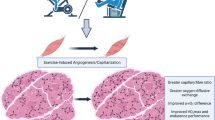
Abstract
The dynamic biochemical and mechanical environment around blood vessels during muscle activity generates powerful stimuli for vascular remodelling. Ultimately, this must lead to a coordinated expansion of various elements of the cardiovascular system in order to support enhanced aerobic exercise. Vascular endothelial growth factor plays a central role, and understanding how this is regulated in vivo by changes in transcription and stability of mRNA, production of protein and interaction with other growth factors, is a continuing challenge. Exercise hyperaemia leads to an increase in microvascular shear stress, which stimulates endothelial release of nitric oxide, whilst proteolytic modification of the extracellular matrix is induced by mechanical deformation during cyclical contractions or muscle overload. These components of the exercise response lead to different forms of capillary growth, and subsequent expansion of the microcirculation may not have the same functional outcome. In vitro and in vivo studies have shown a complex interplay between different cytokines, receptors and mural cells in directing the necessary tissue re-organisation. The mechanisms involved in arteriogenesis are less well-understood than those of angiogenesis, but application of these data to understanding vascular remodelling in response to exercise may help resolve a range of cardiovascular dysfunction.

Similar content being viewed by others
Abbreviations
- Ang:
-
angiopoietin
- C:F:
-
numerical capillary to fibre ratio
- CD:
-
capillary density
- CHF:
-
chronic (congestive) heart failure
- EC:
-
endothelial cells
- ECM:
-
extracellular matrix
- eNOS:
-
endothelial nitric oxide synthase
- EPC:
-
endothelial progenitor cells
- FGF:
-
fibroblast growth factor
- HIF:
-
hypoxia-inducible factor
- MO2max :
-
maximal oxygen consumption
- MMP:
-
matrix metalloproteinases
- NOS:
-
nitric oxide synthase
- PDGF:
-
platelet-derived growth factor
- PGI2 :
-
prostacyclin
- PlGF:
-
placental growth factor
- RTK:
-
receptor tyrosine kinase
- TGF:
-
transforming growth factor
- TIMPs:
-
tissue inhibitors of matrix metalloproteinases
- VEGF:
-
vascular endothelial growth factor
- VEGFR:
-
VEGF receptor
- VSM:
-
vascular smooth muscle
References
Adair TH, Gay WJ, Montani JP (1990) Growth regulation of the vascular system: evidence for a metabolic hypothesis. Am J Physiol 259:R393–R404
Adair TH (2005) Growth regulation of the vascular system: an emerging role for adenosine. Am J Physiol Regul Integr Comp Physiol 289:R283–R296
Ahmed SK, Egginton S, Jakeman PM et al (1997) Is human skeletal muscle capillary supply modelled according to fibre size or fibre type? Exp Physiol 82:231–234
Amaral SL, Papanek PE, Greene AS (2001) Angiotensin II and VEGF are involved in angiogenesis induced by short-term exercise training. Am J Physiol Heart Circ Physiol 281:H1163–H1169
Andersen P, Henriksson J (1977) Capillary supply of the quadriceps femoris muscle of man: adaptive response to exercise. J Physiol 270:677–690
Asahara T, Masuda H, Takahashi T et al (1999) Bone marrow origin of endothelial progenitor cells responsible for postnatal vasculogenesis in physiological and pathological neovascularization. Circ Res 85:221–228
Babaei S, Teichert-Kuliszewska K, Monge JC et al (1998) Role of nitric oxide in the angiogenic response in vitro to basic fibroblast growth factor. Circ Res 82:1007–1015
Badr I, Brown MD, Egginton S et al (2003) Differences in local environment determine the site of physiological angiogenesis in rat skeletal muscle. Exp Physiol 88:565–568
Bailey AP, Shparago M, Gu JW (2006) Exercise increases soluble vascular endothelial growth factor receptor-1 (sFlt-1) in circulation of healthy volunteers. Med Sci Monit 12:CR45–CR50
Bassett DR Jr (1994) Skeletal muscle characteristics: relationships to cardiovascular risk factors. Med Sci Sports Exerc 26:957–966
Bein K, Simons M (2000) Thrombospondin type 1 repeats interact with matrix metalloproteinase 2. Regulation of metalloproteinase activity. J Biol Chem 275:32167–32173
Benndorf R, Boger RH, Ergun S et al (2003) Angiotensin II type 2 receptor inhibits vascular endothelial growth factor-induced migration and in vitro tube formation of human endothelial cells. Circ Res 93:438–447
Benoit H, Jordan M, Wagner H et al (1999) Effect of NO, vasodilator prostaglandins, and adenosine on skeletal muscle angiogenic growth factor gene expression. J Appl Physiol 86:1513–1518
Blair SN, Goodyear NN, Gibbons LW et al (1984) Physical fitness and incidence of hypertension in healthy normotensive men and women. JAMA 252:487–490
Bongrazio M, Da Silva-Azevedo L, Bergmann EC et al (2006) Shear stress modulates the expression of thrombospondin-1 and CD36 in endothelial cells in vitro and during shear stress-induced angiogenesis in vivo. Int J Immunopathol Pharmacol 19:35–48
Booth FW, Thomason DB (1991) Molecular and cellular adaptation of muscle in response to exercise: perspectives of various models. Physiol Rev 71:541–585
Boushel R (2003) Metabolic control of muscle blood flow during exercise in humans. Can J Appl Physiol 28:754–773
Breen EC, Johnson EC, Wagner H et al (1996) Angiogenic growth factor mRNA responses in muscle to a single bout of exercise. J Appl Physiol 81:355–361
Brooks PC, Clark RA, Cheresh DA (1994) Requirement of vascular integrin alpha v beta 3 for angiogenesis. Science 264:569–571
Brown MD, Egginton S, Hudlická O et al (1996) Appearance of the capillary endothelial glycocalyx in chronically stimulated rat skeletal muscles in relation to angiogenesis. Exp Physiol 81:1043–1046
Brown MD, Walter H, Hansen-Smith FM et al (1998) Lack of involvement of basic fibroblast growth factor (FGF-2) in capillary growth in skeletal muscles exposed to long-term contractile activity. Angiogenesis 2:81–91
Carrow RE, Brown RE, Van Huss WD (1967) Fiber sizes and capillary to fiber ratios in skeletal muscle of exercised rats. Anat Rec 159:33–39
Chandler WL, Levy WC, Stratton JR (1995) The circulatory regulation of TPA and UPA secretion, clearance, and inhibition during exercise and during the infusion of isoproterenol and phenylephrine. Circulation 92:2984–2994
Chang H, Wang BW, Kuan P et al (2003) Cyclical mechanical stretch enhances angiopoietin-2 and Tie2 receptor expression in cultured human umbilical vein endothelial cells. Clin Sci (Lond) 104:421–428
Cherwek DH, Hopkins MB, Thompson MJ et al (2000) Fiber type-specific differential expression of angiogenic factors in response to chronic hindlimb ischemia. Am J Physiol Heart Circ Physiol 279:H932–H938
Chong AY, Caine GJ, Freestone B et al (2004) Plasma angiopoietin-1, angiopoietin-2, and angiopoietin receptor tie-2 levels in congestive heart failure. J Am Coll Cardiol 43:423–428
Clark MG, Rattigan S, Barrett EJ et al (2007) Point:counterpoint: there is/is not capillary recruitment in active skeletal muscle during exercise. J Appl Physiol 104:889–891
Clifford PS, Hellsten Y (2004) Vasodilatory mechanisms in contracting skeletal muscle. J Appl Physiol 97:393–403
Colville-Nash PR, Willoughby DA (1997) Growth factors in angiogenesis: current interest and therapeutic potential. Mol Med Today 3:14–23
Costa C, Incio J, Soares R (2007) Angiogenesis and chronic inflammation: cause or consequence? Angiogenesis 10:149–166
Coyle EF, Martin WH 3rd, Sinacore DR et al (1984) Time course of loss of adaptations after stopping prolonged intense endurance training. J Appl Physiol 57:1857–1864
Czarkowska-Paczek B, Bartlomiejczyk I, Przybylski J (2006) The serum levels of growth factors: PDGF, TGF-beta and VEGF are increased after strenuous physical exercise. J Physiol Pharmacol 57:189–197
Deveci D, Marshall JM, Egginton S (2001) Relationship between capillary angiogenesis, fiber type, and fiber size in chronic systemic hypoxia. Am J Physiol Heart Circ Physiol 281:H241–H252
Deveci D, Egginton S (2002) Muscle ischaemia in rats may be relieved by overload-induced angiogenesis. Exp Physiol 87:479–488
Dickson MC, Martin JS, Cousins FM et al (1995) Defective haematopoiesis and vasculogenesis in transforming growth factor-beta 1 knock out mice. Development 121:1845–1854
Djonov V, Baum O, Burri PH (2003) Vascular remodeling by intussusceptive angiogenesis. Cell Tissue Res 314:107–117
Drygas WK (1988) Changes in blood platelet function, coagulation, and fibrinolytic activity in response to moderate, exhaustive, and prolonged exercise. Int J Sports Med 9:67–72
Duscha BD, Kraus WE, Keteyian SJ et al (1999) Capillary density of skeletal muscle: a contributing mechanism for exercise intolerance in class II–III chronic heart failure independent of other peripheral alterations. J Am Coll Cardiol 33:1956–1963
Egginton S (1990) Morphometric analysis of tissue capillary supply. Adv Comp Environ Physiol 6:73–141
Egginton S, Hudlická O, Brown MD et al (1998) Capillary growth in relation to blood flow and performance in overloaded rat skeletal muscle. J Appl Physiol 85:2025–2032
Egginton S, Hudlická O (2000) Selective long-term electrical stimulation of fast glycolytic fibres increases capillary supply but not oxidative enzyme activity in rat skeletal muscles. Exp Physiol 85:567–573
Egginton S, Zhou AL, Brown MD et al (2001) Unorthodox angiogenesis in skeletal muscle. Cardiovasc Res 49:634–646
Egginton S, Gerritsen M (2003) Lumen formation: in vivo versus in vitro observations. Microcirculation 10:45–61
Estelles A, Aznar J, Tormo G et al (1989) Influence of a rehabilitation sports programme on the fibrinolytic activity of patients after myocardial infarction. Thromb Res 55:203–212
Ferrara N (2001) Role of vascular endothelial growth factor in regulation of physiological angiogenesis. Am J Physiol Cell Physiol 280:C1358–C1366
Folkman J, Klagsbrun M (1987) Angiogenic factors. Science 235:442–447
Folkman J (1995) Angiogenesis in cancer, vascular, rheumatoid and other disease. Nat Med 1:27–31
Folkman J (2006) Angiogenesis. Annu Rev Med 57:1–18
Fuchs S, Baffour R, Zhou YF et al (2001) Transendocardial delivery of autologous bone marrow enhances collateral perfusion and regional function in pigs with chronic experimental myocardial ischemia. J Am Coll Cardiol 37:1726–1732
Fulgenzi G, Graciotti L, Collis MG et al (1998) The effect of alpha 1 adrenoceptor antagonist prazosin on capillary supply, blood flow and performance in a rat model of chronic muscle ischaemia. Eur J Vasc Endovasc Surg 16:71–77
Gavin TP, Wagner PD (2001) Effect of short-term exercise training on angiogenic growth factor gene responses in rats. J Appl Physiol 90:1219–1226
Gavin TP, Robinson CB, Yeager RC et al (2004) Angiogenic growth factor response to acute systemic exercise in human skeletal muscle. J Appl Physiol 96:19–24
Gavin TP, Drew JL, Kubik CJ et al (2007) Acute resistance exercise increases skeletal muscle angiogenic growth factor expression. Acta Physiol (Oxf) 191:139–146
Gigante B, Tarsitano M, Cimini V et al (2004) Placenta growth factor is not required for exercise-induced angiogenesis. Angiogenesis 7:277–284
Gu JW, Shparago M, Tan W et al (2006) Tissue endostatin correlates inversely with capillary network in rat heart and skeletal muscles. Angiogenesis 9:93–99
Gustafsson T, Puntschart A, Kaijser L et al (1999) Exercise-induced expression of angiogenesis-related transcription and growth factors in human skeletal muscle. Am J Physiol 276:H679–H685
Gustafsson T, Bodin K, Sylven C et al (2001) Increased expression of VEGF following exercise training in patients with heart failure. Eur J Clin Invest 31:362–366
Gustafsson T, Kraus WE (2001) Exercise-induced angiogenesis-related growth and transcription factors in skeletal muscle, and their modification in muscle pathology. Front Biosci 6:D75–D89
Gustafsson T, Knutsson A, Puntschart A et al (2002) Increased expression of vascular endothelial growth factor in human skeletal muscle in response to short-term one-legged exercise training. Pflugers Arch 444:752–759
Haas TL, Milkiewicz M, Davis SJ et al (2000) Matrix metalloproteinase activity is required for activity-induced angiogenesis in rat skeletal muscle. Am J Physiol Heart Circ Physiol 279:H1540–H1547
Hanahan D (1997) Signaling vascular morphogenesis and maintenance. Science 277:48–50
Hang J, Kong L, Gu JW et al (1995) VEGF gene expression is upregulated in electrically stimulated rat skeletal muscle. Am J Physiol 269:H1827–H1831
Hansen-Smith F, Egginton S, Zhou AL et al (2001) Growth of arterioles precedes that of capillaries in stretch-induced angiogenesis in skeletal muscle. Microvasc Res 62:1–14
Hansen-Smith FM, Hudlická O, Egginton S (1996) In vivo angiogenesis in adult rat skeletal muscle: early changes in capillary network architecture and ultrastructure. Cell Tissue Res 286:123–136
Hawker MJ, Egginton S (1997) The effect of stimulation frequency on skeletal muscle blood flow in the anaesthetized rat. J Physiol (Lond) 499P:P22–P23
Hellsten Y, Rufener N, Nielsen JJ et al (2007) Passive leg movement enhances interstitial VEGF protein, endothelial cell proliferation and eNOS mRNA content in human skeletal muscle. Am J Physiol Regul Integr Comp Physiol 294:R975–R982
Hepple RT (2000) Skeletal muscle: microcirculatory adaptation to metabolic demand. Med Sci Sports Exerc 32:117–123
Hepple RT, Hogan MC, Stary C et al (2000) Structural basis of muscle O(2) diffusing capacity: evidence from muscle function in situ. J Appl Physiol 88:560–566
Hiatt WR, Regensteiner JG, Hargarten ME et al (1990) Benefit of exercise conditioning for patients with peripheral arterial disease. Circulation 81:602–609
Hoffner L, Nielsen JJ, Langberg H et al (2003) Exercise but not prostanoids enhance levels of vascular endothelial growth factor and other proliferative agents in human skeletal muscle interstitium. J Physiol 550:217–225
Hoppeler H, Vogt M (2001) Muscle tissue adaptations to hypoxia. J Exp Biol 204:3133–3139
Hudlická O, Tyler KR, Wright AJ et al (1984) Growth of capillaries in skeletal muscle. Progr Appl Microcirc 5:5
Hudlická O, Brown M, Egginton S (1992) Angiogenesis in skeletal and cardiac muscle. Physiol Rev 72:369–417
Hudlická O, Brown MD, Egginton S et al (1994) Effect of long-term electrical stimulation on vascular supply and fatigue in chronically ischemic muscles. J Appl Physiol 77:1317–1324
Hudlická O, Brown MD, Silgram H (2000) Inhibition of capillary growth in chronically stimulated rat muscles by N(G)-nitro-l-arginine, nitric oxide synthase inhibitor. Microvasc Res 59:45–51
Huonker M, Schmid A, Schmidt-Trucksass A et al (2003) Size and blood flow of central and peripheral arteries in highly trained able-bodied and disabled athletes. J Appl Physiol 95:685–691
Ingjer F (1979) Effects of endurance training on muscle fibre ATP-ase activity, capillary supply and mitochondrial content in man. J Physiol 294:419–432
Isner JM, Asahara T (1999) Angiogenesis and vasculogenesis as therapeutic strategies for postnatal neovascularization. J Clin Invest 103:1231–1236
Jain RK, Schlenger K, Hockel M et al (1997) Quantitative angiogenesis assays: progress and problems. Nat Med 3:1203–1208
Jensen L, Bangsbo J, Hellsten Y (2004) Effect of high intensity training on capillarization and presence of angiogenic factors in human skeletal muscle. J Physiol 557:571–582
Joyner M (2007) Exercise hyperaemia: are there any answers yet? J Physiol 583:817
Kivela R, Silvennoinen M, Touvra AM et al (2006) Effects of experimental type 1 diabetes and exercise training on angiogenic gene expression and capillarization in skeletal muscle. FASEB J 20:1570–1572
Kivela R, Silvennoinen M, Lehti M et al (2007) Effects of acute exercise, exercise training, and diabetes on the expression of lymphangiogenic growth factors and lymphatic vessels in skeletal muscle. Am J Physiol Heart Circ Physiol 293:H2573–H2579
Koskinen SO, Wang W, Ahtikoski AM et al (2001) Acute exercise induced changes in rat skeletal muscle mRNAs and proteins regulating type IV collagen content. Am J Physiol Regul Integr Comp Physiol 280:R1292–R1300
Kraus RM, Stallings HW 3rd, Yeager RC et al (2004) Circulating plasma VEGF response to exercise in sedentary and endurance-trained men. J Appl Physiol 96:1445–1450
Krogh A (1919) The supply of oxygen to the tissues and the regulation of the capillary circulation. J Physiol 52:457–474
Krogh A (1919) The number and distribution of capillaries in muscles with calculations of the oxygen pressure head necessary for supplying the tissue. J Physiol 52:409–415
Laufs U, Werner N, Link A et al (2004) Physical training increases endothelial progenitor cells, inhibits neointima formation, and enhances angiogenesis. Circulation 109:220–226
Laughlin MH, Korthuis RJ, Duncker DJ et al (1996) Control of blood flow to cardiac and skeletal muscle during exercise. In: Rowell LB, Shepherd JT (eds) Handbook of physiology: sect. 12 exercise: regulation and integration of multiple systems. American Physiological Society, Bethesda, pp 705–769
Laughlin MH, Welshons WV, Sturek M et al (2003) Gender, exercise training, and eNOS expression in porcine skeletal muscle arteries. J Appl Physiol 95:250–264
Lawler J (2000) The functions of thrombospondin-1 and-2. Curr Opin Cell Biol 12:634–640
Lelkes PI (ed) (1999) Endothelium and mechanical forces. Harwood Academic, London
Leveen P, Pekny M, Gebre-Medhin S et al (1994) Mice deficient for PDGF B show renal, cardiovascular, and hematological abnormalities. Genes Dev 8:1875–1887
Lloyd PG, Yang HT, Terjung RL (2001) Arteriogenesis and angiogenesis in rat ischemic hindlimb: role of nitric oxide. Am J Physiol Heart Circ Physiol 281:H2528–H2538
Lloyd PG, Prior BM, Yang HT et al (2003) Angiogenic growth factor expression in rat skeletal muscle in response to exercise training. Am J Physiol Heart Circ Physiol 284:H1668–H1678
Mai JV, Edgerton VR, Barnard RJ (1970) Capillarity of red, white and intermediate muscle fibers in trained and untrained guinea pigs. Experientia 26:1222–1223
Maisonpierre PC, Suri C, Jones PF et al (1997) Angiopoietin-2, a natural antagonist for Tie2 that disrupts in vivo angiogenesis. Science 277:55–60
Mandriota SJ, Pepper MS (1997) Vascular endothelial growth factor-induced in vitro angiogenesis and plasminogen activator expression are dependent on endogenous basic fibroblast growth factor. J Cell Sci 110(Pt 18):2293–2302
Marin P, Andersson B, Krotkiewski M et al (1994) Muscle fiber composition and capillary density in women and men with NIDDM. Diabetes Care 17:382–386
Matsunaga T, Weihrauch DW, Moniz MC et al (2002) Angiostatin inhibits coronary angiogenesis during impaired production of nitric oxide. Circulation 105:2185–2191
Milkiewicz M, Brown MD, Egginton S et al (2001) Association between shear stress, angiogenesis, and VEGF in skeletal muscles in vivo. Microcirculation 8:229–241
Milkiewicz M, Hudlická O, Verhaeg J et al (2003) Differential expression of Flk-1 and Flt-1 in rat skeletal muscle in response to chronic ischaemia: favourable effect of muscle activity. Clin Sci (Lond) 105:473–482
Milkiewicz M, Pugh CW, Egginton S (2004) Inhibition of endogenous HIF inactivation induces angiogenesis in ischaemic skeletal muscles of mice. J Physiol 560:21–26
Milkiewicz M, Hudlická O, Shiner R et al (2006) Vascular endothelial growth factor mRNA and protein do not change in parallel during non-inflammatory skeletal muscle ischaemia in rat. J Physiol 577:671–678
Milkiewicz M, Ispanovic E, Doyle JL et al (2006) Regulators of angiogenesis and strategies for their therapeutic manipulation. Int J Biochem Cell Biol 38:333–357
Milkiewicz M, Kelland C, Colgan S et al (2006) Nitric oxide and p38 MAP kinase mediate shear stress-dependent inhibition of MMP-2 production in microvascular endothelial cells. J Cell Physiol 208:229–237
Morbidelli L, Chang CH, Douglas JG et al (1996) Nitric oxide mediates mitogenic effect of VEGF on coronary venular endothelium. Am J Physiol 270:H411–H415
Morrow NG, Kraus WE, Moore JW et al (1990) Increased expression of fibroblast growth factors in a rabbit skeletal muscle model of exercise conditioning. J Clin Invest 85:1816–1820
Mujika I, Padilla S (2001) Muscular characteristics of detraining in humans. Med Sci Sports Exerc 33:1297–1303
Neufeld G, Cohen T, Gengrinovitch S et al (1999) Vascular endothelial growth factor (VEGF) and its receptors. FASEB J 13:9–22
O’Reilly MS, Holmgren L, Shing Y et al (1994) Angiostatin: a novel angiogenesis inhibitor that mediates the suppression of metastases by a Lewis lung carcinoma. Cell 79:315–328
O’Reilly MS, Boehm T, Shing Y et al (1997) Endostatin: an endogenous inhibitor of angiogenesis and tumor growth. Cell 88:277–285
Olfert IM, Breen EC, Mathieu-Costello O et al (2001) Skeletal muscle capillarity and angiogenic mRNA levels after exercise training in normoxia and chronic hypoxia. J Appl Physiol 91:1176–1184
Olfert IM, Breen EC, Gavin TP et al (2006) Temporal thrombospondin-1 mRNA response in skeletal muscle exposed to acute and chronic exercise. Growth Factors 24:253–259
Piepoli MF, Scott AC, Capucci A et al (2001) Skeletal muscle training in chronic heart failure. Acta Physiol Scand 171:295–303
Pipp F, Heil M, Issbrucker K et al (2003) VEGFR-1-selective VEGF homologue PlGF is arteriogenic: evidence for a monocyte-mediated mechanism. Circ Res 92:378–385
Ponjee GA, Janssen GM, van Wersch JW (1993) Prolonged endurance exercise and blood coagulation: a 9 month prospective study. Blood Coagul Fibrinolysis 4:21–25
Pugh CW, Ratcliffe PJ (2003) Regulation of angiogenesis by hypoxia: role of the HIF system. Nat Med 9:677–684
Richardson RS, Grassi B, Gavin TP et al (1999) Evidence of O2 supply-dependent VO2 max in the exercise-trained human quadriceps. J Appl Physiol 86:1048–1053
Richardson RS, Wagner H, Mudaliar SR et al (1999) Human VEGF gene expression in skeletal muscle: effect of acute normoxic and hypoxic exercise. Am J Physiol 277:H2247–H2252
Richardson RS, Wagner H, Mudaliar SR et al (2000) Exercise adaptation attenuates VEGF gene expression in human skeletal muscle. Am J Physiol Heart Circ Physiol 279:H772–H778
Risau W (1997) Mechanisms of angiogenesis. Nature 386:671–674
Rivard A, Fabre JE, Silver M et al (1999) Age-dependent impairment of angiogenesis. Circulation 99:111–120
Rivilis I, Milkiewicz M, Boyd P et al (2002) Differential involvement of MMP-2 and VEGF during muscle stretch- versus shear stress-induced angiogenesis. Am J Physiol Heart Circ Physiol 283:H1430–H1438
Roberts KC, Nixon C, Unthank JL et al (1997) Femoral artery ligation stimulates capillary growth and limits training-induced increases in oxidative capacity in rats. Microcirculation 4:253–260
Robinson DM, Ogilvie RW, Tullson PC et al (1994) Increased peak oxygen consumption of trained muscle requires increased electron flux capacity. J Appl Physiol 77:1941–1952
Roca J, Gavin TP, Jordan M et al (1998) Angiogenic growth factor mRNA responses to passive and contraction-induced hyperperfusion in skeletal muscle. J Appl Physiol 85:1142–1149
Rowell LB (1986) Human circulation: regulation during physical stress. Oxford University Press, Oxford, p 428
Rullman E, Rundqvist H, Wagsater D et al (2007) A single bout of exercise activates matrix metalloproteinase in human skeletal muscle. J Appl Physiol 102:2346–2351
Saltin B, Gollnick PD (1983) Skeletal muscle adaptability: significance for metabolism and performance. In: Peachey LD, Adrian RH, Geiger SR (eds) Handbook of physiology: sect. 10 skeletal muscle. American Physiological Society, Bethesda, pp 555–599
Schantz P, Henriksson J, Jansson E (1983) Adaptation of human skeletal muscle to endurance training of long duration. Clin Physiol 3:141–151
Schmidt-Nielsen K, Pennycuik P (1961) Capillary density in mammals in relation to body size and oxygen consumption. Am J Physiol 200:746–750
Segal SS (2005) Regulation of blood flow in the microcirculation. Microcirculation 12:33–45
Semenza GL (2007) Vasculogenesis, angiogenesis, and arteriogenesis: mechanisms of blood vessel formation and remodeling. J Cell Biochem 102:840–847
Shweiki D, Itin A, Soffer D et al (1992) Vascular endothelial growth factor induced by hypoxia may mediate hypoxia-initiated angiogenesis. Nature 359:843–845
Simoneau JA, Colberg SR, Thaete FL et al (1995) Skeletal muscle glycolytic and oxidative enzyme capacities are determinants of insulin sensitivity and muscle composition in obese women. FASEB J 9:273–278
Skorjanc D, Jaschinski F, Heine G et al (1998) Sequential increases in capillarization and mitochondrial enzymes in low-frequency-stimulated rabbit muscle. Am J Physiol 274:C810–C818
Spier SA, Delp MD, Stallone JN et al (2007) Exercise training enhances flow-induced vasodilation in skeletal muscle resistance arteries of aged rats: role of PGI2 and nitric oxide. Am J Physiol Heart Circ Physiol 292:H3119–H3127
Staton CA, Lewis C, Bicknell R (eds) (2006) Angiogenesis assays. A critical appraisal of current techniques. Wiley, Chichester, p 390
Stavri GT, Zachary IC, Baskerville PA et al (1995) Basic fibroblast growth factor upregulates the expression of vascular endothelial growth factor in vascular smooth muscle cells. Synergistic interaction with hypoxia. Circulation 92:11–14
Stein I, Neeman M, Shweiki D et al (1995) Stabilization of vascular endothelial growth factor mRNA by hypoxia and hypoglycemia and coregulation with other ischemia-induced genes. Mol Cell Biol 15:5363–5368
Stetler-Stevenson WG, Seo DW (2005) TIMP-2: an endogenous inhibitor of angiogenesis. Trends Mol Med 11:97–103
Suhr F, Brixius K, de Marees M et al (2007) Effects of short-term vibration and hypoxia during high-intensity cycling exercise on circulating levels of angiogenic regulators in humans. J Appl Physiol 103:474–483
Sun HW, Li CJ, Chen HQ et al (2007) Involvement of integrins, MAPK, and NF-kappaB in regulation of the shear stress-induced MMP-9 expression in endothelial cells. Biochem Biophys Res Commun 353:152–158
Sundberg CJ (1994) Exercise and training during graded leg ischaemia in healthy man with special reference to effects on skeletal muscle. Acta Physiol Scand Suppl 615:1–50
Tesch PA, Thorsson A, Kaiser P (1984) Muscle capillary supply and fiber type characteristics in weight and power lifters. J Appl Physiol 56:35–38
Tesch PA (1988) Skeletal muscle adaptations consequent to long-term heavy resistance exercise. Med Sci Sports Exerc 20:S132–S134
Thurston G (2003) Role of Angiopoietins and Tie receptor tyrosine kinases in angiogenesis and lymphangiogenesis. Cell Tissue Res 314:61–68
Tomanek RJ, Schatteman GC (2000) Angiogenesis: new insights and therapeutic potential. Anat Rec 261:126–135
Tornling G, Adolfsson J, Unge G et al (1980) Capillary neoformation in skeletal muscle of dipyridamole-treated rats. Arzneimittelforschung 30:791–792
Tronc F, Mallat Z, Lehoux S et al (2000) Role of matrix metalloproteinases in blood flow-induced arterial enlargement: interaction with NO. Arterioscler Thromb Vasc Biol 20:E120–E126
Tsurumi Y, Murohara T, Krasinski K et al (1997) Reciprocal relation between VEGF and NO in the regulation of endothelial integrity. Nat Med 3:879–886
Tuttle JL, Nachreiner RD, Bhuller AS et al (2001) Shear level influences resistance artery remodeling: wall dimensions, cell density, and eNOS expression. Am J Physiol Heart Circ Physiol 281:H1380–H1389
van Groningen JP, Wenink AC, Testers LH (1991) Myocardial capillaries: increase in number by splitting of existing vessels. Anat Embryol (Berl) 184:65–70
van Royen N, Piek JJ, Buschmann I et al (2001) Stimulation of arteriogenesis; a new concept for the treatment of arterial occlusive disease. Cardiovasc Res 49:543–553
Wang JS, Jen CJ, Kung HC et al (1994) Different effects of strenuous exercise and moderate exercise on platelet function in men. Circulation 90:2877–2885
Wang Y, Chang J, Li YC et al (2004) Shear stress and VEGF activate IKK via the Flk-1/Cbl/Akt signaling pathway. Am J Physiol Heart Circ Physiol 286:H685–H692
Williams JL, Cartland D, Hussain A et al (2006) A differential role for nitric oxide in two forms of physiological angiogenesis in mouse. J Physiol 570:445–454
Williams JL, Cartland D, Rudge JS et al (2006) VEGF trap abolishes shear stress- and overload-dependent angiogenesis in skeletal muscle. Microcirculation 13:499–509
Williams JL, Weichert A, Zakrzewicz A et al (2006) Differential gene and protein expression in abluminal sprouting and intraluminal splitting forms of angiogenesis. Clin Sci (Lond) 110:587–595
Wu LW, Mayo LD, Dunbar JD et al (2000) Utilization of distinct signaling pathways by receptors for vascular endothelial cell growth factor and other mitogens in the induction of endothelial cell proliferation. J Biol Chem 275:5096–5103
Xiang L, Naik J, Hester RL (2005) Exercise-induced increase in skeletal muscle vasodilatory responses in obese Zucker rats. Am J Physiol Regul Integr Comp Physiol 288:R987–R991
Yamaguchi S, Yamaguchi M, Yatsuyanagi E et al (2002) Cyclic strain stimulates early growth response gene product 1-mediated expression of membrane type 1 matrix metalloproteinase in endothelium. Lab Invest 82:949–956
Yang HT, Ogilvie RW, Terjung RL (1994) Peripheral adaptations in trained aged rats with femoral artery stenosis. Circ Res 74:235–243
Zheng W, Seftor EA, Meininger CJ et al (2001) Mechanisms of coronary angiogenesis in response to stretch: role of VEGF and TGF-beta. Am J Physiol Heart Circ Physiol 280:H909–H917
Zhou AL, Egginton S, Brown MD et al (1998) Capillary growth in overloaded, hypertrophic adult rat skeletal muscle: an ultrastructural study. Anat Rec 252:49–63
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Egginton, S. Invited review: activity-induced angiogenesis. Pflugers Arch - Eur J Physiol 457, 963–977 (2009). https://doi.org/10.1007/s00424-008-0563-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-008-0563-9