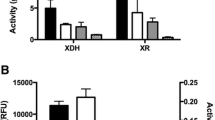
Abstract
The sulfuric acid hydrolysate of lignocellulosic biomass, such as wood chips, from the forest industry is an important material for fuel bioethanol production. In this study, we constructed a recombinant yeast strain that can ferment xylose and cellooligosaccharides by integrating genes for the intercellular expressions of xylose reductase and xylitol dehydrogenase from Pichia stipitis, and xylulokinase from Saccharomyces cerevisiae and a gene for displaying β-glucosidase from Aspergillus acleatus on the cell surface. In the fermentation of the sulfuric acid hydrolysate of wood chips, xylose and cellooligosaccharides were completely fermented after 36 h by the recombinant strain, and then about 30 g/l ethanol was produced from 73 g/l total sugar added at the beginning. In this case, the ethanol yield of this recombinant yeast was much higher than that of the control yeast. These results demonstrate that the fermentation of the lignocellulose hydrolysate is performed efficiently by the recombinant Saccharomyces strain with abilities for xylose assimilation and cellooligosaccharide degradation.





Similar content being viewed by others
References
Aristidou A, Penttilä M (2000) Metabolic engineering applications to renewable resource utilization. Curr Opin Biotechnol 11:187–198
Bradford MM (1976) Photometric methods for protein determination. Anal Biochem 72:248–254
Cho KM, Yoo YJ (1999) Novel SSF process for ethanol production from microcrystalline cellulose using δ-integrated recombinant yeast, Saccharomyces cerevisiae L2612δGC. J Microbiol Biotechnol 9:340–345
Claassen PAM, van Lier JB, Lopez Contreras AM, van Niel EMJ, Sijtsma L, Stams AJM, de Vries SS, Weusthuis RA (1999) Utilization of biomass for the supply of energy carriers. Appl Microbiol Biotechnol 52:741–755
Eliasson A, Christensson C, Wahlbom CF, Hahn-Hägerdal B (2000) Anaerobic xylose fermentation by recombinant Saccharomyces cerevisiae carrying XYL1, XYL2, and XKS1 in minimal medium chemostat cultures. Appl Environ Microbiol 66:3381–3386
Farone WA, Cuzens JE (1997) Strong acid hydrolysis of cellulosic and hemicellulosic materials. US Patent 5597714
Farone WA, Cuzens JE (1998) Method of removing silica or silicates from solids resulting from the strong acid hydrolysis of cellulosic and hemicellulosic materials. US Patent 5782982
Fujita Y, Takasaki S, Ueda M, Tanaka A, Okada H, Morikawa Y, Kawaguchi T, Arai M, Fukuda H, Kondo A (2002) Direct and efficient production of ethanol from cellulosic material with a yeast strain displaying cellulolytic enzymes. Appl Environ Microbiol 68:5136–5141
Fujita Y, Ito J, Ueda M, Fukuda H, Kondo H (2004) Synergistic saccharification, and direct fermentation to ethanol, of amorphous cellulose by use of an engineered yeast strain codisplaying three types of cellulolytic enzymes. Appl Environ Microbiol 70:1207–1212
Hahn-Hägerdal B, Wahlbom CF, Gardonyi M, Zyl WH van, Cordero Otero RR, Jönsson LJ (2001) Metabolic engineering of Saccharomyces cerevisiae for xylose utilization. Adv Biochem Eng Biotechnol 73:53–84
Ho NW, Lin FP, Huang S, Andrews PC, Tsao GT (1990) Purification, characterization, and amino terminal sequence of xylose reductase from Candida shehatae. Enzyme Microb Technol 12:33–39
Ho NW, Chen Z, and Brainard AP (1998) Genetically engineered Saccharomyces yeast capable of effective cofermentation of glucose and xylose. Appl Environ Microbiol 64:1852–1859
Ho NW, Chen Z, Brainard AP, Sedlak M (1999) Successful design and development of genetically engineered Saccharomyces yeasts for effective co-fermentation of glucose and xylose from cellulosic biomass to fuel ethanol. Adv Biochem Eng Biotechnol 65:163–192
Jeffries TW, Jin YS (2004) Metabolic engineering for improved fermentation of pentoses by yeasts. Appl Microbiol Biotechnol 63:495–509
Jeppsson M, Johansson B, Hahn-Hägerdal B, Gorwa-Grauslund MF (2002) Reduced oxidative pentose phosphate pathway flux in recombinant xylose-utilizing Saccharomyces cerevisiae strains improves the ethanol yield from xylose. Appl Environ Microbiol 68:1604–1609
Katahira S, Fujita Y, Mizuike A, Fukuda H, Kondo A (2004) Construction of a xylan-fermenting yeast strain through codisplay of xylanolytic enzymes on the surface of xylose-utilizing Saccharomyces cerevisiae cells. Appl Environ Microbiol 70:5407–5414
Lee JK, Bong-Seong K, Sang-Yong K (2003) Cloning and characterization of xyl1 gene, encoding an NADH-pefering xylose reductase from Candida parapsilosis, and its functional expression in Candida tropicalis. Appl Environ Microbiol 69:6179–6188
Lipke PN, Wojciechowicz D, Kurjan J (1989) AGα1 is the structural gene for the Saccharomyces cerevisiae α-agglutinin, a cell surface glycoprotein involved in cell–cell interactions during mating. Mol Cell Biol 9:3155–3165
McMillan JD (1997) Bioethanol production: status and prospects. Renew Energy 10:295–302
Murai T, Ueda M, Kawaguchi T, Arai M, Tanaka A (1998) Assimilation of cellooligosaccharides by a cell surface-engineered yeast expressing β-glucosidase and carboxymethylcellulase from Aspergillus aculeatus. Appl Environ Microbiol 64:4857–4861
Olsson L, Hahn-Hägerdal B (1993) Fermentative performance of bacteria and yeasts in lignocellulose hydrolysate. Proc Biochem 28:249–257
Ryabova OB, Chmil OM, Sibirny AA (2003) Xylose and cellobiose fermentation to ethanol by the thermotolerant methylotrophic yeast Hansenula polymorpha. FEMS Yeast Res 4:157–164
Saha BC (2003) Hemicellulose bioconversion. J Ind Microbiol 30:279–291
Shamanna DK, Sanderson KE (1979) Uptake and catabolism d-xylose in Salmonella typhimurium LT2. J Bacteriol 139:64–70
Smiley KL, Bolen PL (1982) Demonstration of d-xylose reductase and d-xylitol dehydrogenase in Pachysolen tannophilus. Biotecnol Lett 4:607–610
Sonderegger M, Jeppson M, Larsson C, Gorwa-Grauslund MF, Boles E, Olsson L, Spencer-Martins I, Hähn-Hagerdal B, Sauer U (2004) Fermentation performance of engineered and evolved xylose-fermenting Saccharomyces cerevisiae strains. Biotechnol Bioeng 87:90–98
Tajima M, Nogi Y, Fukasawa T (1985) Primary structure of the Saccharomyces cerevisiae GAL7 gene. Yeast 1:67–77
Toivari MH, Aristidou A, Ruohonen L, Penttilä M (2001) Conversion of xylose to ethanol by recombinant Saccharomyces cerevisiae: importance of xylulokinase (XKS1) and oxygen availability. Metab Eng 3:236–249
van Rensburg P, van Zyl WH, Pretorius IS (1998) Engineering yeast for efficient cellulose degradation. Yeast 14:67–76
Verduyn C, van Kleef R, Frank J, Schreuder H, van Dijken JP, Scheffers WA (1985) Properties of the NAD(P)H-dependent xylose reductase from the xylose-fermenting yeast Pichia stipitis. Biochem J 226:669–677
Walfridsson M, Anderlund M, Bao X, Hahn-Hägerdal B (1997) Expression of different levels of enzymes from Pichia stipitisXYL1 and XYL2 genes in Saccharomyces cerevisiae and its effects on product formation during xylose utilization. Appl Microbiol Biotechnol 48:218–224
Watanabe S, Kodaki T, Makino K (2005) Complete reversal coenzyme specificity of xylitol dehydrogenase and increase of thermostability by the introduction of structural zinc. J Biol Chem 280:10340–10349
Acknowledgements
We thank Takeshi Sumiyoshi, JGC corporation, for providing the lignocellulose hydrolysate and Professor Mitsuyoshi Ueda of the Laboratory of Biomacromolecular Chemistry, Department of Applied Biochemistry, Division of Applied Life Sciences, Graduate School of Agriculture, Kyoto University, for his invaluable suggestions. This work was financed by the New Energy and Industrial Technology Development Organization (NEDO), Tokyo, Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Katahira, S., Mizuike, A., Fukuda, H. et al. Ethanol fermentation from lignocellulosic hydrolysate by a recombinant xylose- and cellooligosaccharide-assimilating yeast strain. Appl Microbiol Biotechnol 72, 1136–1143 (2006). https://doi.org/10.1007/s00253-006-0402-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-006-0402-x