Summary
Background
Glycoxidation of collagens contributes to development of vascular complications in diabetes.
Aim of the study
Since flavonoids are potent antioxidants present in vegetal foods, it was interesting to examine their effect on the formation of a cross-linking advanced glycation endproduct, pentosidine, in collagens.
Methods
Collagen was incubated with glucose (250 mM), in the presence of different flavonoids. Pentosidine was measured by HPLC, hydroxyproline colorimetrically.
Results
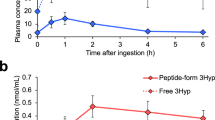
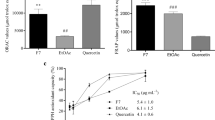
Monomeric flavonoids (25 and 250 µM) markedly reduced pentosidine/hydroxyproline values in a concentration- and structure-dependent manner. In decreasing order of their specific inhibitory activity, they rank as follows: myricetin ≥ quercetin > rutin > (+)catechin > kaempferol. Thus 3′-OH or 4-oxo + Delta2–3 increase the inhibitory activity; conjugation by Rha-Glc on 3-OH decreases it. Procyanidin oligomers from grape seed were more active than pine bark procyanidin oligomers: this may be related to the galloyl residues present in grape seed oligomers only. Procyanidin oligomers are known to be cleaved into monomers in the gastric milieu and monomeric flavonoids to be absorbed and recovered at micromolar concentrations (with a long plasmatic half-life) in extracellular fluids, in contact with collagens.
Conclusion
Flavonoids are very potent inhibitors of pentosidine formation in collagens. They are active at micromolar concentrations; these might be achieved in plasma of diabetic patients after oral intake of natural flavonoids.



Similar content being viewed by others
References
Aherne SA, O’Brien NM (2002) Dietary flavonols: chemistry, food content, and metabolism. Nutrition 18:75–81
Balant L, Burki B, Wermeille M, Golden G (1979) Comparison of some pharmacokinetic parameters of (+)-cyanidanol-3 obtained with specific and non-specific analytical methods. Arzneimitteforschung 29:1758–1762
Carando S, Teissedre P-L, Pascual-Martinez L, Cabanis J-C (1999) Levels of flavan-3-ols in french wines. J Agric Food Chem 47:4161–4166
Cervantes-Laurean D, Schramm DD, Jacobson EL, Halaweish I, Bruckner GG, Boissonneault GA (2006) Inhibition of advanced glycation end product formation on collagen by rutin and its metabolites. J Nutr Biochem 17:531–540
De Groot H, Rauen U (1998) Tissue injury by reactive oxygen species and the protective effects of flavonoids. Fundam Clin Pharmacol 12:249–255
De Pascual-Teresa S, Santos-Buelga C, Rivas-Gonzalo JC (2000) Quantitative analysis of flavan-3-ols in spanish foodstuffs and beverages. J Agric Food Chem 48:5331–5337
De Vries JH, Hollman PC, van Amersfoort I, Olthof MR, Katan MB (2001) Red wine is a poor source of bioavailable flavonols in men. J Nutr 131:745–748
Elgawish A, Glomb M, Friedlander MA, Monnier VM (1996) Involvement of hydrogen peroxide in collagen crosslinking by high glucose in vitro and in vivo. J Biol Chem 271:12964–12971
Fuleki T, Ricardo-Da-Silva JM (2003) Effects of cultivar and processing method on the contents of catechins and procyanidins in grape juice. J Agric Food Chem 51:640–646
Graefe EU, Wittig J, Mueller S, Tiethling AK, Uehleke B, Drewelow B, Pforte B, Jacobasch G, Derendorf H, Veit M (2001) Pharmacokinetics and bioavailability of quercetin glycosides in humans. J Clin Pharmacol 41:492–499
Gu L, Kelm M, Hammerstone JF, Beecher G, Holden J, Haytowitz D, Prior RL (2003) Screening of foods containing proanthocyanidins and their structural characterization using LC-MS/MS and thiolytic degradation. J Agric Food Chem 51:7513–7521
Hakkinen SH, Karenlampi SO, Heinonen M, Mykkanen HM, Torronen AR (1999) Content of the flavanols quercetin, myricetin, and kaempferol in 25 edibles berries. J Agric Food Chem 47:2274–2279
Hammerstone JF, Lazarus SA, Schmitz HH (2000) Procyanidin content and variation in some commonly consumed foods. J Nutr 130(Suppl):2086S–2092S
Hertog MGL, Hollman PCH, Katan MB (1992) Content of potentially anticarcinogenic flavonoids of 28 vegetables and 9 fruits commonly consumed in The Netherlands. J Agric Food Chem 40:2379–2383
Hertog MGL, Hollman PCH, van de Putte B (1993) Content of potentially anti-carcinogenic flavonoids of tea infusions, wines, and fruit juices. J Agric Food Chem 41:1242–1246
Holt RR, Lazarus SA, Cameron Sullards M, Yan Zhu Q, Schramm DD, Hammerstone JF, Fraga CG, Schmitz HH, Keen CL (2002) Procyanidin dimer B2 [epicatechin-(4b–8)-epicatechin] in human plasma after the consuption of a flavanol-rich cocoa. Am J Clin Nutr 76:798–804
Kiho T, Usui S, Hirano K, Aizawa K, Inakuma T (2004) Tomato paste fraction inhibiting the formation of advanced glycation end-products. Biosci Biotechnol Biochem 68: 200–205
Kris-Etherton PM, Lefevre M, Beecher GR, Gross MD, Keen CL, Etherton TD (2004) Bioactive compounds in nutrition and health-research methodologies for establishing biological function: the antioxidant and anti-inflammatory effects of flavonoids on atheroscerosis. Annu Rev Nutr 24:511–538
Lamuela-Raventos RM, Andres-Lacueva C, Permanyer J, Izquierdo-Pulido M (2001) More antioxidants in cocoa. J Nutr 131:834
Lee M-J, Wang Z-Y, Li H, Chen L, Sun Y, Gobbo S, Balentine DA, Yang CS (1995) Analysis of plasma and urinary tea polyphenols in human subjects. Cancer Epidemiol Biomark Prev 4:393–399
Leenen R, Roodenburg AJ, Tijburg LB, Wiseman SA (2000) A single dose of tea with or without milk increases plasma antioxidant activity in humans. Eur J Clin Nutr 54:87–92
Manach C, Scalbert A, Morand C, Remesy C, Jimenez L (2004) Polyphenols: food sources and bioaviability. Am J Clin Nutr 79:727–747
Manach C, Williamson G, Morand C, Scalbert A, Remesy C (2005) Bioavailability and bioefficacy of polyphenols in humans. I. Review of 97 bioavailability studies. Am J Nutr 81(Suppl):230S–242S
Matsuda H, Wang T, Managi H, Yoshikawa M (2003) Structural requirements of flavonoids for inhibition of protein glycation and radical scavenging activities. Bioorg Med Chem 11:5317–5323
Monnier VM, Bautista O, Kenny D, Sell DR, Fogarty J, Dahms W, Cleary PA, Lachin J, Genuth S, the DCCT Skin Collagen Ancillary Study Group (1999) Skin collagen glycation, glycoxidation, and cross-linking are lower in subjects with long-term intensive versus conventional therapy of type−1 diabetes: relevance of glycated collagen products versus HbA1c as markers of diabetic complications. Diabetes 48:870–880
Monnier V, Fogarty JF, Monnier CS, Sell DR (1999) Glycation, glycoxidation and other Maillard recation products. In: Byung Pal Yu (ed) Methods in aging research. CRC Press, New York, pp 657-681
Monnier VM, Glomb M, Elgawish A, Sell DR (1996) The mechanism of collagen cross-linking in diabetes: a puzzle nearing resolution. Diabetes 45(Suppl 3):S67–S72
Morimitsu Y, Yoshida K, Esaki S, Hirota H (1995) Protein glycation inhibitors from thyme (Thymus vulgaris). Biosci Biotech Biochem 59:2018–2021
Nakagawa K, Okuda S, Miyazawa T (1997) Dose-dependent incorporation of tea catechins, (-)-epigallocatechin-3-gallate and (-)-epigallocatechin, into human plasma. Biosci Biotechnol Biochem 61:1981–1985
Odetti PR, Borgoglio A, De Pascale A, Rolandi R, Adezati L (1990) Prevention of diabetes-increased aging effect on rat collagen-linked fluorescence by aminoguanidine and rutin. Diabetes 39:796–801
Olthof MR, Hollman PC, Vree TB, Katan MB (2000) Bioavailabilities of quercetin−3-glucoside and quercetin−4′-glucoside do not differ in humans. J Nutr 130:1200–1203
Pannala AS, Chan TS, O’Brien PJ, Rice-Evans CA (2001) Flavonoid B-ring chemistry and antioxidant activity: fast reaction kinetics. Biochem Biophys Res Commun 282:1161–1168
Plumb GW, De Pascual-Teresa S, Santos-Buelga C, Cheynier V, Williamson G (1998) Antioxidant properties of catechins and proanthocyanidins: effect of polymerisation, galloylation and glycosylation. Free Radic Res 29:351–358
Price DL, Rhett PM, Thorpe SR, Baynes JW (2001) Chelating activity of advanced glycation end-product inhibitors. J Biol Chem 276:48967–48972
Sano A, Yamakoshi J, Tokutake S, Tobe K, Kubota Y, Kikuchi M (2003) Procyanidin B1 is detected in human serum after intake of proanthocyanidin-rich grape seed extract. Biosci Biotechnol Biochem 67:1140–1143
Scalbert A, Williamson G (2000) Dietary intake and bioavailability of polyphenols. J Nutr 130:2073S–2085S
Sell DR, Lapolla A, Odetti P, Fogarty J, Monnier VM (1992) Pentosidine formation in skin correlates with severity of complications in individuals with long-standing IDDM. Diabetes 41:1286–1292
Spencer JP, Chaudry F, Pannala AS, Srai SK, Debnam E, Rice-Evans C (2000) Decomposition of cocoa procyanidins in the gastric milieu. Biochem Biophys Res Comm 272:236–241
Sternberg M, Borsos AM, Roux A, Adam C, Urios P (2002) Compared inhibition of pentosidine formation in type-I collagen incubated with glucose by catechin, myricetin, kaempferol and quercetin: role of flavonoid structure. Diabetologia 45(Suppl 2):393
Sternberg MC, M’Bemba J, Urios P, Borsos AM, Roux A, Adam C, Berne C, Selam JL, Slama G (2003) Skin-collagen pentosidine and fluorescence at 370/440 nm as markers of diabetes duration and complications in a french population. Diabetologia 46(Suppl 2):A406
Stitt A, Gardiner TA, Anderson NL, Canning P, Frizzell N, Duffy N, Boyle C, Januszewski AS, Chachich M, Baynes JW, Thorpe SR (2002) The AGE inhibitor pyridoxamine inhibits development of retinopathy in experimental diabetes. Diabetes 51:2826–2832
Tourino S, Selga A, Jimenez A, Julia L, Lozano C, Lizarraga D, Cascante M, Torres JL (2005) Procyanidin fractions from pine (Pinus pinaster) bark: radical scavenging power in solution, antioxidant activity in emulsion, and antiproliferative effect in melanoma cells. J Agric Food Chem 53:4728–4735
Urios P, Grigorova-Borsos AM, Feing-Kwong-Chan S, Sternberg M (2000a) Quercetin and rutin inhibit pentosidine formation on type-I collagen incubated with glucose. Exp Clin Endocrinol Diabetes 18/5:A11
Urios P, Kassab I, Borsos AM, Guillot R, Peyroux J, Sternberg M (2000b) Long-term treatment with a purified micronized flavonoid fraction reduces urinary albumin clearance and restores albuminemia in normotensive and hypertensive diabetic rats. Diabetes Res Clin Pract 50, Suppl 1: S362
Vertommen J, Van den Enden M, Simoens L, De Leew I (1994) Flavonoid treatment reduces glycation and lipid peroxidation in experimental diabetic rats. Phytother Res 8:430–432
Waterhouse AL (2002) Wine phenolics. Ann NY Acad Sci 957:21–36
Woessner JF (1961) The determination of hydroxyproline and protein samples containing small proportions of this imino acid. Arch Biochem Biophys 93:440–447
Wu C-H, Yen G-C (2005) Inhibitory effect of naturally occuring flavonoids on the formation of advanced glycation endproducts. J Agric Food Chem 53:3167–3173
Yang Y, Chien M (2000) Characterization of grape procyanidins using high-performance liquid chromatography/mass spectrometry and matrix-assisted laser desorption/ionization time-of-flight mass spectrometry. J Agric Food Chem 48:3990–3996
Yokozawa T, Nakagawa T (2004) Inhibitory effects of Luobuma tea and its components against glucose-mediated protein damage. Food Chem Toxicol 42:975–981
Acknowledgements
This work was supported by the “Fondation pour la Recherche Médicale” and “Naturalia et Biologia”. We thank J. Peyroux for his help and critical reviewing, S. Feing, J. Garaud, A. Roux and C. Adam for their technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Urios, P., Grigorova-Borsos, AM. & Sternberg, M. Flavonoids inhibit the formation of the cross-linking AGE pentosidine in collagen incubated with glucose, according to their structure. Eur J Nutr 46, 139–146 (2007). https://doi.org/10.1007/s00394-007-0644-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-007-0644-0