Abstract
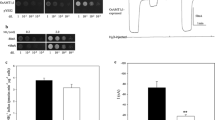
Lsi1 (OsNIP2;1) is the first silicon (silicic acid) transporter identified in plant, which belongs to the nodulin 26-like intrinsic membrane protein (NIP) subfamily. In this study, we characterized the function of this transporter by using the Xenopus laevis oocyte expression system. The transport activity of Lsi1 for silicic acid was significantly inhibited by HgCl2 but not by low temperature. Lsi1 also showed an efflux transport activity for silicic acid. The substrate specificity study showed that Lsi1 was able to transport urea and boric acid; however, the transport activity for silicic acid was not affected by the presence of equimolar urea and was decreased only slightly by boric acid. Furthermore, among the NIPs subgroup, OsNIP2;2 showed transport activity for silicic acid, whereas OsNIP1;1 and OsNIP3;1 did not. We propose that Lsi1 and its close homologues form a unique subgroup of NIP with a distinct ar/R selectivity filter, which is located in the narrowest region on the extra-membrane mouth and govern the substrate specificity of the pore.








Similar content being viewed by others
References
Sripanyakorn S, Jugdaohsingh R, Thompson RPH, Powell JJ (2005) Dietary silicon and bone health. Nutr Bull 30:222–230
Ma JF, Yamaji N (2006) Silicon uptake and accumulation in higher plants. Trends Plant Sci 11:392–397
Takahashi E, Hino K (1978) Silica uptake by plant with special reference to the forms of dissolved silica. J Soil Sci Manure Jpn 49:357–360
Ma JF, Tamai K, Yamaji N, Mitani N, Konishi S, Katsuhara M, Ishiguro M, Murata Y, Yano M (2006) A silicon transporter in rice. Nature 440:688–691
Yamaji N, Ma JF (2007) Spatial distribution and temporal variation of the rice silicon transporter Lsi1. Plant Physiol 143:1306–1313
Ma JF, Tamai K, Ichii M, Wu GF (2002) A rice mutant defective in Si uptake. Plant Physiol 130:2111–2117
Wallace IS, Roberts DM (2004) Homology modeling of representative subfamilies of Arabidopsis major intrinsic proteins. Classification based on the aromatic/arginine selectivity filter. Plant Physiol 135:1059–1068
Forrest KL, Bhave M (2007) Major intrinsic proteins (MIPs) in plants: a complex gene family with major impacts on plant phenotype. Funct Integr Genomics 7:263–289
Dean RM, Rivers RL, Zeidel ML, Roberts DM (1999) Purification and functional reconstitution of soybean nodulin 26. An aquaporin with water and glycerol transport properties. Biochemistry 38:347–353
Choi WG, Roberts DM (2007) Arabidopsis NIP2;1: a major intrinsic protein transporter of lactic acid induced by anoxic stress. J Biol Chem 282:24209–24218
Wallace IS, Roberts DM (2005) Distinct transport selectivity of two structural subclasses of the nodulin-like intrinsic protein family of plant aquaglyceroporin channels. Biochemistry 44:16826–16834
Takano J, Wada M, Ludewig U, Schaaf G, von Wiren N, Fujiwara T (2006) The arabidopsis major intrinsic protein NIP5;1 is essential for efficient boron uptake and plant development under boron limitation. Plant Cell 18:1498–1509
Preston GM, Carroll TP, Guggino WB, Agre P (1992) Appearance of water channels in Xenopus oocytes expressing red cell CHIP28 protein. Science 256:385–387
Ma JF, Goto S, Tamai K, Ichii M (2001) Role of root hairs and lateral roots in silicon uptake by rice. Plant Physiol 127:1773–1780
Ma JF, Yamaji N, Mitani N, Tamai K, Konishi S, Fujiwara T, Katsuhara M, Yano M (2007) An efflux transporter of silicon in rice. Nature 448:209–212
Tamai K, Ma JF (2003) Characterization of silicon uptake by rice roots. New Phytol 158:431–436
Daniels MJ, Chaumont F, Erik Mirkov T, Chrispeels MJ (1996) Characterization of a new vacuolar membrane aquaporin sensitive to mercury at a unique site. Plant Cell 8:587–599
Agre P, Kozono D (2003) Aquaporin water channels: molecular mechanisms for human diseases. FEBS Lett 555:72–78
Maurel C (2007) Plant aquaporins: novel functions and regulation properties. FEBS Lett 581:2227–2236
Tajkhorshid E, Nollert P, Jensen MO, Miercke LJ, O'Connell J, Stroud RM, Schulten K (2002) Control of the selectivity of the aquaporin water channel family by global orientational tuning. Science 296:525–530
Ilan B, Tajkhorshid E, Schulten K, Voth GA (2003) Electrostatic tuning of permeation and selectivity in aquaporin water channels. Biophys J 85:2884–2899
Wallace IS, Choi WG, Roberts DM (2006) The structure, function and regulation of the nodulin 26-like intrinsic protein family of plant aquaglyceroporins. Biochim Biophys Acta 1758:1165–1175
Thomas D, Bron P, Ranchy G, Duchesne L, Cavalier A, Rolland JP, Raguenes-Nicol C, Hubert JF, Haase W, Delamarche C (2002) Aquaglyceroporins, one channel for two molecules. Biochim Biophys Acta 1555:181–186
Wang Y, Schulten K, Tajkhorshid E (2005) What makes an aquaporin a glycerol channel? A comparative study of AqpZ and GlpF. Structure 13:1107–1118
Wu B, Beitz E (2007) Aquaporins with selectivity for unconventional permeants. Cell Mol Life Sci 64:2413–2421
Johansson I, Karlsson M, Shukla VK, Chrispeels MJ, Larsson C, Kjellbom P (1998) Water transport activity of the plasma membrane aquaporin PM28A is regulated by phosphorylation. Plant Cell 10:451–459
Guenther JF, Chanmanivone N, Galetovic MP, Wallace IS Cobb JA, Roberts DM (2003) Phosphorylation of soybean nodulin 26 on serine 262 enhances water permeability and is regulated developmentally and by osmotic signals. Plant Cell 15:981–991
Katsuhara M, Akiyama Y, Koshio K, Shibasaka M, Kasamo K (2002) Functional analysis of water channels in barley roots. Plant Cell Physiol 43:85–93
Weaver CD, Crombie B, Stacey G, Roberts DM (1991) Calcium-dependent phosphorylation of symbiosome membrane proteins from nitrogen-fixing soybean nodules: evidence for phosphorylation of nodulin-26. Plant Physiol 95:222–227
Weaver CD, Roberts DM (1992) Determination of the site of phosphorylation of nodulin 26 by the calcium-dependent protein kinase from soybean nodules. Biochemistry 31:8954–8959
Ma JF, Takahashi E (2002) Soil, Fertilizer, and Plant Silicon Research in Japan. Elsevier Science, Amsterdam
Acknowledgment
This research was supported by a grant-in-aid for Scientific Research on Priority Areas from the Ministry of Education, Culture, Sports, Science and Technology of Japan (nos. 15380053 and 17078008 to J.F.M.) and a grant from the Ministry of Agriculture, Forestry and Fisheries of Japan (Rice Genome Project IP-5003 to J.F.M.). We thank Dr. Maki Katsuhara and Dr. Nobuyuki Uozumi for instruction in oocyte assay. We also thank Fang Jie Zhao for his critical reading of this paper.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mitani, N., Yamaji, N. & Ma, J.F. Characterization of substrate specificity of a rice silicon transporter, Lsi1. Pflugers Arch - Eur J Physiol 456, 679–686 (2008). https://doi.org/10.1007/s00424-007-0408-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-007-0408-y