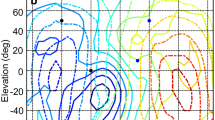
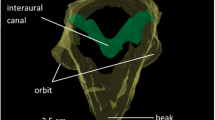
Abstract
The barn owl (Tyto alba) possesses several specializations regarding auditory processing. The most conspicuous features are the directionally sensitive facial ruff and the asymmetrically arranged ears. The frequency-specific influence of these features on sound has consequences for sound localization that might differ between low and high frequencies. Whereas the high-frequency range (>3 kHz) is well investigated, less is known about the characteristics of head-related transfer functions for frequencies below 3 kHz. In the present study, we compared 1/3 octaveband-filtered transfer functions of barn owls with center frequencies ranging from 0.5 to 9 kHz. The range of interaural time differences was 600 μs at frequencies above 4 kHz, decreased to 505 μs at 3 kHz and increased again to about 615 μs at lower frequencies. The ranges for very low (0.5–1 kHz) and high frequencies (5–9 kHz) were not statistically different. Interaural level differences and monaural gains increased monotonically with increasing frequency. No systematic influence of the body temperature on the measured localization cues was observed. These data have implications for the mechanism underlying sound localization and we suggest that the barn owl’s ears work as pressure receivers both in the high- and low-frequency ranges.








Similar content being viewed by others
Abbreviations
- FFT:
-
Fast Fourier transformation
- HRTF:
-
Head-related transfer function
- HRIR:
-
Head-related impulse response
- ITD:
-
Interaural time difference
- ILD:
-
Interaural level difference
References
Blauert J (1997) Spatial hearing. MIT Press, Cambridge
Brainard MS, Knudsen EI, Esterly SD (1992) Neural derivation of sound source location: resolution of spatial ambiguities in binaural cues. J Acoust Soc Am 91:1015–1027
Bühler P, Epple W (1980) The vocalizations of the barn owl. J Ornithol 121:36–70
Calford MB (1988) Constraints on the coding of sound frequency imposed by the avian interaural canal. J Comp Physiol A 162:491–502
Calford MB, Piddington RW (1988) Avian interaural canal enhances interaural delay. J Comp Physiol A 162:503–510
Campenhausen M, Wagner H (2006) Influence of the facial ruff on the sound-receiving characteristics of the barn owl’s ears. J Comp Physiol A 192:1073–1082
Christensen-Dalsgaard J, Manley GA (2005) Directionality of the lizard ear. J Exp Biol 208:1209–1217
Coles RB, Guppy A (1988) Directional hearing in the barn owl (Tyto alba). J Comp Physiol A 163:117–133
Dyson ML, Klump GM, Gauger B (1998) Absolute hearing thresholds and critical masking ratios in the european barn owl: a comparison with other owls. J Comp Physiol A 182:695–702
Egnor SEI (2001) Effects of binaural decorrelation on neural and behavioral processing of interaural level differences in the barn owl (Tyto alba). J Comp Physiol A 187:589–595
Fay RR, Feng AS (1987) Mechanisms for directional hearing among nonmammalian vertebrates. In: Yost WA, Gourevitch G (eds) Directional hearing. Springer, New York, pp 179–213
Harper NS, McAlpine D (2004) Optimal neural population coding of an auditory spatial cue. Nature 430:682–686
Hyson RL, Overholt EM, Lippe WR (1994) Cochlear microphonic measurements of interaural time differences in the chick. Hear Res 81:109–118
Johnston RF (1990) Variation in size and shape in pigeons, Columbia livia. Wilson Bull 102:213–225
Keller CH, Hartung K, Takahashi TT (1998) Head-related transfer functions of the barn owl: measurement and neural responses. Hear Res 118:13–34
Klump G (2000) Sound localization in birds. In: Dooling RJ, Popper AN, Fay RR (eds) Comparative hearing: birds and reptiles. Springer, New York
Knudsen EI (1981) The hearing of the barn owl. Sci Am 245:113–125
Konishi M (1973) Locatable and nonlocatable acoustic signals for barn owls. Am Nat 107:775–785
Köppl C (1997) Phase locking to high frequencies in the auditory nerve and cochlear nucleus magnocellularis of the barn owl, Tyto alba. J Neurosci 17:3312–3321
Köppl C, Gleich O (1997) Evoked cochlear potentials in the barn owl. J Comp Physiol A 193:601–612
Köppl C, Gleich O (2007) Evoked cochlear potentials in the barn owl. J Comp Physiol A 193(6):601–612
Larsen ON, Dooling RJ, Ryals BM (1997) Roles of intracranial air pressure in bird audition. In: Lewis ER, Long GR, Lyon RF, Narins PM, Steele CR, Hecht-Poinar E (eds) Diversity in auditory mechanics. World Scientific, Singapore, pp 11–17
Larsen ON, Dooling RJ, Michelsen A (2006) Role of pressure difference reception in the directional hearing of budgerigars (Melopsittacus undulatus). J Comp Physiol A 192:1063–1072
Lewald J (1990) The directionality of the ear of the pigeon (Columbia livia). J Comp Physiol A 167:533–543
Michelsen A, Larsen ON (2008) Pressure difference receiving ears. Bioinsp Biomim 3:1–18. doi:10.1088/1748-3182/3/1/011001
Moiseff A (1989) Binaural disparity cues available to the barn owl for sound localization. J Comp Physiol A 164:629–636
Moiseff A, Konishi M (1981a) The owl’s interaural pathway is not involved in sound localization. J Comp Physiol 144:299–304
Moiseff A, Konishi M (1981b) Neuronal and behavioral sensitivity to binaural time differences in the owl. J Neurosci 1:40–48
Okanoya K, Dooling RJ (1987) Hearing in passerine and psittacine birds: a comparative study of absolute and masked auditory thresholds. J Comp Psych 101:7–15
Poganiatz I, Wagner H (2001) Sound-localization experiments with barn owls in virtual space: influence of broadband interaural level difference on head-turning behavior. J Comp Physiol A 187:225–233
Poganiatz I, Nelken I, Wagner H (2001) Sound-localization experiments with barn owls in virtual space: influence of interaural time difference on head-turning behavior. JARO 2:1–21
Roulin A (2001) Food supply differentially affects sibling negotiation and competition in the barn owl (Tyto alba). Behav Ecol Sociobiol 49:514–519
Singheiser M, Plachta DTT, Brill S, Bremen P, van der Willigen R, Wagner H (2010) Target-approaching behavior of barn owls (Tyto alba): influence of sound frequency. J Comp Physiol A 196:227–240
Spezio ML, Keller CH, Marrocco RT, Takahashi TT (2000) Head-related transfer functions of the Rhesus monkey. Hear Res 144:73–88
Sterbing SJ, Hartung K, Hoffmann KP (2003) Spatial tuning to virtual sounds in the inferior colliculus of the guinea pig. J Neurophysiol 90:2648–2659
Tollin DJ, Koka K (2009) Postnatal development of sound pressure transformations by the head and pinnae of the cat: monaural characteristics. J Acoust Soc Am 125:980–994
Vonderschen K, Wagner H (2009) Tuning to interaural time difference and frequency differs between the auditory arcopallium and the external nucleus of the inferior colliculus. J Neurophysiol 101:2348–2361
Wada Y (1924) Beiträge zur vergleichenden Physiologie der Gehörorgane. Pflugers Arch Ges Physiol 202:46–69
Wagner H (1993) Sound-localization deficits induced by lesions in the barn owl’s auditory space map. J Neurosci 13:371–386
Wagner H, Mazer JA, von Campenhausen M (2002) Response properties of neurons in the core of the central nucleus of the inferior colliculus of the barn owl. Eur J Neurosci 15:1343–1352
Wagner H, Asadollahi A, Bremen P, Endler F, Vonderschen K, von Campenhausen M (2007) Distribution of interaural time difference in the barn owl’s inferior colliculus in the low- and high-frequency ranges. J Neurosci 27:4191–4200
Weber T, Thorson J, Huber F (1981) Auditory behaviour of the cricket. I. Dynamics of compensated walking and discrimination paradigms on the Kramer treadmill. J Comp Physiol A 141:215–232
Acknowledgments
We thank Frank Endler for help with the setup and stimulus software, and Gillian Melton for proofreading. Care and treatment of the owls were approved by the Landespräsidium für Natur, Umwelt und Verbraucherschutz Nordrhein-Westfalen, Recklinghausen, Germany, and principles of laboratory animal care (NIH publication No. 85-23, revised 1985) were followed.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hausmann, L., von Campenhausen, M. & Wagner, H. Properties of low-frequency head-related transfer functions in the barn owl (Tyto alba). J Comp Physiol A 196, 601–612 (2010). https://doi.org/10.1007/s00359-010-0546-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00359-010-0546-0