Abstract
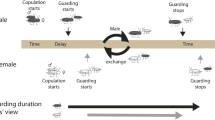
Sexual conflict is common in nature, but detailed behavioral studies on the role female resistance behavior plays in shaping mating patterns are rare. I manipulated female resistance to examine its effects on pairing dynamics in two ecologically different freshwater amphipods. I found evidence for female behavior playing a role in both the outcome of pre-pairing interactions and the initiation of pairing in both species. In these species, the male optimum pairing duration is greater than the value preferred by females or compromised pairing durations observed under natural conditions, thus indicating sexual conflict. Furthermore, the proportion of male–female encounters producing male grasping was greater and the duration of such interactions was longer when female resistance was reduced. Thus, sexual conflict over pairing duration may select simultaneously for female resistance and for male persistence both of which mediate the outcome of pre-pairing interactions in Hyalella. Contact precopulatory mate guarding and the interactions that precede it are common components of crustacean and insect mating systems, suggesting that such conflicts may play an important role in the evolution of mating traits in many taxa.




Similar content being viewed by others
References
Arnqvist G (1992) Pre-copulatory fighting in a water strider: inter-sexual conflict or mate assessment. Anim Behav 43:559–567
Arnqvist G, Rowe L (2005) Sexual conflict. Princeton University Press, Princeton, New Jersey
Borowsky B (1984) The use of the males’ gnathopods during precopulation in some gammaridean amphipods. Crustaceana 47:245–250
Bousfield EL (1958) Fresh-water amphipod crustaceans of glaciated North America. Can Field-Nat 72:55–113
Chapman TG, Arnqvist G, Bangham J, Rowe L (2003) Sexual conflict. Trends Ecol Evol 18:41–47
Cothran RD (2004) Precopulatory mate guarding affects predation risk in two freshwater amphipod species. Anim Behav 68:1133–1138
Dick JTA, Elwood RW (1996) Effects of natural variation in sex ratio and habitat structure on mate-guarding decisions in amphipods (Crustacea). Behaviour 133:985–996
Edwards TD, Cowell BC (1992) Population dynamics and secondary production of Hyalella azteca (Amphipoda) in Typha stands of a subtropical Florida lake. J North Am Benthol Soc 11:69–79
Gavrilets S (2000) Rapid evolution of reproductive barriers driven by sexual conflict. Nature 403:886–889
Härdling R, Kaitala A (2005) The evolution of repeated mating under sexual conflict. J Evol Biol 18:106–115
Holmes SJ (1903) Sex recognition among amphipods. Biol Bull 5:288–292
Jormalainen V (1998) Precopulatory mate guarding: male competitive strategy and intersexual conflict. Q Rev Biol 73:275–304
Jormalainen V, Merilaita S (1995) Female resistance and duration of mate-guarding in 3 aquatic peracarids (Crustacea). Behav Ecol Sociobiol 36:43–48
Jormalainen V, Shuster SM (1999) Female reproductive cycle and sexual conflict over precopulatory mate-guarding in Thermosphaeroma isopods. Ethology 105:233–246
Jormalainen V, Tuomi J, Yamamura N (1994) Intersexual conflict over precopula duration in mate guarding Crustacea. Behav Process 32:265–283
Jormalainen V, Merilaita S, Riihimäki J (2001) Costs of intersexual conflict in the isopod Idotea baltica. J Evol Biol 14:763–772
Krupa J, Sih A (1993) Experimental studies on water strider mating dynamics: spatial variation in density and sex ratio. Behav Ecol Sociobiol 33:107–120
Magurran AE, Seghers BH (1994a) Sexual conflict as a consequence of ecology—evidence from guppy, Poecilia reticulata, populations in Trinidad. Proc R Soc Lond B Biol Sci 255:31–36
Magurran AE, Seghers BH (1994b) A cost of sexual harassment in the guppy, Poecilia reticulata. Proc R Soc Lond B Biol Sci 258:89–92
Martin OY, Hosken DJ (2003) The evolution of reproductive isolation through sexual conflict. Nature 423:979–982
Martin OY, Hosken DJ (2004) Reproductive consequences of population divergence through sexual conflict. Curr Biol 14:906–910
Parker GA (1974a) Courtship persistence and female-guarding as male time investment strategies. Behaviour 48:157–184
Parker GA (1974b) Assessment strategy and evolution of fighting behaviour. J Theor Biol 47:223–243
Parker GA (1979) Sexual selection and sexual conflict. In: Blum MS, Blum NA (eds) Sexual selection and reproductive competition in insects. Academic, New York
Pickard DP, Benke AC (1996) Production dynamics of Hyalella azteca (Amphipoda) among different habitats in a small wetland in the southeastern U S A. J North Am Benthol Soc 15:537–550
Pizzari TR, Snook R (2003) Perspective: Sexual conflict and sexual selection: chasing away paradigm shifts. Evolution 57:1223–1236
Quinn GP, Keough MJ (2002) Experimental design and data analysis for biologists. Cambridge University Press, Cambridge
Ridley M (1983) The explanation of organic diversity: the comparative method and adaptations for mating. Clarendon, Oxford
Robinson BW, Doyle RW (1985) Trade-off between male reproduction (amplexus) and growth in the amphipod Gammarus lawrencianus. Biol Bull 168:482–488
Rowe L, Day T (2006) Detecting sexual conflict and sexually antagonistic coevolution. Philos Trans R Soc Lond B 361:277–285
Rowe L, Arnqvist G, Sih A, Krupa JJ (1994) Sexual conflict and the evolutionary ecology of mating patterns: water striders as a model system. Trends Ecol Evol 9:289–293
Sparkes TC, Keogh DP, Haskins KE (2000) Female resistance and male preference in a stream-dwelling isopod: effects of female molt characteristics. Behav Ecol Sociobiol 47:145–155
Strong DR (1972) Life history variation among populations of an amphipod (Hyalella azteca). Ecology 53:1103–1111
Strong JDR (1973) Amphipod amplexus, the significance of ecotypic variation. Ecology 54:1383–1388
Sutcliffe DW (1992) Reproduction in Gammarus (Crustacea, Amphipoda): basic processes. Freshw Forum 2:102–129
Thornhill R (1980) Rape in Panorpa scorpionflies and a general rape hypothesis. Anim Behav 28:52–59
Thornhill R, Alcock J (1983) The evolution of insect mating systems. Harvard University Press, Cambridge
Wellborn GA (1994) Size-biased predation and prey life histories: a comparative study of freshwater amphipod populations. Ecology 75:2104–2117
Wellborn GA (1995) Determinates of reproductive success in a freshwater amphipod species that experience different mortality regimes. Anim Behav 50:353–363
Wellborn GA, Bartholf SA (2005) Ecological context and the importance of body and gnathopod size for pairing success in two amphipod species. Oecologia 143:308–316
Wellborn GA, Cothran RD (2007) Evolution and ecology of mating behavior in freshwater amphipods. In: Duffy E, Thiel M (eds) Evolutionary ecology of social and sexual systems: crustaceans as model organisms. Cambridge University Press, Cambridge
Wellborn GA, Cothran R, Bartholf S (2005) Life history and allozyme diversification in regional species of the Hyalella azteca (Crustacea: Amphipoda) species complex. Biol J Linn Soc 84:161–175
Yamamura N, Jormalainen V (1996) Compromised strategy resolves intersexual conflict over pre-copulatory guarding duration. Evol Ecol 10:661–68
Acknowledgments
This research was funded by grants from The University of Oklahoma Graduate Student Senate, and scholarships from the University of Oklahoma, College of Arts and Sciences (Sturgis Scholarship), Department of Zoology (Blanche Adams Memorial Scholarship) and Biological Station (Summer Fellowship). My graduate advisor Gary Wellborn provided insightful comments and encouragement throughout the preparation of the manuscript. I thank my advisory committee Wayne Elisens, Ola Fincke, Rosemary Knapp, and Douglas Mock for guidance and constructive criticisms. Veijo Jormalainen and an anonymous reviewer offered constructive comments on an earlier version of the manuscript. This manuscript formed part of the requirements for a Doctor of Philosophy degree at the University of Oklahoma. The experiments herein comply with the current laws of the United States of America.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by P. Backwell
Rights and permissions
About this article
Cite this article
Cothran, R.D. Phenotypic manipulation reveals sexual conflict over precopula duration. Behav Ecol Sociobiol 62, 1409–1416 (2008). https://doi.org/10.1007/s00265-008-0570-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00265-008-0570-z