, Komatsu K-I (1991) Production of R-(-)-mandelic acid from mandelonitrile by Alcaligenes faecalis ATCC 8750. Appl Environ Microbiol 57:3028–3032
Acknowledgements
The authors would like to express their appreciation to Dr Maryke Henton, of Onderstepoort Veterinary Institute’s Bacteriology Laboratory, South Africa, for assistance with microbial taxonomy; Dr Gert Marais for additional microbial strains, and Dr Andreas Bommarius for suggesting target compounds. We would also like to thank Jülich Fine Chemicals (Germany), Novozymes (Denmark), BioCatalytics Inc. (USA), and Prof. Lutz Fischer of the Institute of Food Technology, University of Stuttgart-Hohenheim for donation of biocatalysts. D. Brady would like to express his appreciation to the Delft University of Technology for a fellowship to support this work, to Drs LM van Langen, RK Mitra, and ML Bode for useful discussions, N Wilde and C van der Westhuizen for technical assistance, and Dr Lesley Robertson and staff of the Department of Biotechnology (Delft University of Technology) for use of facilities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Brady, D., Beeton, A., Zeevaart, J. et al. Characterisation of nitrilase and nitrile hydratase biocatalytic systems. Appl Microbiol Biotechnol 64, 76–85 (2004). https://doi.org/10.1007/s00253-003-1495-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-003-1495-0