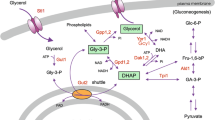
Abstract
Saccharomyces cerevisiae uses different mechanisms to adapt to changes in environmental osmolarity. Upon hyperosmotic shock, cells first mobilize a rapid rescue system that prevents excessive loss of ions and water; then in the adaptation period they accumulate a compatible solute (glycerol). When subjected to hypoosmotic shock, they rapidly release intracellular stocks of glycerol to reduce intracellular osmolarity and prevent bursting. The plasma membrane Nha1 alkali metal cation/H+ antiporter is not important in helping the cells to survive a sudden drop in external osmolarity, but is involved in the cell response to hyperosmotic shock. For this role, its long hydrophilic C-terminus is indispensable. The capacity of the Nha1 antiporter to transport potassium is regulated by Hog1 kinase. Upon sorbitol-mediated stress, the Nha1p potassium export activity decreases in order to maintain a higher intracellular concentration of solutes. The C-terminal-less Nha1 version is not inactivated and its potassium efflux activity renders cells very sensitive to hyperosmotic shock. Taken together, our results suggest an important role of Nha1p and its C-terminus in the immediate response to hyperosmotic shock as part of the rapid rescue mechanism.






Similar content being viewed by others
References
Albertyn J, Hohmann S, Thevelein JM, Prior BA (1994) GPD1, which encodes glycerol-3-phosphate dehydrogenase, is essential for growth under osmotic stress in Saccharomyces cerevisiae, and its expression is regulated by the high-osmolarity glycerol response pathway. Mol Cell Biol 14:4135–4144
Banuelos MA, Sychrova H, Bleykasten-Grosshans C, Souciet JL, Potier S (1998) The Nha1 antiporter of Saccharomyces cerevisiae mediates sodium and potassium efflux. Microbiol SGD 144:2749–2758
Bill RM, Hedfalk K, Karlgren S, Mullins JGL, Rydstrom J, Hohmann S (2001) Analysis of the pore of the unusual major intrinsic protein channel, yeast Fps1p. J Biol Chem 276:36543–36549
Brett CL, Donowitz M, Rao R (2005) Evolutionary origins of eukaryotic sodium/proton exchangers. Am J Physiol Cell Physiol 288:C223–C239
Csonka LN, Hanson AD (1991) Prokaryotic osmoregulation: genetics and physiology. Annu Rev Microbiol 45:569–606
Hill JE, Myers AM, Koerner TJ, Tzagoloff A (1986) Yeast/E. coli shuttle vectors with multiple unique restriction sites. Yeast 2:163–167
Hohmann S, Mager WH (1997) Yeast stress responses. R.G. Landes Company, Austin, TX
Kinclova O, Ramos J, Potier S, Sychrova H (2001) Functional study of the Saccharomyces cerevisiae Nha1p C-terminus. Mol Microbiol 40:656–668
Mitsui K, Kamauchi S, Nakamura N, Inoue H, Kanazawa H (2004) A conserved domain in the tail region of the Saccharomyces cerevisiae Na+/H+ antiporter (Nha1p) plays important roles in localization and salinity-resistant cell-growth. J Biochem (Tokyo) 135:139–148
Nass R, Rao R (1999) The yeast endosomal Na+/H+ exchanger, Nhx1, confers osmotolerance following acute hypertonic shock. Microbiol SGD 145:3221–3228
Proft M, Struhl K (2004) MAP kinase-mediated stress relief that precedes and regulates the timing of transcriptional induction. Cell 118:351–361
Rodriguez-Navarro A (2000) Potassium transport in fungi and plants. Biochim Biophys A 1469:1–30
Simon E, Clotet J, Calero F, Ramos J, Arino J (2001) A screening for high copy suppressors of the sit4 hal3 synthetically lethal phenotype reveals a role for the yeast Nha1 antiporter in cell cycle regulation. J Biol Chem 276:29740–29747
Stotz A, Linder P (1990) The ADE2 gene from Saccharomyces cerevisiae: sequence and new vectors. Gene 95:91–98
Sychrova H (2004) Yeast as a model organism to study transport and homeostasis of alkali metal cations. Physiol Res 53:S91–S98
Sychrova H, Ramirez J, Pena A (1999) Involvement of Nha1 antiporter in regulation of intracellular pH in Saccharomyces cerevisiae. FEMS Microbiol Lett 171:167–172
Tamas MJ et al (1999) Fps1p controls the accumulation and release of the compatible solute glycerol in yeast osmoregulation. Mol Microbiol 31:1087–1104
Acknowledgement
This work was supported by Grant Agency AS CR (A5011407).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by S. Hohmann
Rights and permissions
About this article
Cite this article
Kinclova-Zimmermannova, O., Sychrova, H. Functional study of the Nha1p C-terminus: involvement in cell response to changes in external osmolarity. Curr Genet 49, 229–236 (2006). https://doi.org/10.1007/s00294-005-0050-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00294-005-0050-1