Abstract
Purpose
Reporter genes can provide a way of noninvasively assessing gene activity in vivo. However, current reporter gene strategies may be limited by the immunogenicity of foreign reporter proteins, endogenous expression, or unwanted biological activity. We have developed a reporter gene based on carcinoembryonic antigen (CEA), a human protein with limited normal tissue expression.
Methods
To construct a CEA reporter gene for PET, a CEA minigene (N-A3) was fused to the extracellular and transmembrane domains of the human FcγRIIb receptor. The NA3-FcγRIIb recombinant gene, driven by a CMV promoter, was transfected in Jurkat (human T cell leukemia) cells. Expression was analyzed by flow cytometry, immunohistochemistry (IHC), and microPET imaging.
Results
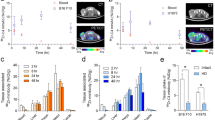
Flow cytometry identified Jurkat clones stably expressing NA3-FcγRIIb at low, medium, and high levels. High and medium NA3-FcγRIIb expression could also be detected by Western blot. Reporter gene positive and negative Jurkat cells were used to establish xenografts in athymic mice. IHC showed staining of the tumor with high reporter gene expression; medium and low N-A3 expression was not detected. MicroPET imaging, using an anti-CEA 124I-labeled single-chain Fv-Fc antibody fragment, demonstrated that only high N-A3 expression could be detected. Specific accumulation of activity was visualized at the N-A3 positive tumor as early as 4 h. MicroPET image quantitation showed tumor activity of 1.8 ± 0.2, 15.2 ± 1.3, and 4.6 ± 1.2 percent injected dose per gram (%ID/g) at 4, 20, and 48 h, respectively. Biodistribution at 48 h demonstrated tumor uptake of 4.8 ± 0.8%ID/g.
Conclusion
The CEA N-A3 minigene has the potential to be used as a reporter gene for imaging cells in vivo.





Similar content being viewed by others
References
Alam J, Cook JL. Reporter genes: application to the study of mammalian gene transcription. Anal Biochem 1990;188(2):245–54.
Serganova I, Blasberg R. Reporter gene imaging: potential impact on therapy. Nucl Med Biol 2005;32(7):763–80.
Tjuvajev JG, Doubrovin M, Akhurst T, Cai S, Balatoni J, Alauddin MM, et al. Comparison of radiolabeled nucleoside probes (FIAU, FHBG, and FHPG) for PET imaging of HSV1-tk gene expression. J Nucl Med 2002;43(8):1072–83.
MacLaren DC, Gambhir SS, Satyamurthy N, Barrio JR, Sharfstein S, Toyokuni T, et al. Repetitive, non-invasive imaging of the dopamine D2 receptor as a reporter gene in living animals. Gene Ther 1999;6(5):785–91.
Ur E, Bomanji J, Mather SJ, Britton KE, Wass JA, Grossman AB, et al. Localization of neuroendocrine tumours and insulinomas using radiolabelled somatostatin analogues, 123I-Tyr3-octreotide and 111In-pentatreotide. Clin Endocrinol (Oxf) 1993;38(5):501–6.
Haberkorn U, Henze M, Altmann A, Jiang S, Morr I, Mahmut M, et al. Transfer of the human NaI symporter gene enhances iodide uptake in hepatoma cells. J Nucl Med 2001;42(2):317–25.
Altmann A, Kissel M, Zitzmann S, Kubler W, Mahmut M, Peschke P, et al. Increased MIBG uptake after transfer of the human norepinephrine transporter gene in rat hepatoma. J Nucl Med 2003;44(6):973–80.
Bonini C, Ferrari G, Verzeletti S, Servida P, Zappone E, Ruggieri L, et al. HSV-TK gene transfer into donor lymphocytes for control of allogeneic graft-versus-leukemia. Science 1997;276(5319):1719–24.
Verzeletti S, Bonini C, Marktel S, Nobili N, Ciceri F, Traversari C, et al. Herpes simplex virus thymidine kinase gene transfer for controlled graft-versus-host disease and graft-versus-leukemia: clinical follow-up and improved new vectors. Hum Gene Ther 1998;9(15):2243–51.
Liang Q, Satyamurthy N, Barrio JR, Toyokuni T, Phelps MP, Gambhir SS, et al. Noninvasive, quantitative imaging in living animals of a mutant dopamine D2 receptor reporter gene in which ligand binding is uncoupled from signal transduction. Gene Ther 2001;8(19):1490–8.
Ajjan RA, Kamaruddin NA, Crisp M, Watson PF, Ludgate M, Weetman AP. Regulation and tissue distribution of the human sodium iodide symporter gene. Clin Endocrinol (Oxf) 1998;49(4):517–23.
Smanik PA, Ryu KY, Theil KS, Mazzaferri EL, Jhiang SM. Expression, exon–intron organization, and chromosome mapping of the human sodium iodide symporter. Endocrinology 1997;138(8):3555–8.
Spitzweg C, Joba W, Eisenmenger W, Heufelder AE. Analysis of human sodium iodide symporter gene expression in extrathyroidal tissues and cloning of its complementary deoxyribonucleic acids from salivary gland, mammary gland, and gastric mucosa. J Clin Endocrinol Metab 1998;83(5):1746–51.
Serganova I, Ponomarev V, Blasberg R. Human reporter genes: potential use in clinical studies. Nucl Med Biol 2007;34(7):791–807.
Dubey P, Su H, Adonai N, Du S, Rosato A, Braun J, et al. Quantitative imaging of the T cell antitumor response by positron-emission tomography. Proc Natl Acad Sci U S A 2003;100(3):1232–7.
Koehne G, Doubrovin M, Doubrovina E, Zanzonico P, Gallardo HF, Ivanova A, et al. Serial in vivo imaging of the targeted migration of human HSV-TK-transduced antigen-specific lymphocytes. Nat Biotechnol 2003;21(4):405–13.
Benchimol S, Fuks A, Jothy S, Beauchemin N, Shirota K, Stanners CP. Carcinoembryonic antigen, a human tumor marker, functions as an intercellular adhesion molecule. Cell 1989;57(2):327–34.
Shively JE, Beatty JD. CEA-related antigens: molecular biology and clinical significance. Crit Rev Oncol Hematol 1985;2(4):355–99.
Toth CA, Thomas P, Broitman SA, Zamcheck N. Receptor-mediated endocytosis of carcinoembryonic antigen by rat liver Kupffer cells. Cancer Res 1985;45(1):392–7.
Raben D, Buchsbaum DJ, Khazaeli MB, Rosenfeld ME, Gillespie GY, Grizzle WE, et al. Enhancement of radiolabeled antibody binding and tumor localization through adenoviral transduction of the human carcinoembryonic antigen gene. Gene Ther 1996;3(7):567–80.
Kenanova V, Olafsen T, Crow DM, Sundaresan G, Subbarayan M, Carter NH, et al. Tailoring the pharmacokinetics and positron emission tomography imaging properties of anti-carcinoembryonic antigen single-chain Fv-Fc antibody fragments. Cancer Res 2005;65(2):622–31.
You YH, Hefta LJ, Yazaki PJ, Wu AM, Shively JE. Expression, purification, and characterization of a two domain carcinoembryonic antigen minigene (N-A3) in Pichia pastoris. The essential role of the N-domain. Anticancer Res 1998;18(5A):3193–201.
Schrewe H, Thompson J, Bona M, Hefta LJ, Maruya A, Hassauer M, et al. Cloning of the complete gene for carcinoembryonic antigen: analysis of its promoter indicates a region conveying cell type-specific expression. Mol Cell Biol 1990;10(6):2738–48.
Brooks DG, Qiu WQ, Luster AD, Ravetch JV. Structure and expression of human IgG FcRII(CD32). Functional heterogeneity is encoded by the alternatively spliced products of multiple genes. J Exp Med 1989;170(4):1369–85.
Olafsen T, Kenanova VE, Wu AM. Tunable pharmacokinetics: modifying the in vivo half-life of antibodies by directed mutagenesis of the Fc fragment. Nat Protoc 2006;1(4):2048–60.
Neumaier M, Shively L, Chen FS, Gaida FJ, Ilgen C, Paxton RJ, et al. Cloning of the genes for T84.66, an antibody that has a high specificity and affinity for carcinoembryonic antigen, and expression of chimeric human/mouse T84.66 genes in myeloma and Chinese hamster ovary cells. Cancer Res 1990;50(7):2128–34.
Neville DM Jr., Scharff J, Srinivasachar K. In vivo T-cell ablation by a holo-immunotoxin directed at human CD3. Proc Natl Acad Sci U S A 1992;89(7):2585–9.
Defrise M, Kinahan PE, Townsend DW, Michel C, Sibomana M, Newport DF. Exact and approximate rebinning algorithms for 3-D PET data. IEEE Trans Med Imaging 1997;16(2):145–58.
Loening AM, Gambhir SS. AMIDE: a free software tool for multimodality medical image analysis. Mol Imaging 2003;2(3):131–7.
Ravetch JV, Bolland S. IgG Fc receptors. Annu Rev Immunol 2001;19:275–90.
Thompson JA, Grunert F, Zimmermann W. Carcinoembryonic antigen gene family: molecular biology and clinical perspectives. J Clin Lab Anal 1991;5(5):344–66.
Berk DA, Yuan F, Leunig M, Jain RK. Direct in vivo measurement of targeted binding in a human tumor xenograft. Proc Natl Acad Sci U S A 1997;94(5):1785–90.
Kenanova V, Olafsen T, Williams LE, Ruel NH, Longmate J, Yazaki PJ, et al. Radioiodinated versus radiometal-labeled anti-carcinoembryonic antigen single-chain Fv-Fc antibody fragments: optimal pharmacokinetics for therapy. Cancer Res 2007;67(2):718–26.
Zalutsky MR, Noska MA, Colapinto EV, Garg PK, Bigner DD. Enhanced tumor localization and in vivo stability of a monoclonal antibody radioiodinated using N-succinimidyl 3-(tri-n-butylstannyl)benzoate. Cancer Res 1989;49(20):5543–9.
Geissler F, Anderson SK, Venkatesan P, Press O. Intracellular catabolism of radiolabeled anti-mu antibodies by malignant B-cells. Cancer Res 1992;52(10):2907–15.
Yaghoubi S, Barrio JR, Dahlbom M, Iyer M, Namavari M, Satyamurthy N, et al. Human pharmacokinetic and dosimetry studies of [(18)F]FHBG: a reporter probe for imaging herpes simplex virus type-1 thymidine kinase reporter gene expression. J Nucl Med 2001;42(8):1225–34.
Voges J, Reszka R, Gossmann A, Dittmar C, Richter R, Garlip G, et al. Imaging-guided convection-enhanced delivery and gene therapy of glioblastoma. Ann Neurol 2003;54(4):479–87.
Gangopadhyay A, Thomas P. Processing of carcinoembryonic antigen by Kupffer cells: recognition of a penta-peptide sequence. Arch Biochem Biophys 1996;334(1):151–7.
Ravetch JV, Kinet JP. Fc receptors. Annu Rev Immunol 1991;9:457–92.
Hulett MD, Witort E, Brinkworth RI, McKenzie IF, Hogarth PM. Identification of the IgG binding site of the human low affinity receptor for IgG Fc gamma RII. Enhancement and ablation of binding by site-directed mutagenesis. J Biol Chem 1994;269(21):15287–93.
Acknowledgments
We thank the UCLA Jonsson Comprehensive Cancer Center and Center for AIDS Research Flow Cytometry Core facility. We are especially grateful to Dr. David Stout, Waldemar Ladno, and Judy Edwards at the University of California Los Angeles for their assistance with the microPET/CT scans. We also acknowledge the assistance of Sofia Loera at the City of Hope Comprehensive Cancer Center Anatomic Pathology Core Facility for performing the immunohistochemistry work. This work was supported by P50 CA 086306; National Institute of Health (NIH) grants CA 043904 and CA 086306; Department of Defense grants DAMD 17-00-1-203 and DAMD 17-00-1-0150. A.M.W., A.C., H.H., and J.B. are members of the UCLA Jonsson Comprehensive Cancer Center (NIH CA 016042).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kenanova, V., Barat, B., Olafsen, T. et al. Recombinant carcinoembryonic antigen as a reporter gene for molecular imaging. Eur J Nucl Med Mol Imaging 36, 104–114 (2009). https://doi.org/10.1007/s00259-008-0921-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-008-0921-z