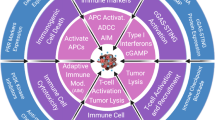
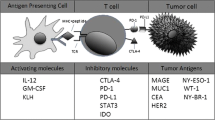
Abstract
After decades of work to develop immune-based therapies for cancer, the first drugs designed specifically to engage the host anti-tumor immune response for therapeutic benefit were recently approved for clinical use. Sipuleucel-T, a vaccine for advanced prostate cancer, and ipilimumab, a monoclonal antibody that mitigates the negative impact of cytotoxic T lymphocyte antigen-4 signaling on tumor immunity, provide a modest clinical benefit in some patients. The arrival of these drugs in the clinic is a significant advance that we can capitalize on for even better clinical outcomes. The strategic and scientifically rational integration of vaccines and other direct immunomodulators with standard cancer therapeutics should lead to therapeutic synergy and high rates of tumor rejection. This review focuses on the use of cyclophosphamide, doxorubicin, and HER-2-specific monoclonal antibodies to dissect mechanisms of immune tolerance relevant to breast cancer patients and illustrates how appropriate preclinical models can powerfully inform clinical translation. The immune-modulating activity of targeted, pathway-specific, small molecule therapeutics is also discussed. Fully understanding how cancer drugs impact the immune system should lead to the ultimate personalized cancer medicine: effective combinatorial immunotherapy strategies that simultaneously target signaling pathways essential for tumor growth and progression, and systematically break multiple, distinct immune tolerance pathways to maximize tumor rejection and effect cure.
Similar content being viewed by others
References
Kantoff P, Higano C, Shore N, Berger E, Small E et al (2010) Sipuleucel-T immunotherapy for castration-resistant prostate cancer. N Engl J Med 363:411–422
Hodi F, O’Day S, McDermott D, Weber R, Sosman J et al (2010) Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med 363:711–723
Robert C, Thomas L, Bondarenko I, O’Day S et al (2011) Ipilimumab plus decarbazine for previously untreated melanoma. N Engl J Med 364:2517–2526
Emens L, Jaffee E (2007) Immunotherapy and cancer therapeutics: why partner? In: Prendergast G, Jaffee E (eds) Cancer immunotherapy and immune suppression, 1st edn. Academic Press, Elsevier, London, pp 207–233
Emens L (2010) Chemoimmunotherapy. Cancer J J Princ Prac Oncol 16:295–303
Emens L, Machiels J, Reilly R, Jaffee E (2001) Chemotherapy: friend or foe to cancer vaccines? Curr Opin Mol Ther 3:77–84
Ferris R, Jaffee E, Ferrone S (2010) Tumor-antigen-targeted, monoclonal antibody0based immunotherapy: clinical response, cellular immunity, and immunoescape. J Clin Oncol 28:4390–4399
Guy CT, Webster MA, Schaller M, Parsons TJ, Cardiff RD et al (1992) Expression of the neu protooncogene in the mammary epithelium of transgenic mice induces metastatic disease. Proc Natl Acad Sci U S A 89:10578–10582
Reilly RT, Gattlieb MB, Ercolini AM, Machiels JP, Kane CE et al (2000) HER-2/neu is a tumor rejection target in tolerized HER-2/neu transgenic mice. Cancer Res 60:3569–3576
Machiels J, Reilly R, Emens L, Ercolini A, Lei R et al (2001) Cyclophospha-mide, doxorubicin, and paclitaxel enhance the antitumor immune response of granulocyte/macrophage-colony stimulating factor-secreting whole-cell vaccines in HER-2/neu tolerized mice. Cancer Res 61:3689–3697
Ercolini A, Machiels J, Chen Y, Slansky J, Giedlen M et al (2003) Identification and characterization of the immunodominant rat HER-2/neu MHC class I epitope presented by spontaneous mammary tumors from HER-2/neu-transgenic mice. J Immunol 170:4273–4280
Uram J, Black C, Flynn E, Huang L, Armstrong T et al (2011) Nondominant CD8+ T cells are active players in the vaccine-induced antitumor immune response. J Immunol 186:3847–3857
Nigam A, Yacavone R, Zahurak M, Johns C, Pardoll D et al (1998) Immunomodulatory properties of antineoplastic drugs administered in conjunction with GM-CSF-secreting cancer cell vaccines. Int J Cancer 12:161–170
Ercolini A, Ladle B, Manning E, Pfannenstiel L, Armstrong T et al (2005) Recruitment of latent pools of high-avidity CD8+ T cells to the antitumor immune response. J Exp Med 201:1519–1602
Reilly R, Machiels J, Emens L, Ercolini A, Okoye F et al (2001) The collaboration of both humoral and cellular HER-2/neu–targeted immune responses is required for the complete eradication of HER-2/neu-expressing tumors. Cancer Res 61:880–883
Wolpoe M, Lutz E, Murata S, Ivie S et al (2003) HER-2/neu-specific monoclonal antibodies collaborate with HER-2/neu-targeted granulocyte macrophage colony-stimulating whole cell vaccination to augment CD8+ T cell effector function and tumor-free survival in HER-2/neu-transgenic mice. J Immunol 171:2161–2169
Kim P, Armstrong T, Song H, Wolpoe M, Weiss V et al (2008) Antibody association with HER-2/neu-targeted vaccine enhances CD8+ T cell responses in mice through Fc-mediated activation of DCs. J Clin Investig 188:1700–1711
Davis-Sproul JM, Harris MP, Davidson NE, Kobrin BJ, Jaffee EM et al (2005) Cost-effective manufacture of an allogenic GM-CSF-secreting breast tumor vaccine in an academic cGMP facility. Cytotherapy 7:46–56
Emens LA, Asquith JM, Leatherman JM, Kobrin BJ, Petrik S, Laiko M, Levi J, Daphtary MM, Biedrzycki B, Wolff AC, Stearns V, Disis ML, Ye X, Piantadosi S, Fetting JH, Davidson NE, Jaffee EM (2009) Timed sequential treatment with cyclophosphamide, doxorubicin, and an allogeneic granulocyte-macrophage colony-stimulating factor-secreting breast tumor vaccine: a chemotherapy dose-ranging factorial study of safety and immune activation. J Clin Oncol 27:5911–5918
Emens L, Gupta R, Petrik S, Laiko M, Levi J et al (2011) A feasibility study of combination therapy with trastuzumab (T), cyclophoshamide (CY), and al allogenic GM-CSF-secreting breast tumor vaccine for the treatment of HER-2+ breast cancer. Proc Am Soc Clin Oncol (abstract 2435)
Emens LA (2005) Trastuzumab: targeted therapy for the management of HER-2/neu-overexpressing metastatic breast cancer. Am J Ther 12:243–253
Cobleigh MA, Vogel CL, Tripathy D, Robert NJ, Scholl S et al (1999) Multinational study of the efficacy and safety of humanized anti-HER2 monoclonal antibody in women who have HER2-overexpressing metastatic breast cancer that has progress after chemotherapy for metastatic disease. J Clin Oncol 17:2639–2648
Vogel CL, Cobleigh MA, Tripathy D, Gutheil JC, Harris LN et al (2002) Efficiacy and safety of trastuzumab as a single agent in the first-line treatment of HER2-overexpressing metastatic breast cancer. J Clin Oncol 20:719–726
Clynes R, Towers T, Presta L, Ravetch J (2000) Inhibitory Fc receptors modulate in vivo cytoxicity against tumor targets. Nat Med 6:443–446
Manning E, Ullman J, Leatherman J, Asquith J, Hansen T et al (2007) A vascular endothelial growth factor receptor-2 inhibitor enhances antitumor immunity through an immune-based mechanism. Clin Cancer Res 13:3951–3959
Prewett M, Huber J, Li Y, Santiago A, O’Connor W, King K, Overholser J, Hooper A, Pytowski B, Witte L, Bohlen P, Hicklin DJ (1999) Antivascular endothelial growth factor receptor (fetal liver kinase 1) monoclonal antibody inhibits tumor angiogenesis and growth of several mouse and human tumors. Cancer Res 50:5209–5218
Gabrilovich D, Ishida T, Nadaf S, Ohm J, Carbone D (1999) Antibodies to vascular endothelial growth factor enhance the efficacy of cancer immunotherapy by improving endogenous dendritic cell function. Clin Cancer Res 5:2963–2970
Ozao-Choy J, Ma G, Kao J, Wang G, Meseck M et al (2009) The novel role of tyrosine kinase inhibitor in the reversal of immune suppression and modulation of tumor microenvironment for immune-based cancer therapies. Cancer Res 69:2514–2522
Edwards JP, Emens LA (2010) The multikinase inhibitor sorafenib reverses the suppression of IL-12 and enhancement of IL-10 by PGE in murine macrophages. Int Immunopharmacol 10:1220–1228
Boni A, Cogdill A, Dang P, Udayakumar D, Njauw C et al (2010) Selective BRAFV600E inhibition enhances T-cell recognition of melanoma without affecting lymphocyte function. Cancer Res 70:5213–5219
Acknowledgments
This work was supported by the Department of Defense (Clinical Translational Research Award W81XWH-07-1-0485), the American Cancer Society (RSG CCE 112685), the Specialized Programs in Research Excellence (SPORE) in Breast Cancer (P50CA88843), Genentech Incorporated, the Gateway Foundation, the Avon Foundation, and the V Foundation.
Conflict of interest
Dr. Emens receives research funding from Genentech, Incorporated, and has received honoraria for participating on regional advisory panels for Genentech, Incorporated, and Roche Incorporated. Under a licensing agreement between Biosante, Incorporated, and the Johns Hopkins University, the University is entitled to milestone payments and royalty on sales of the GM-CSF-secreting breast cancer vaccine. The terms of these arrangements are being managed by the Johns Hopkins University in accordance with its conflict of interest policies.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Emens, L.A. Re-purposing cancer therapeutics for breast cancer immunotherapy. Cancer Immunol Immunother 61, 1299–1305 (2012). https://doi.org/10.1007/s00262-012-1247-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-012-1247-z