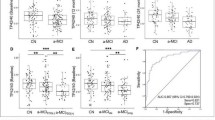
Abstract
Previous studies of Aβ plasma as a biomarker for Alzheimer’s disease (AD) obtained conflicting results. We here included 715 subjects with baseline Aβ1-40 and Aβ1-42 plasma measurement (50% with 4 serial annual measurements): 205 cognitively normal controls (CN), 348 patients mild cognitive impairment (MCI) and 162 with AD. We assessed the factors that modified their concentrations and correlated these values with PIB PET, MRI and tau and Aβ1-42 measures in cerebrospinal fluid (CSF). Association between Aβ and diagnosis (baseline and prospective) was assessed. A number of health conditions were associated with altered concentrations of plasma Aβ. The effect of age differed according to AD stage. Plasma Aβ1-42 showed mild correlation with other biomarkers of Aβ pathology and were associated with infarctions in MRI. Longitudinal measurements of Aβ1-40 and Aβ1-42 plasma levels showed modest value as a prognostic factor for clinical progression. Our longitudinal study of complementary measures of Aβ pathology (PIB, CSF and plasma Aβ) and other biomarkers in a cohort with an extensive neuropsychological battery is significant because it shows that plasma Aβ measurements have limited value for disease classification and modest value as prognostic factors over the 3-year follow-up. However, with longer follow-up, within subject plasma Aβ measurements could be used as a simple and minimally invasive screen to identify those at increased risk for AD. Our study emphasizes the need for a better understanding of the biology and dynamics of plasma Aβ as well as the need for longer term studies to determine the clinical utility of measuring plasma Aβ.





Similar content being viewed by others
References
(1998) Consensus report of the Working Group on: “Molecular and Biochemical Markers of Alzheimer’s Disease”. The Ronald and Nancy Reagan Research Institute of the Alzheimer’s Association and the National Institute on Aging Working Group. Neurobiology of aging 19(2): 109–116
Alexander G, Furey M, Grady C et al (1997) Association of premorbid intellectual function with cerebral metabolism in Alzheimer’s disease: implications for the cognitive reserve hypothesis. Am J Psychiatry 154(2):165–172
Arvanitakis Z, Lucas JA, Younkin LH, Younkin SG, Graff-Radford NR (2002) Serum creatinine levels correlate with plasma amyloid [beta] protein. Alzheimer Dis Assoc Disord 16(3):187–190
Blasko I, Kemmler G, Krampla W et al (2005) Plasma amyloid [beta] protein 42 in non-demented persons aged 75 years: effects of concomitant medication and medial temporal lobe atrophy. Neurobiol Aging 26(8):1135–1143
Blennow K, De Meyer G, Hansson O et al (2009) Evolution of Abeta42 and Abeta40 levels and Abeta42/Abeta40 ratio in plasma during progression of Alzheimer’s disease: a multicenter assessment. J Nutr Health Aging 13(3):205–208
Butters N, Granholm E, Salmon D, Grant I, Wolfe J (1987) Episodic and semantic memory: a comparison of amnesic and demented patients. J Clin Exp Neuropsychol 9(5):479–497
Carmichael O, Schwarz C, Drucker D et al (2010) Longitudinal changes in white matter disease and cognition in the first year of the alzheimer disease neuroimaging initiative. Arch Neurol 67(11):1370–1378
Cosentino SA, Stern Y, Sokolov E et al (2010) Plasma {beta}-amyloid and cognitive decline. Arch Neurol 67(12):1485–1490
Chen M, Inestrosa NC, Ross GS, Fernandez HL (1995) Platelets are the primary source of amyloid [beta]-peptide in human blood. Biochem Biophys Res Commun 213(1):96–103
Davatzikos C, Bhatt P, Shaw LM, Batmanghelich KN and Trojanowski JQ (2010) Prediction of MCI to AD conversion, via MRI, CSF biomarkers, and pattern classification. Neurobiol aging
de Souza LC, Lamari F, Belliard S et al (2011) Cerebrospinal fluid biomarkers in the differential diagnosis of Alzheimer’s disease from other cortical dementias. J Neurol Neurosurg Psychiatry 82(3):240–246
Devanand DP, Schupf N, Stern Y et al (2011) Plasma Aβ and PET PiB binding are inversely related in mild cognitive impairment. Neurology
Fagan AM, Mintun MA, Shah AR et al (2009) Cerebrospinal fluid tau and ptau(181) increase with cortical amyloid deposition in cognitively normal individuals: implications for future clinical trials of Alzheimer’s disease. EMBO Mol Med 1(8–9):371–380
Fagan AM, Shaw LM, Xiong C et al (2011) Comparison of Analytical Platforms for Cerebrospinal Fluid Measures of {beta}-Amyloid 1-42, Total tau, and P-tau181 for Identifying Alzheimer Disease Amyloid Plaque Pathology. Arch Neurol
Fei M, Jianghua W, Rujuan M, Wei Z, Qian W (2011) The relationship of plasma A[beta] levels to dementia in aging individuals with mild cognitive impairment. J Neurol Sci 305(1–2):92–96
Folstein MF, Folstein SE, McHugh PR (1975) Mini-mental state. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 12(3):189–198
Fukumoto H, Tennis M, Locascio JJ et al (2003) Age but not diagnosis is the main predictor of plasma amyloid {beta}-protein levels. Arch Neurol 60(7):958–964
Goldberg KM, Iglewicz B (1992) Bivariate extensions of the boxplot. Technometrics 34:307–320
Goodglass H, Kaplan E (1983) The assessment of aphasia and related disorders. Lea & Febiger, Philadelphia
Graff-Radford NR, Crook JE, Lucas J et al (2007) Association of low plasma Abeta42/Abeta40 Ratios with increased imminent risk for mild cognitive impairment and alzheimer disease. Arch Neurol 64(3):354–362
Gurol ME, Irizarry MC, Smith EE et al (2006) Plasma β-amyloid and white matter lesions in AD, MCI, and cerebral amyloid angiopathy. Neurology 66(1):23–29
Ikonomovic MD, Klunk WE, Abrahamson EE et al (2008) Post-mortem correlates of in vivo PiB-PET amyloid imaging in a typical case of Alzheimer’s disease. Brain 131(6):1630–1645
Jack CR Jr, Bernstein MA, Fox NC et al (2008) The Alzheimer’s disease neuroimaging initiative (ADNI): MRI methods. J Magn Reson Imaging 27(4):685–691
Jack CR Jr, Knopman DS, Jagust WJ et al (2010) Hypothetical model of dynamic biomarkers of the Alzheimer’s pathological cascade. Lancet Neurol 9(1):119–128
Jagust WJ, Bandy D, Chen K et al (2010) The Alzheimer’s disease neuroimaging Initiative positron emission tomography core. Alzheimers Dement 6(3):221–229
Kaplan E, Goodglass H, Weintraub S (1983) Boston naming test. Lea & Febiger, Philadelphia
Lachno DR, Vanderstichele H, De Groote G et al (2009) The influence of matrix type, diurnal rhythm and sample collection and processing on the measurement of plasma beta-amyloid isoforms using the INNO-BIA plasma Abeta forms multiplex assay. J Nutr Health Aging 13(3):220–225
Lambert J-C, Schraen-Maschke S, Richard F et al (2009) Association of plasma amyloid β with risk of dementia. Neurology 73(11):847–853
Lambert JC, Dallongeville J, Ellis KA et al (2011) Association of plasma aβ peptides with blood pressure in the elderly. PLoS ONE 6(4):e18536
Laske C, Sopova K, Gkotsis C et al (2010) Amyloid-β peptides in plasma and cognitive decline after 1 year follow-up in alzheimer’s disease patients. J Alzheimers Dis 21(4):1263–1269
Lewczuk P, Kornhuber J, Vanmechelen E et al (2010) Amyloid beta peptides in plasma in early diagnosis of Alzheimer’s disease: a multicenter study with multiplexing. Exp Neurol 223(2):366–370
Locascio JJ, Fukumoto H, Yap L et al (2008) Plasma amyloid {beta}-protein and c-reactive protein in relation to the rate of progression of Alzheimer disease. Arch Neurol 65(6):776–785
Lopez OL, Kuller LH, Mehta PD et al (2008) Plasma amyloid levels and the risk of AD in normal subjects in the Cardiovascular Health Study. Neurology 70(19):1664–1671
Lui JK, Laws SM, Li Q-X et al (2010) Plasma amyloid-β as a biomarker in Alzheimer’s disease: the AIBL study of aging. J Alzheimers Dis 20(4):1233–1242
Mathis CA, Wang Y, Holt DP et al (2003) Synthesis and Evaluation of 11C-Labeled 6-Substituted 2-Arylbenzothiazoles as Amyloid Imaging Agents. J Med Chem 46(13):2740–2754
Mayeux R, Tang M-X, Jacobs DM et al (1999) Plasma amyloid β-peptide 1-42 and incipient Alzheimer’s disease. Ann Neurol 46(3):412–416
Mayeux R, Honig LS, Tang M-X et al (2003) Plasma Aβ40 and Aβ42 and Alzheimer’s disease: relation to age, mortality, and risk. Neurology 61(9):1185–1190
Mehta PD, Pirttila T, Patrick BA, Barshatzky M, Mehta SP (2001) Amyloid [beta] protein 1-40 and 1-42 levels in matched cerebrospinal fluid and plasma from patients with Alzheimer disease. Neurosci Lett 304(1–2):102–106
Petersen RC, Aisen PS, Beckett LA et al (2010) Alzheimer’s disease neuroimaging initiative (ADNI): clinical characterization. Neurology 74(3):201–209
R Development Core Team (2010) R: A language and environment for statistical computing R Foundation for Statistical Computing, Vienna, Austria. ISBN 3-900051-07-0. http://www.R-project.org/
Reitan R (1958) Validity of the trail making test as an indicator of organic brain damage. Percept Mot Skills 8:271–276
Rentz DM, Locascio JJ, Becker JA et al (2010) Cognition, reserve, and amyloid deposition in normal aging. Ann Neurol 67(3):353–364
Rey A (1964) L’examen clinique en psychologie. Presses Universitaires de France, Paris
Rosen W, Mohs R, Davis K (1984) A new rating scale for Alzheimer’s disease. Am J Psychiatry 141(11):1356–1364
Ryan JR, Paolo AM (1992) A screening procedure for estimating premorbid intelligence in the elderly. Clin Neuropsychol 6(1):53–62
Schupf N, Tang MX, Fukuyama H et al (2008) Peripheral Aβ subspecies as risk biomarkers of Alzheimer’s disease. Proc Nat Acad Sci 105(37):14052–14057
Schupf N, Zigman WB, Tang M-X et al (2010) Change in plasma Aβ peptides and onset of dementia in adults with Down syndrome. Neurology 75(18):1639–1644
Seppälä TT, Herukka S-K, Hänninen T et al (2010) Plasma Aβ42 and Aβ40 as markers of cognitive change in follow-up: a prospective, longitudinal, population-based cohort study. J Neurol Neurosurg Psychiatry 81(10):1123–1127
Shaw LM, Vanderstichele H, Knapik-Czajka M et al (2009) Cerebrospinal fluid biomarker signature in Alzheimer’s disease neuroimaging initiative subjects. Ann Neurol 65(4):403–413
Shaw LM, Vanderstichele H, Knapik-Czajka M et al (2011) Qualification of the analytical and clinical performance of CSF biomarker analyses in ADNI. Acta Neuropathol 121(5):597–609
Song F, Poljak A, Valenzuela M, et al (2011) Meta-Analysis of Plasma Amyloid-beta levels in Alzheimer’s Disease. J Alzheimers Dis (1875-8908 (Electronic))
Sundelof J, Giedraitis V, Irizarry MC et al (2008) Plasma beta amyloid and the risk of alzheimer disease and dementia in elderly men: a prospective, population-based cohort study. Arch Neurol 65(2):256–263
Taylor K, Salmon D, Rice V et al (1996) Longitudinal examination of american national adult reading test (AMNART) performance in dementia of the Alzheimer type (DAT): validation and correction based on degree of cognitive decline. J Clin Exp Neuropsychol 18(6):883–891
Van Dijk EJ, Prins ND, Vermeer SE et al (2004) Plasma amyloid β, apolipoprotein E, lacunar infarcts, and white matter lesions. Ann Neurol 55(4):570–575
van Oijen M, Hofman A, Soares HD, Koudstaal PJ, Breteler MM (2006) Plasma Abeta(1-40) and Abeta(1-42) and the risk of dementia: a prospective case-cohort study. Lancet Neurol 5(8):655–660
Wechsler D (1987) Wechsler Memory Scale. Rev ed. Psychological Corp, San Antonio
Weigand SD, Vemuri P, Wiste HJ et al (2011) Transforming cerebrospinal fluid Abeta42 measures into calculated Pittsburgh compound B units of brain Abeta amyloid. Alzheimers Dement 7(2):133–141
Wilcox RR, Schönbrodt FD (2009) The WRS package for robust statistics in R (version 0.12.1). http://r-forge.r-project.org/projects/wrs/
Yaffe K, Weston A, Graff-Radford NR et al (2011) Association of plasma β-amyloid level and cognitive reserve with subsequent cognitive decline. JAMA 305(3):261–266
Acknowledgments
We thank our ADNI colleagues for their contributions to the work summarized here which has been supported mainly by the ADNI U01 AG024904. ADNI is funded by the National Institute of Aging, the National Institute of Biomedical Imaging and Bioengineering (NIBIB), and the Foundation for the National Institutes of Health, through generous contributions from the following companies and organizations: Pfizer Inc., Wyeth Research, Bristol-Myers Squibb, Eli Lilly and Company, GlaxoSmithKline, Merck & Co. Inc., AstraZeneca AB, Novartis Pharmaceuticals Corporation, the Alzheimer’s Association, Eisai Global Clinical Development, Elan Corporation plc, Forest Laboratories, and the Institute for the Study of Aging (ISOA), with participation from the U.S. Food and Drug Administration. Other support has come from AG10124 and the Marian S. Ware Alzheimer Program. VMYL is the John H. Ware 3rd Professor for Alzheimer’s Disease Research and JQT is the William Maul Measy-Truman G. Schnabel Jr. M.D. Professor of Geriatric Medicine and Gerontology. We thank the ADNI Biomarker Core for the analyses. We thank Donald Baldwin and the Molecular Diagnosis Genotyping Facility at the University of Pennsylvania Medical Center for provision of the APOε genotyping data. J.B.T.’s work was supported by a grant from the Alfonso Martín Escudero foundation. L.S.’s, C.J.’s and M.W.’s work is partially supported by NIH.
Conflict of interest
J.Q.T., V.M.Y.L., A.W.T., S.X.X., E.T., M.F., M.W., C.J., P.A., W.J. and J.B.T. have no conflicts of interest. L.S. belongs to the advisory board of Innogenetics Technical. H.V. works at INNX-Fujirebio.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Toledo, J.B., Vanderstichele, H., Figurski, M. et al. Factors affecting Aβ plasma levels and their utility as biomarkers in ADNI. Acta Neuropathol 122, 401–413 (2011). https://doi.org/10.1007/s00401-011-0861-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-011-0861-8