Abstract
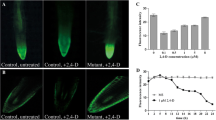
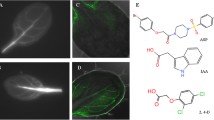
The diageotropica (dgt) mutant of tomato (Lycopersicon esculentum Mill.) is known to lack a number of typical auxin responses. Here we show that rapid auxin-induced growth of seedling hypocotyls is completely abolished by the mutation over the full range of auxin concentrations tested, and also in early phases of the time course. Protoplasts isolated from wild-type hypocotyls respond to auxin by a rapid increase in cell volume, which we measured by image analysis at a high temporal resolution. A similar swelling could be triggered by antibodies directed against a part of the putative auxin-binding domain (box-a) of the auxin-binding protein 1 (ABP1). Induction of swelling both by auxin and by the antibody was not observed in the protoplasts isolated from the dgt mutant. However, dgt protoplasts are able to respond to the stimulator of the H+-ATPase, fusicoccin, with normal swelling. We propose that dgt is a signal-transduction mutation interfering with an auxin-signalling pathway that uses ABP1 as a receptor.




Similar content being viewed by others
Abbreviations
- ABP:
-
auxin-binding protein
- CCD:
-
charge-coupled device
- 2,4-D:
-
2,4-dichlorophenoxyacetic acid
- dgt :
-
diageotropica
- FC:
-
fusicoccin
References
Barbier-Brygoo H, Ephritikhine G, Klämbt D, Maurel C, Palme K, Schell J, Guern J (1991) Perception of the auxin signal at the plasma membrane of tobacco mesophyll protoplasts. Plant J 1:83–93
Chen JG, Ullah H, Young JC, Sussman MR, Jones AM (2001) ABP1 is required for organized cell elongation and division in Arabidopsis embryogenesis. Genes Dev 15:902–911
Christian M, Lüthen H (2000) New methods to analyse auxin-induced growth I: Classical auxinology goes Arabidopsis. Plant Growth Regul 32:107–114
Claussen M, Lüthen H, Böttger M (1996) Inside or outside? The localisation of the receptor relevant to auxin induced growth. Physiol Plant 98:861–867
Coenen C, Bierfreund N, Lüthen H, Neuhaus G (2002) Developmental regulation of proton ATPase dependent auxin responses in the diageotropica mutant of tomato (Lycopersicon esculentum). Physiol Plant 114:461–471
Daniel SG, Rayle DL, Cleland RE (1989) Auxin physiology of the tomato mutant diageotropica. Plant Physiol 91:804–807
Davies PJ (1974) The uptake and elution of indoleacetic acid by pea stem sections in relation to auxin induced growth. In: Plant growth substances 1973. Hirokawa, Tokyo, pp 767–778
Fischer C, Lüthen H, Böttger M, Hertel R (1992) Initial transient growth inhibition in maize coleoptiles following auxin application. J Plant Physiol 141:88–92
Fujino DW, Nissen SJ, Jones AD, Burger DW, Bradford KJ (1988) Quantification of indole-3-acetic acid in dark-grown seedlings of the diageotropica and epinastic mutants of tomato (Lycopersicon esculentum Mill.). Plant Physiol 88:780–784
Gehring CA, McConchie RM, Venis MA, Parrish RW (1998) Auxin binding protein antibodies and peptides influence stomatal opening and alter cytoplasmic pH. Planta 205:581–586
Hertel R (1995) Auxin binding protein 1 is a red herring. J Exp Bot 46:461–462
Jones AM (1994) Auxin binding proteins. Annu Rev Plant Physiol Plant Mol Biol 45:393–420
Jones AM, Im KH, Savka MA, Wu MJ, DeWitt G, Shillito R, Binns AN (1998) Auxin dependent cell expansion mediated by overexpressing auxin-binding protein 1. Science 282:1114–1117
Kelly MO, Bradford KJ (1986) Insensitivity of the diageotropica mutant to auxin. Plant Physiol 82:713–717
Krautwig B (1993) Etablierung von in vitro-Kultursystemen aus Mikrosporen von Mais (Zea mays L.). Thesis, University of Hamburg, Germany
Madlung A, Behringer FJ, Lomax TL (1999) Ethylene plays multiple nonprimary roles in modulating the gravitropic response in tomato. Plant Physiol 120:897–906
Marrè E (1979) Fusicoccin: a tool in plant physiology. Annu Rev Plant Physiol 30:273–288
Mito N, Bennett AB (1995) The diageotropica mutation and synthetic auxins differentially affect the expression of auxin-regulated genes in tomato. Plant Physiol 109:293–297
Muday GK, Lomax TL, Rayle DL (1995) Characterization of the growth and auxin physiology of roots of the tomato mutant, diageotropica. Planta 195:548–553
Nebenführ A, White TJ, Lomax TL (2000) The diageotropica mutation alters auxin induction of a subset of the Aux/IAA gene family in tomato. Plant Mol Biol 44:73–84
Palmgren MG (1991) Regulation of plant plasma membrane H+-ATPase. Plant Physiol 83:314–323
Park WJ (1998) Effect of epibrassinolide on hypocotyl growth of the tomato mutant diageotropica. Planta 207:120–124
Parsons A, Firn RD, Digby J (1988) The role of the coleoptile apex in controlling organ elongation. I. The effects of decapitation and apical incisions. J Exp Bot 39:1331–1341
Rice MS, Lomax TL (2000) The auxin-resistant diageotropica mutant of tomato responds to gravity via an auxin-mediated pathway. Planta 210:906–913
Rück A, Palme K, Venis MA, Napier RM, Felle HH (1993) Patch clamp analysis establishes a role for an auxin binding protein in the auxin stimulation of plasma membrane current in Zea mays protoplasts. Plant J 4:41–46
Steffens B, Lüthen H (2000) New methods to analyse auxin-induced growth II: The swelling reaction of protoplasts — a model system for the analysis of auxin signal transduction? Plant Growth Regul 32:115–122
Steffens B, Feckler C, Palme K, Christian M, Böttger M, Lüthen H (2001) The auxin signal for protoplast swelling is perceived by extracellular ABP1. Plant J 27:591–599
Tietze-Haß E, Dörffling K (1977) Initial phases of indolylacetic acid induced growth in coleoptile segments of Avena sativa L. Planta 135:192–195
Venis MA (1995) Auxin binding protein 1 is a red herring ? — Oh no it isn't. J Exp Bot 46:463–465
Venis MA, Napier RM, Barbier-Brygoo H, Maurel C, Perrot-Rechenmann C, Guern J (1992) Antibodies to a peptide from maize auxin-binding protein have auxin agonist activity. Proc Natl Acad Sci USA 89:7208–7212
Vesper MJ, Kuss CL (1990) Physiological evidence that the primary site of auxin action in maize coleoptiles is an intracellular site. Planta 182:486–491
Weiler EW, Jourdan PS, Conrad W (1981) Levels of indole-3-acetic acid in intact and decapitated coleoptiles as determined by a specific and highly sensitive solid phase immunoassay. Planta 153:561–571
Zobel RW (1974) Control of morphogenesis in the ethylene-requiring tomato mutant, diageotropica. Can J Bot 52:735–743
Acknowledgements
We express our thanks to Dr. Catharina Coenen for giving a generous supply of dgt and wild-type seeds. Dr. Christian Feckler and Prof. Klaus Palme (Universität Freiburg) supplied the anti-intern antibodies used in this study. We also thank the Deutsche Forschungsgemeinschaft for funding within the program "molecular analysis of phytohormone action". This work is based in part on the PhD thesis of May Christian at the University of Hamburg.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Christian, M., Steffens, B., Schenck, D. et al. The diageotropica mutation of tomato disrupts a signalling chain using extracellular auxin binding protein 1 as a receptor. Planta 218, 309–314 (2003). https://doi.org/10.1007/s00425-003-1090-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-003-1090-8