Abstract
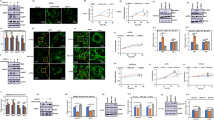
The bioflavonoid quercetin has long been known to exert anti-tumor effects, although the underlying mechanisms remain unknown. Investigation of the potential interference of this anti-oxidant with the efficacy of cell stress-inducing anti-cancer drugs revealed extensive intracellular vacuolation induced by quercetin in epithelial cancer cells that led to cell cycle arrest and ensuing apoptosis. Accumulation of biomarkers of autophagy, including fluorescent autophagy markers and acidotropic dyes characterized these vacuoles as phagolysosomes. Prior to the formation of autophagosomes, an immediate and pronounced inhibition of the autophagy-controlling mTOR activity in quercetin-treated cancer cells occurred, accompanied by a marked reduction in the phosphorylation of the mTOR substrates 4E-BP1 and p70S6 kinase. Assessment of cellular proteasome activity revealed an effective and immediate inhibition of the activity of the proteasome by quercetin in cancer cells. In addition to the formation of autophagosomes, accumulation of poly-ubiquitinated protein aggregates was observed. Thus, proteasome inhibition by quercetin can be regarded as a major cause of quercetin-induced cancer cell death. These results suggest potential new applications for quercetin in cancer science and identify quercetin as an easy-to-handle agent to study proteasome activity, mTOR signaling and autophagy in human cancer cells for cell biological purposes.








Similar content being viewed by others
References
Berkers CR, Verdoes M, Lichtman E, Fiebiger E, Kessler BM, Anderson KC, Ploegh HL, Ovaa H, Galardy PJ (2005) Activity probe for in vivo profiling of the specificity of proteasome inhibitor bortezomib. Nat Methods 2:357–362
Bernales S, McDonald KL, Walter P (2006) Autophagy counterbalances endoplasmic reticulum expansion during the unfolded protein response. PLoS Biol 4:e423
Bischoff SC (2008) Quercetin: potentials in the prevention and therapy of disease. Curr Opin Clin Nutr Metab Care 11:733–740
Boots AW, Haenen GR, Bast A (2008) Health effects of quercetin: from antioxidant to nutraceutical. Eur J Pharmacol 585:325–337
Brancolini C (2008) Inhibitors of the ubiquitin-proteasome system and the cell death machinery: How many pathways are activated? Curr Mol Pharmacol 1:24–37
Brüning A (2011) Analysis of nelfinavir-induced endoplasmic reticulum stress. Methods Enzymol 491C:127–142
Brüning A, Burger P, Vogel M, Rahmeh M, Friese K, Lenhard M, Burges A (2008) Bortezomib treatment of ovarian cancer cells mediates endoplasmic reticulum stress, cell cycle arrest, and apoptosis. Invest New Drugs doi:101007/s10637-008-9206-4
Brüning A, Friese K, Burges A, Mylonas I (2010a) Tamoxifen enhances the cytotoxic effects of nelfinavir in breast cancer cells. Breast Cancer Res 12:R45
Brüning A, Gingelmaier A, Friese K, Mylonas I (2010b) New prospects for nelfinavir in non-HIV-related diseases. Curr Mol Pharmacol 3:91–97
Brüning A, Vogel M, Mylonas I, Friese K, Burges A (2011) Bortezomib targets the caspase-like proteasome activity in cervical cancer cells, triggering apoptosis that can be enhanced by nelfinavir. Current Cancer Drug Targets 11:799–809
Buchberger A, Bukau B, Sommer T (2010) Protein quality control in the cytosol and the endoplasmic reticulum: brothers in arms. Mol Cell 40:238–252
Cecconi F, Levine B (2008) The role of autophagy in mammalian development: cell makeover rather than cell death. Dev Cell 15:344–357
Chen D, Frezza M, Schmitt S, Kanwar J, Dou QP (2011) Bortezomib as the first proteasome inhibitor anticancer drug: current status and future perspectives. Curr Cancer Drug Targets 11:239–253
Ciechanover A (2005) Proteolysis: from the lysosome to ubiquitin and the proteasome. Nat Rev Mol Cell Biol 6:79–87
Curran MP, McKeage K (2009) Bortezomib: a review of its use in patients with multiple myeloma. Drugs 69:859–888
Dechsupa S, Kothan S, Vergote J, Leger G, Martineau A, Berangeo S, Kosanlavit R, Moretti JL, Mankhetkorn S (2007) Quercetin, Siamois 1 and Siamois 2 induce apoptosis in human breast cancer MDA-mB-435 cells xenograft in vivo. Cancer Biol Ther 6(1):56–61
Gilardini A, Marmiroli P, Cavaletti G (2008) Proteasome inhibition: a promising strategy for treating cancer, but what about neurotoxicity? Curr Med Chem 15:3025–3035
Glick D, Barth S, Macleod KF (2010) Autophagy: cellular and molecular mechanisms. J Pathol 221:3–12
Hayashi-Nishino M, Fujita N, Noda T, Yamaguchi A, Yoshimori T, Yamamoto A (2009) A subdomain of the endoplasmic reticulum forms a cradle for autophagosome formation. Nat Cell Biol 11:1433–1437
Johnston JA, Ward CL, Kopito RR (1998) Aggresomes: a cellular response to misfolded proteins. J Cell Biol 143:1883–1898
Kim EJ, Choi CH, Park JY, Kang SK, Kim YK (2008) Underlying mechanism of quercetin-induced cell death in human glioma cells. Neurochem Res 33(6):971–979
Kostenko S, Moens U (2009) Heat shock protein 27 phosphorylation: kinases, phosphatases, functions and pathology. Cell Mol Life Sci 66:3289–3307
Kroemer G, Mariño G, Levine B (2010) Autophagy and the integrated stress response. Mol Cell 40:280–293
Lafarga M, Berciano MT, Pena E, Mayo I, Castaño JG, Bohmann D, Rodrigues JP, Tavanez JP, Carmo-Fonseca M (2002) Clastosome: a subtype of nuclear body enriched in 19S and 20S proteasomes, ubiquitin, and protein substrates of proteasome. Mol Biol Cell 13(8):2771–2782
Mizushima N, Levine B, Cuervo AM, Klionsky DJ (2008) Autophagy fights disease through cellular self-digestion. Nature 451:1069–1075
Moretti L, Cha YI, Niermann KJ, Lu B (2007) Switch between apoptosis and autophagy: radiation-induced endoplasmic reticulum stress? Cell Cycle 6:793–798
Murakami A, Ashida H, Terao J (2008) Multitargeted cancer prevention by quercetin. Cancer Lett 269:315–325
Navon A, Ciechanover A (2009) The 26 S proteasome: from basic mechanisms to drug targeting. J Biol Chem 284:33713–33718
Nguyen TT, Tran E, Nguyen TH, Do PT, Huynh TH, Huynh H (2004) The role of activated MEK-ERK pathway in quercetin-induced growth inhibition and apoptosis in A549 lung cancer cells. Carcinogenesis 25:647–659
Psahoulia FH, Moumtzi S, Roberts ML, Sasazuki T, Shirasawa S, Pintzas A (2007) Quercetin mediates preferential degradation of oncogenic Ras and causes autophagy in Ha-RAS-transformed human colon cells. Carcinogenesis 28:1021–1031
Savulescu AF, Rotem A, Harel A (2011) Proteasomes crossing the nuclear border. Nucleus 2(4)
Uchiyama Y, Shibata M, Koike M, Yoshimura K, Sasaki M (2008) Autophagy-physiology and pathophysiology. Histochem Cell Biol 129:407–420
Wu WK, Sakamoto KM, Milani M, Aldana-Masankgay G, Fan D, Wu K, Lee CW, Cho CH, Yu J, Sung JJ (2010) Macroautophagy modulates cellular response to proteasome inhibitors in cancer therapy. Drug Resist Update 13:87–92
Yang Z, Klionsky DJ (2010) Mammalian autophagy: core molecular machinery and signaling regulation. Curr Opin Cell Biol 22:124–131
Zhou X, Ikenoue T, Chen X, Li L, Inoki K, Guan KL (2009) Rheb controls misfolded protein metabolism by inhibiting aggresome formation and autophagy. Proc Natl Acad Sci USA 106:8923–8928
Acknowledgments
The generous supply of bortezomib by Millenium Phamaceuticals, Cambridge, MA, USA, and the generous supply of nelfinavir by Pfizer, Groton, CT, USA, are gratefully appreciated. The authors are much obliged to the highly skilful and motivated technical assistance of Petra Burger (DNA fragmentation assay), Martina Rahmeh (proteasome assay analysis) and Marianne Vogel (FACScan analysis).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Klappan, A.K., Hones, S., Mylonas, I. et al. Proteasome inhibition by quercetin triggers macroautophagy and blocks mTOR activity. Histochem Cell Biol 137, 25–36 (2012). https://doi.org/10.1007/s00418-011-0869-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-011-0869-0