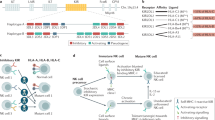
Abstract
Natural killer (NK) lymphocytes represent the first line of defense against virally infected cells and tumor cells. The role of NK cells in immune responses has been markedly explored, mainly due to the identification of NK cell receptors and their ligands, but also through the analysis of mechanisms underlying the effects of various cytokines on NK cell development and function. A population of lymphocytes that shares function and receptors with NK cells is represented by natural killer T (NKT) cells. NKT lymphocytes are regulators of both innate and adaptive immune responses, but have also been reported to function as effector antitumor cells. The marked progress in our understanding of the biology, development, and function of NK/NKT cells has provided the basis for their potential application in tumor clinical trials.
Similar content being viewed by others
References
Trincieri G (1989) Biology of natural killer cells. Adv Immunol 47:187–376
Smyth MJ, Godfrey DI, Trapani JA (2001) A fresh look at tumor immunosurveillance. Nature Immunol 2:293–299
Lanier LL (1998) NK cell receptors. Annu Rev Immunol 16:359–394
Robertson MJ, Ritz J (1990) Biology and clinical relevance of human natural killer cells. Blood 76:2421–2438
Farag SS, Fehniger TA, Ruggeri L, Velardo A,Caligiuri MA (2002) Natural killer cell receptors; new biology and insights into the graft-versus-leukemia effect. Blood 100:1935–1945
Bendelac A, Rivera MN, Park SH, Roark JH (1997) Mouse CD1-specific NK1 T cells: development, specificity, and function. Annu Rev Immunol 15:535–562
Lantz O, Bendelac A (1994) An invariant T cell receptor alpha chain is used by a unique subset of major histocompatibility complex class I-specific CD4+ and CD4-8- T cells in mice and humans. J Exp Med 180:1097–1106
Cui J, Shin T, Kawano T, Sato H, Kondo R, Toura Y, Kaneko H, Koseki Kanno M, Taniguchi M (1997) Requirement for Vα14 NKT cells in IL-12-mediated rejection of tumors. Science 278:1623–1626
Kawano TJ, Cui Y, Koezuka I, Toura Y, Kaneko K, Motoki H, Ueno R, Nakagawa H, Sato E, Kondo R (1997) CD1d-restricted and TCR-mediated activation of Vα14 NKT cells by glycosylceramides. Science 278:1626–1629
Burdin N, Brossay L, Koezuka Y, Smiley ST, Grusby MJ, Gui M, Taniguchi M, Hayakawa K, Kronenberg M (1998) Selective ability of mouse CD1 to present glycolipids: α-galactosylceramide specifically stimulates Vα14+ NKT lymphocytes. J Immunol 161:3271–3281
Bendelac A, Killeen N, Littman DR, Schwartz RH (1994) A subset of CD4+ thymocytes selected by MHC class I molecules. Science 263:1774–1778
Ohteki T, MacDonald HR (1994) Major histocompatibility complex class I-related molecules control the development of CD4+8- and CD4-8- subsets of natural killer 1.1+ T cell receptor-α/β+ cells in the liver of mice.J Exp Med 180:699–704
Zlotnik A, Godfrey DI, Fischer M, Suda T (1992) Cytokine production by mature and immature CD4-CD8- T cells: αβ-Τ cell receptor+ CD4-CD8- T cells produce IL-4. J Immunol 149:1211–1215
Arase H, Arase H, Nakagawa H, Good RA, Onoe K (1993) NK1.1+ CD4+CD8- thymocytes with specific lymphokine secretion. Eur J Immunol 23:307–310
Dao T, Mehal WZ, Crispe IN (1998) IL-18 augments perforin-dependent cytotoxicity of liver NKT- cells. J Immunol 161:2217–2222
Arase H, Arase N, Kobayashi Y, Nishimura Y, Yonehara S, Onoe K (1994) Cytotoxicity of fresh NK1.1+ T cell receptor α/β+ thymocytes against a CD4+CD8+ thymocyte population associated with intact Fas antigen expression on the target. J Exp Med 180:423–432
Koyasu S (1994) CD3+CD16+ NK1.1+B220+ large granular lymphocytes arise from both alpha-beta TCR+CD4-CD8- and gamma-delta TCR+CD4-CD8- cells. J Exp Med 179:1957–1972
Koyasu S, D’Adamio L, Arulanandam AR, Abraham S, Clayton LK, Reinherz EL (1992) T cell receptor complexes containing FcεRI gamma homodimers in lieu of CD3 ζ and CD3 ζ components: a novel isoform expressed on large granular lymphocytes. J Exp Med 175:203–209
Wilson MT, Singh AK, Van Kaer L (2002) Immunotherapy with ligands of natural killer T cells. Trends Mol Med 8:225–231
Lee PT, Benlogha K, Teyton L, Bendelac A (2002) Distinct functional lineages of human: Vα24 natural killer T cells. J Exp Med 195:637–641
Gumperz JE, Miyaka S, Yamamura T, Brenner MB (2002) Functionally distinct subsets of CD1d-restricted natural killer T cells revealed by CD1d tetramer staining. J Exp Med 195:625–636
Douagi I, Colucci F, Di Santo JP, Cumano A (2002) Identification of the earliest prethymic bipotent T/NK progenitor in murine fetal liver. Blood 99:473–471
Yu H, Fehniger TA, Fuchshuber P (1998) Flt3 ligand promotes the generation of a distinct CD34(+) human natural killer cell progenitor that responds to interleukin-15. Blood 92:3647–3657
Carayol G, Robin C, Bourhis JH (1998) NK cells differentiated from bone marrow, cord blood and peripheral blood stem cells exhibit similar phenotype and functions. Eur J Immunol 28:1991–2002
Muench MO, Humeau L, Paek B (2000) Differential effects of interleukin-3, interleukin-7, interleukin-15, and granulocyte-macrophage colony-stimulating factor in the generation of natural killer and B cells from primitive human fetal liver progenitors. Exp Hematol 28:961–973
Bennett IM, Zatsepina O, Zamai L, Azzoni L, Mikheeva T, Perussia B (1996) Definition of a natural killer NKR-P1A+/CD56-/CD16-functionally immature human NK cell subset that differentiates in vitro in the presence of interleukin 12. J Exp Med 184:1845–1856
Gaddy J, Broxmeyer HE (1997) Cord blood CD16+56-cells with low lytic activity are possible precursors of mature natural killer cells. Cell Immunol 180:132–142
Williams NS, Moore TA, Schatzle JD (1997) Generation of lytic natural killer 1.1+Ly-49-cells from multipotential murine bone marrow progenitors in a stroma-free culture: definition of cytokine requirements and developmental intermediates. J Exp Med 186:1609–1614
Perez SA, Gkika DG, Sotiropoulou PA, Mahaira LG, Niarchos DK, Gritzapis AD, Kavalakis GJ, Antsaklis AI, Baxevanis CN, Papamichail M (2003) A novel myeloid like NK cell progenitor in human umbilical cord blood. Blood 101:1–7
Hammong KJL, Pelikan SB, Crowe MY, Randle-Barrett E, Nakayama T, Taniguchi M, Smyth MJ, van Driel IR, Scollay R, Baxter AG, Godfrey DI (1999) NKT cells are phenotypically and functionally diverse. Eur J Immunol 29:3768–3781
Bendelac A, Killeen N, Littman DR, Schwartz RH (1994) A subset of CD4+ thymocytes selected by MHC class I molecules. Science 263:1774–1778
Levitsky H, Golumbek P, Pardoll D (1991) The fate of CD4-CD8- T cells receptor αβ+ thymocytes. J Immunol 146:1113–1118
Hashimoto W, Takeda K, Anzai R, Ogasawara K, Sakihara H, Sugiura H, Seki S, Kumagai K (1995) Cytotoxic NK1.1 Ag+ αβ T cells with intermediate TCR induced in the liver of mice by IL-12. J Immunol 154:4333–4340
Woo S-Y, Jung Y-J, Ryn .-H, Park H-Y, Kie J-H, Im S-A, Chung W-S, Han H-S, Seoh J-Y (2003) In vitro differentiation of natural killer T cells from human cord blood CD34+ cells. Brit J Heamatol 121:148–156
Wagtmann N, Rajagopalan S, Winter CC, Peruzzi M, Long EO (1995) Killer cell inhibitory receptors specific for HLA-C and HLA-B identified by direct binding and functional transfer. Immunity 3:801–809
Vitale M, Sivori S, Pende D (1996) Physical and functional independency of p70 and p58 natural killer (NK) cell receptors for HLA class I: their role in the definition of different groups of alloreactive NK cell clones. Proc Natl Acad Sci U S A 93:1453–1457
Blassoni R, Cantoni C, Falco M (1996) The human leukocyte antigen (HLA)-C-specific “activatory” or “inhibitory” natural killer cell receptors display highly homologous extracellular domains but differ in their transmembrane and intracytoplasmic portions. J Exp Med 183:645–650
Colonna M, Samaridis J (1995) Cloning of immunoglobulin-superfamily members associated with HLA-C and HLA-B recognition by human natural killer cells. Science 268:405–408
Vales-Gomez M, Reyburn HT, Mandelbolm M, Strominger JL (1998) Kinetics of interaction of HLA-C ligands with natural killer cell inhibitory receptors. Immunity 9:337–344
Wilson MJ, Torkar M, Trowsdale J (1997) Genomic organization of a human killer cell inhibitory receptor gene. Tissue Antigens. 49:574–579
Vales-Gomez M, Reyburn HT, Erskins RA, Lopez-Botet M, Strominger JL (1999) Kinetics and peptide dependency of the binding of the inhibitory NK receptor CD94/NKG2-A and the activating receptor CD94/NKG2-C to HLA-E. EMBO J 18:4250–4260
Aramburu J, Balboa MA, Ramirez A (1990) A novel functional cell surface dimmer (Kp43) expressed by natural killer cells and T cell receptor-gamma/delta + T lymphocytes. Inhibition of the IL-2-dependent proliferation by anti-Kp43 monoclonal antibody. J Immunol 144:3238–3247
Lopez-Botet M, Carretero M, Bellon T, Perez-Villar JJ, Liano M, Navarro F (1998) The CD94/NKG2 C-type lectin receptor complex. Curr Top Microbiol Immunol 230:41–52
Lee N, Liano M, Carretero M (1998) HLA-E is a major ligand for the natural killer inhibitory receptor CD94/NKG2A. Proc Natl Acad Sci U S A 95:5199–5204
Liano M, Lee N, Navarro F (1998) HLA-E-bound peptides influence recognition by inhibitory and triggering CD94/NKG2 receptors: preferential responses to an HLA-G-derived nonamer. Eur J Immunol 28:2854–2863
Bauer S, Groh V, Wu J (1999) Activation of NK cells and T cells by NKG2D, a receptor for stress-inducible MICA. Science 285:727–729
Groh V, Bahram S, Bauer S, Herman A, Beauchamp M, Spies T (1996) Cell stress-regulated human major histocompatibility complex class I gene expressed in gastrointestinal epithelium. Proc Natl Acad Sci U S A 93:12445–12450
Moretta A, Bottino C, Vitale M, Pende D, Cantonis C, Mingari MC, Biassoni R, Moretta L (2001) Activating receptors and coreceptors involved in human natural killer cell-mediated cytalysis. Annu Rev Immunol 19:197–223
Ortaldo JR, Young AA (2003) Expression of IFN-γ upon triggering of activating Ly4aD NK receptors in vitr and in vivo: costimulation with IL-12 or IL18 overrides inhibitory receptors. J Immunol 170:1763–1769
Groh V, Rhinehart R, Randolph-Habecker J, Topp M Riddell S, Spies T (2001) Co-stimulation of CD8 alphabeta T-cells by NKG2D via engagement by NK induced on virus-infected cells. Nat Immunol 2:255–260
Kaye J, Browne H, Stoffel M, Minson T (1992) The UL16 gene of human cytomegalovirus encodes a glycoprotein that is dispensable for growth in vitro. J Virol 66:6609–6615
Li P, Wille ST, Bauer S, Morris DL, Spies T, Strong RK (1999) Crystal structure of the MHC class I homolog MIC-A, a gammadelta T cell ligand. Immunity 10:577–584
Groh V, Bahram S, Bauer S, Herman A, Beauchamp M, Spies T (1996) Cell stress-regulated human major histocompatibility complex class I gene expressed in gastrointestinal epithelium. Proc Natl Acad Sci U S A 93:12445–12450
Das H, Groh V, Kuijl C, Sugita M, Morita CT, Spies T, Bukowski JF (2001) MICA engagement by human Vgamma2 Vdelta2 T cells enhances their antigen-dependent effector function. Immunity 15:83–93
Groh V, Rhinehart R, Secrist H, Bauer S, Grabstein KH, Spies T (1999) Broad tumor associated expression and recognition by tumor-derived gamma delta T cells of MICA and MICB. Proc Natl Acad Sci U S A 96:6879–6884
Sutherland CL, Chalupny NJ, Cosman D (2001) The UL16-binding proteins, a novel family of MHC class I-related ligands for NKG2D, activate natural killer functions. Immunol Rev 181:185–192
Pende D, Cantoni C, Rivera P, Vitale M, Castriconi R, Marcenaro S, Nanni M, Biassoni R, Bottino C, Moretta A, Moretta L (2001) Role of NKG2D in tumor cell lysis mediated by human NK cells: cooperation with natural cytotoxicity receptors and capability of recognizing tumors of non-epithelial origin. Eur J Immunol 31:1076–1086
Trapani JA, Davis J, Sutton VR, Smyth MJ (2000) Proapoptotic functions of cytotoxic of cytotoxic lymphocyte granule constituents in vitro and in vivo. Curr Opin Immunol 12:323–329
Smyth MJ, Crowe NY, Godfrey DI (2001) NK cells and NKT cells collaborate in host protection from methylcholanthrene-induced fibrosarcoma. Int Immunol 13:459–463
Davis JE, Smyth MJ, Trapani JA (2001) Granzyme A- and B-deficient killer lymphocytes are defective in eliciting DNA freagmentation but retain potent in vivo anti-tumor capacity. Eur J Immunol 31:39–47
Ashkenazi A (2002) Targeting death and decoy receptors of the tumour-necrosis factor superfamily. Nature Rev Cancer 2:420–430
Bradley M, Zeytun A, Rafi-Janajreh A, Nagarkatti PS, Nagarkatti M (1996) Role of spontaneous and interleukin-2-induced natural killer cell activity in the cytotoxicity and rejection of Fas+ and Fas- tumor cells. Blood 92:4248–4255
Screpanti V, Wallin RP, Ljunggren HG, Grandien A (2001) A central role for death receptor-mediated apoptosis in the rejection of tumors by NK cells. J Immunol 167:2068–2073
Takeda K, Hayakawa Y, Smyth MJ, Kayagaki N, Yamaguchi N, Kakuta S, Iwakura Y, Yagita H, Okumura K (2001) Involvement of tumor necrosis factor-related apoptosis-inducing ligand in surveillance of tumor metastasis by liver natural killer cells. Nature Med 7:94–100
Cretney E, Takeda K, Yagita H, Glaccum M, Peschon JJ, Smyth MJ (2002) Increased susceptibility to tumor initiation and metastasis in TNF-related apoptosis-inducing ligand-deficient mice. J Immunol 168:1356–1361
Smyth MJ, Cretney E, Takeda K, Wiltrout RH, Sedger LM, Kayagaki N, Yagita H, Okumura K (2001) Tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) contributes to interferon gamma-dependent natural killer cell protection from tumor metastasis. J Exp Med 193:661–670
Lee RK, Spielman J, Zhao DY, Olsen KJ, Podack ER (1996) Perforin, Fas ligand and tumor necrosis factor are the major cytotoxic molecules used by lymphokine activated killer cells. J Immunol 157:1914–1919
Ratner A, Clark WR (1993) Role of TNF-α in CD8+ cytotoxic T lymphocyte-mediated lysis. J Immunol 150:4303–4308
Smyth MJ, Sedgwich JD (1998) Delayed kinetics of tumor necrosis factor-mediated bystander lysis by peptide specific CD8+ cytotoxic T lymphocytes. Eur J Immunol 28:4162–4169
Sarin A, Conan CM, Henkart PA (1995) Cytotoxic effect of TNF and lymphotoxin on T lymphocytes. J Immunol 155:3716–3721
Zheng L, Fisher G, Miller RE, Perschon J, Lynch DH, Lenardo MJ (1995) Induction of apoptosis in mature T cells by tumor necrosis factor. Nature 377:348–350
Baxevanis CN, Voutsas IF, Tsitsilonis OE, Tsiatas ML, Gritzapis AD, Papamichail M (2000) Compromised anti-tumor responses in tumor necrosis factor-α knockout mice. Eur J Immunol 30:1957–1966
Loza MJ, Zamai L, Azzoni L, Rosatti E, Perussia M (2002) Expression of type 1 (interferon gamma) and type 2 (interleukin-13, interleukin-5) cytokines at distinct stages of natural killer cell differentiation from progenitor cells. Blood 99:1273–1281
Brion C, Nguyen KB, Pien GC, Cousens LP, Salazar-Mather TP (1999) Natural killer cells in antiviral defense: function and regulation by innate cytokines. Annu Rev Immunol 17:189–220
Street SE, Cretney E, Smyth MJ (2001) Perforin and interferon-amma activities independently control tumor inititation growth and metastasis. Blood 97:192–197
Hayakawa Y, Takeda K, Yagita H, Smyth MJ, van Kaer L, Okumura K, Saiki I (2002) IFN-gamma-mediated inhibition of tumor angiogenesis by natural killer T-cell ligand, alpha-galactosylceramide. Blood 100:1728–1733
Cifone MG, D’Alo S, Parroni R, Millimaggi D, Biordi L, Martinotti S, Santani A (1999) Interleukin-2-activated rat natural killer cells express inducible nitric oxide synthase that contributes to cytotoxic function and interferon-gamma production. Blood 93:3876–3884
Baxevanis CN, Dedoussis GVZ, Papadopoulos NG, Missitzis I, Beroukas C, Stathopoulos GP, Papamichail M (1995) Enhanced human lymphokine-activated killer cell function after brief exposure to granulocyte-macrophage-colony stimulating factor. Cancer 76:1253–1260
Goodier MR, Londei M (2000) Lipopolysaccharide stimulates the proliferation of human CD56+CD3- NK cells: a regulatory role of monocytes and IL-10. J Immunol 165:139–147
Cai G, Kanstelein RA, Hunter CA (1999) IL-10 enhances NK cell proliferation, cytotoxicity and production of IFN-γ when combined with IL-18. Eur J Immunol 29:2658–2663
Wysocka M, Kubin M, Vieira LQ, Ozmen L, Garotta G, Scott P, Trinchieri G (1995) Interleukin-12 is required for interferon-gamma production and lethality in lipopolysaccharide-induced shock in mice. Eur J Immunol 25:672–680
Orange JS, Salazar-Mather TP, Opal SM, Spencer RL, Miller AH, McEwen BS, Biron CA (1995) Mechanism of interleukin 12-mediated toxicities during experimental viral infections: role of tumor necrosis factor and glucocorticoids. J Exp Med 181:901–912
Zitvogel L (2002) Dendritic and natural killer cells cooperate in the control/switch of innate immunity. J Exp Med 195:F9–F14
Piccioli D, Sbrana S, Melandri E, Valiante NM (2002) Contact-dependent stimulation and inhibition of dendritic cells by natural killer cells. J Exp Med 195:335–341
Bendelac A, Rivera MN, Park SH, Roark JH (1997) Mouse CD1-specific NK1 T cells: development, specificity, and function. Annu Rev Immunol 15:535–562
Godfrey D, Hammond KJL, Poulton LD, Smyth MJ, Baxter AG (2000) NKT cells: facts, functions and fallacies. Immunol Today 21:573–583
Joyce S (2001) CD1d and natural T cells: how their properties jump-start the immune system. Cell Mol Life Sci 58:442–469
Smyth MJ, Taniguchi M, Street SE (2000) The antitumor activity of IL-12: mechanisms of innate immunity that are dose and model dependent. J Immunol 165:2665
Smyth MJ, Thia KY, Street SE, Cretney. E, Trapani JA, Taniguchi M, Kawano S, Pelikan B, Crowe NY, Godfrey DI (2000) Differential tumor surveillance by natural killer (NK) and NKT cells. J Exp Med 191:661
Terabe M, Matsui O, Noben-Trauth N, Chen H, Watson C, Donaldson DD, Carbone DP, Paul WE, Berzofsky J (2000) NKT cell-mediated repression of tumor immunosurveillance by IL-13 and the IL-4R-STAT6 pathway. Nat Immunol 1:515–520
Moodycliffe AM, Nghiem D, Clydesdale G, Ullrich SE (2000) Immune suppression and skin cancer development: regulation by NKT cells. Nat Immunol 1:521–525
Baxevanis CN, Gritzapis AD, Papamichail M (2003) In vivo antitumor activity of NKT cells activated by the combination of IL-12 and IL-18. J Immunol 171:2953–2959
Suzuki H, Duncan GS, Takimoto H, Mak TW (1997) Abnormal development of intestinal intraepithelial lymphocytes and peripheral natural killer cells in mice lacking the IL-2 receptor β chain. J Exp Med 185:499–508
Ohteki TS, Ho H, Suzuki TW, Mak, Ohashi PS (1997) Role for IL-15/IL-15 receptor β-chain in natural killer 1.1+ T cell receptor-αβ+ cell development. J Immunol 159:5931–5938
Kennedy MK, Glaceum M, Brown SN, Butz EA, Viney JL, Embers M, Matsuki N, Charrier K, Sedger L, Willis CR, Brasel K, Morrissey PJ, Stocking K, Schuh JC, Joyce S, Peschon JJ (2000) Reversible defects in natural killer and memory CD8 T cell lineages in interleukin 15-deficient mice. J Exp Med 191:771–780
Zhang X, Sun S, Hwang I, Tough DF, Sprent J (1998) Potent and selective stimulation of memory-phenotype CD8+ T cells in vivo by IL-15. Immunity 8:591–600
Carson WE, Giri JG, Lindemann MJ, Linett ML, Ahdieh M, Paxton R, Anderson D, Eisenmann J, Grabstein K, Caliguiri MA (1994) Interleukin (IL) 15 is a novel cytokine that activates human natural killer cells via components of the IL-2 receptor. J Exp Med 180:1395–1403
Carson WE, Ross ME, Baiocchi RA, Marien MJ, Boiani N, Grabstein K, Caliguiri MA (1995) Endogenous production of interleukin 15 by activated human monocytes is critical for optimal production of interferon-γ by natural killer cells in vitro. J Clin Invest 96:2578–2584
Carson WE, Fehniger TA, Haldar S, Eckhert K, Lindemann MJ, Lai C-F, Croce CM, Baumann H, Caliguiri MA (1997) A potential role for interleukin-15 in the regulation of human natural killer cell survival. J Clin Invest 99:937–942
Fehniger TA, Cooper MA, Caligiuri MA (2002) Interleukin-2 and interleukin-15: immunotherapy for cancer. Cytokine Growth Factor Rev 13:159–183
Hunter CA, Gabriel KE, Radzanowski T, Neyer LE, Remington JS (1997) Type I interferons enhance production of IFN-γ by NK cells. Immunol Lett 59:1–5
Matinainen S, Paananen A, Miettinen M, Kurimoto M, Timonen T, Julkunen I, Sareneva T (2001) IFN-alpha and IL-18 synergistically enhance IFN-gamma production in human NK cells: differential regulation of Stat4 activation and IFN-gamma gene expression by IFN-alpha and IL-12. Eur J Immunol 31:2236–2245
Tripp CS, Wolf SE, Unanue ER (1993) Interleukin-12 and tumor necrosis factor α are costimulators of interferon γ production by natural killer cells in severe combined immunodeficiency mice with listeriosis, and interleukin 10 is a physiological antagonist. Proc Natl Acad Sci U S A 90:3725–3729
Hunter CA, Timans JC, Pisacane P, Menon S, Cai G, Walker W, Aste-Amezaga M, Chizzonite R, Bazan JF, Kastelein RA (1997) Comparison of the effects of interleukin-1α, interleukin-1β and interferon-γ-inducing factor on the production of interferon-γ by natural killer. Eur J Immunol 27:2787–2792
Cai G, Kastelein RA, Hunter CA (1999) IL-10 enhances NK cell proliferation, cytotoxicity and production of IFN-γ when combined with IL-18. Eur J Immunol 29:2658–2555
Trinchieri G (1995) Interleukin-12: a proinflammatory cytokine with immunoregulatory functions that bridge innate resistance and antigen-specific adoptive immunity. Annu Rev Immunol 13:251–285
Trinchieri G (1997) Cytokines acting on or secreted by macrophages during intracellular infection (IL-15, IL-12, IFN-γ). Curr Opin Immunol 9:17–21
Brunda MJ (1994) Interleukin-12. J Leucocyte Biol 55:280–290
Robertson MJ, Ritz J (1996) Interleukin-12: basic biology and potential applications in cancer treatment. Oncologist 1:88–97
Okamura H, Tsutsi H, Komatsu T, Yutsudo M, Hakura A, Taminoto T, Torigoe K, Okura T, Nukada Y, Hattori K, Akita K, Namba M, Tanabe F, Konishi K, Fukunda S, Kurimoto M (1995) Cloning of a new cytokine that induces IFN-γ production by T cells. Nature 378:88–92
Okamoto I, Kohno K, Tanimoto T, Ikegami H,Kurimoto M (1999) Development of CD8+ effector T cells is differentially regulated by IL-18 and IL-12. J Immunol 162:3202–3210
Osaki T, Pero JM, Cai W, Okamura H, Robbins PD, Kurimoto M, Nagata S, Lotze MT, Tahara H (1998) IFN-γ-inducing factor/IL-18 administration mediated IFN-γ- and IL-12-independent antitumor effects. J Immunol 160:1742–1749
Micalleh MJ, Yoshida K, Kawai S, Hamaya T, Kohno K, Arai S, Tanimoto T, Torigoe K, Fujii M, Ikeda M, Kurimoto M (1997) In vivo antitumor effects of murine interferon-γ-inducing factor/interleukin-18 in mice bearing syngeneic Meth A sarcoma malignant ascites. Cancer Immunol Immunother 43:961–970
Takeda K, Tsutsi H, Yoshimoto T, Adachi O, Yoshida N, Kishimoto T, Okamura H, Nakanishi K, Akira S (1998) Defective NK cell activity and Th1 response in IL-18-deficient mice. Immunity 8:383–390
Akamatsu S, Arai N, Hayana T, Arai S, Tanimoto T, Fujii M, Kohno K, Micallef MJ, Ikeda M, Kurimoto M (2002) Antitumor activity of interleukin-18 against the murine T-cell leukemia/lymphoma EL-4 in syngeneic mice. J Immunother 25[Suppl 1]:28–35
Wang W, Hu H, Ju DW, He L, Pan JP, Xia DJ, Zhang LH, Cao X (2001) Intratumoral IL-18 gene transfer improves therapeutic efficacy of antibody-targeted superantigen in established murine melanoma. Gene Ther 8:542–549
Ju DW, Yang Y, Tao Q, Song WG, He L, Chen G, Gu S, Ting CC, Cao X (2000) Interleukin-18 gene transfer increases antitumor effects of suicide gene therapy through efficient induction of antitumor immunity. Gene Ther 19:1672–1680
Tatsumi T, Gambotto A, Robbins PD, Storkus WJ (2002) Interleukin 18 gene transfer expands the repertoire of antitumor Th1-type immunity elicited by dendritic cell-based vaccines in association with enhanced therapeutic efficacy. Cancer Res 62:5853–5860
Leite-de-Moraes MC, Hameg A, Arnoud A, Machavoine F, Koezuka Y, Schneider E, Herbelin A, Dy M (1999) A distinct IL-18-induced pathway to fully activate NKT lymphocytes indendently from TCR engagement. J Immunol 163:5871–5880
Hyodo Y, Matsui K, Hayashi N, Tsutsui T, Kashiwamura SI, Yamauchi H, Hiroishi K, Takeda K, Tagawa Y-I, Twakura Y, Kayagami N, Kurimoto M, Okamura H, Hada T, Yogita H, Akira S, Nakanishi K, Higashino K (1999) IL-18 up-regulates perforin-mediated NK activity without increasing perforin messenger RNA expression by binding to constitutively expressed IL-18 receptor. J Immunol 162:1662–1670
Nagai H, Hara Y, Horikawa T, Fujii M, Kurimoto M, Kamidono S, Ichihashi M (2000) Antitumor effects on mouse melanoma elicited by local secretion of interleukin-12 and their enhancement by treatment with interleukin-18. Cancer Invest 18:206–211
Kishida T, Asada H, Satoh E, Shinya M, Hirai H, Iwai M, Tahara H, Imanishi J, Mazda O (2001) In vivo electroporation-mediated transfer of interleukin-12 and interleukin-18 genes induces significant antitutmor effects against melanoma in mice. Gene Ther 8:1234–1240
Yamanaka R, Yajia N, Tsuchiya N, Homna H, Tanaka R, Ramsey H, Blaese M, Xanthopoulos KG (2002) Administration of interleukin-12 and -18 enhancing the antitumor immunity of genetically modified dendritic cells that had been pulsed with Semliki forest virus-mediated tumor complementary DNA. J Neurosurg 97:1184–1190
Hashimoto W, Tanaka F, Robbins PD, Taniguchi M, Okamura H, Lotze MT, Tahara H (2003) Natural killer, but not natural killer T cells play a necessary role in the promotion of an innate antitumor response induced by IL-18. Int J Cancer 103:508–515
Kruit WH, Goey SH, Lamers CH, Gratama JW, Visser B, Schmitz PI, Eggermont AM, Bolhuis RL, Stoter G (1997) High-dose regimen of interleukin-2 and interferon-alpha in combination with lymphokine-activated killer cells in patients with metastatic renal cell cancer. J Immunother 20:312–320
Kimura H, Yamaguchi Y (1997) A phase III randomized study of interleukin-2 lymphokine-activated killer cell immunotherapy combined with chemotherapy or radiotherapy after curative or noncurative resection of primary lung carcinoma. Cancer 80:42–49
Law TM, Motzer RJ, Mazumdar M, Sell KW, Walther PJ, O’Connell M, Khan A, Vlamis V, Vogelzang NJ, Bajorin DF (1995) Phase III randomized trial of interleukin-2 with or without lymphokine-activated killer cells in the treatment of patients with advanced renal cell carcinoma. Cancer 76:824–832
Soiffer RJ, Murray C, Gonin R, Ritz J (1994) Effect of low-dose interleukin-2 on disease relapse after T-cell-depleted allogeneic bone marrow transplantation. Blood 84:964–971
Caliguiri MA, Murray C, Soiffer RJ, Klumpp TR, Seiden M, Cochran K, Cameron C, Ish C, Buchanan L, Perillo D (1991) Extended continous infusion low-dose recombinant interleukin-2 in advanced cancer; prolonged immunomodulation without significant toxicity. J Clin Oncol 9:2110–2119
Lim SH, Newland AC, Kelsey S, Bell A, Offerman E, Rist C, Gozzard D, Bareford D, Smith MP, Goldstone AH (1992) Continous intravenous infusion of high-dose recombinant interleukin-2 for acute myeloid leukaemia: a phase II study. Cancer Immunol Immunother 34:337–342
Mercopol NJ, Barressi GM, Fehniger TA, Hitt J, Franklin M, Caliguiri MA (1998) Evaluation of natural killer cell expansion and activation in vivo with daily subcutaneous low-dose interleukin-2 plus periodic intermediate-dose pulsing. Cancer Immunol Immunother 46:318–326
Cortes JE, Kantarjian HM, O’Brien S, Giles F, Keating MJ, Freireich EJ, Estey EH (1999) A pilot study of interleukin-2 for adult patients with acute myelogenous leukemia in first complete remission. Cancer 85:1506–1513
Hayes RL, Arbit E, Odaimi M, Pannullo S, Scheff R, Kravchinskiy D, Zaroulis C (2001) Adoptive cellular immunotherapy for the treatment of malignant gliomas. Crit Rev Oncol Hematol 39(1–2):31–42
Kimoto Y, Tanaka T, Tanji Y, Fujiwara A, Taguchi T (1995) Use of human leukocyte antigen-mismatched allogeneic lymphokine-activated killer cells and interleukin-2 in the adoptive immunotherapy of patients with malignancies. Biotherapy 8:41–50
Boughton B, Simpson A, Phaure T, Beatty C (1995) Graft-versus-host disease following interleukin-2/lymphokine-activated killer (LAK) cell immunotherapy in a patient with acute myelogenous leukemia in second complete remission: autologous LAK cells following allogeneic bone marrow transplantation are donor-derived. Cancer Immunol Immunother 41:68–70
Cesano A, Visonneau S, Santoli D (1995) Treatment of experimental glioblastoma with a human MHC non-restricted cytotoxic T-cell line. Cancer Res 55:96–101
Cesano A, Pierson G, Visonneau S, Migliaccio A, Santoli D (1996) Use of a lethally irradiated major histocompatibility complex nonrestricted cytotocix T-cell line for effective purging of marrows containing lysis-sensitive or –resistant leukemic targets. Blood 87:393–403
Yan Y, Steinherz P, Klingemann H-G, Dennig D, Childs BH, McGuirk J, O’Reilly RJ (1998) Antileukemia activity of natural killer cell line against human leukemia. Clin Cancer Res 4:2859–2868
Uherek C, Toon T, Uherek B, Becker B, Schnierle B, Klingemann H-G, Wels W (2002) Retargeting of natural killer-cell cytolytic activity to ErbB2-expressing cancer cells results in efficient and selective tumor cell destruction. Blood 100:1265–1273
Maki G, Krystal G, Dougherty G, Takei F (1998) Induction of sensitivity to NK-mediated cytotoxicity by TNF-alpha treatment possible role of ICAM3 and CD44. Leukemia 12:1565–1572
Klingemann H-G, Wong W, Maki G (1996) A cytotoxic NK-cell line (NK-92) for ex vivo purging of leukemia from blood. Biol Blood Marrow Transplant 2:68–75
Carson WE, Parihar R, Lindenmann MJ, Personeri N, Dierksheide J, Meropol NJ, Baelga J, Caligiuri MA (2001) Interleukin-2 enhances the natural killer cell response to Herceptin-coated HER2/neu-positive breast cancer cells. Eur J Immunol 31:3016–3025
Meropol NJ, Barresi GM, Fehniger TA, Hitt J, Franklin M, Caligiuri MA (1998) Evaluation of natural killer cell expansion and activation in vivo with daily subcutaneous low-dose interleukin-2 plus periodic intermediate dose-pulsing. Cancer Immunol Immunother 46:318–326
Kawano T, Cui J, Koezukay, Toura J, Kaneko Y, Sato H, Kondo E, Harada M, Koseki H, Nakayama T, Tanaka Y, Taniguchi M (1998) Natural killer-like nonspecific tumor cell lysis mediated by specific ligand-activated Vα14 NKT cells. Proc Natl Acad Sci U S A 95:5690–5693
Nishimura T, Kitamura H, Iwakabe K, Yahata T, Ohta A, Sato M, Takeda K, Okumura K, van Kaer L, Kawano T, Taniguchi H, Nakui M, Sekimoto M, Koda T (2000) The interface between innate and acquired immunity: glycolipid antigen presentation by CD1d-expressing dendritic cells to NKT cells induces the differentiation of antigen-specific cytotoxic T lymphocytes. Int Immunol 12:987–994
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was presented at the first Cancer Immunology and Immunotherapy Summer School, 8–13 September 2003, Ionian Village, Bartholomeio, Peloponnese, Greece.
Rights and permissions
About this article
Cite this article
Papamichail, M., Perez, S.A., Gritzapis, A.D. et al. Natural killer lymphocytes: biology, development, and function. Cancer Immunol Immunother 53, 176–186 (2004). https://doi.org/10.1007/s00262-003-0478-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-003-0478-4