Abstract
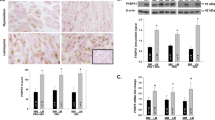
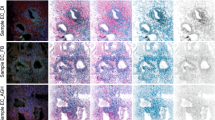
It is thought that the growth of uterine leiomyomas may be mediated by the interaction of estrogen receptor alpha (ERα) and growth factor pathways and that phosphorylation of ERα at serine 118 (ERα-phospho-Ser118) is important in this interaction. In this study, immunoblotting and immunohistochemistry were used to investigate the expression of ERα-phospho-Ser118, phosphorylated p44/42 mitogen-activated protein kinase (phospho-p44/42 MAPK), and proliferating cell nuclear antigen (PCNA) in human leiomyoma and myometrial tissues during the proliferative and secretory phases of the menstrual cycle. We found that tumors taken from the proliferative phase expressed significantly higher levels of ERα-phospho-Ser118, phospho-p44/42 MAPK, and PCNA compared to patient-matched myometria and had significantly higher ERα-phospho-Ser118 and PCNA expression compared to secretory phase tumors. Also, enhanced colocalization and association of phospho-p44/42 MAPK and ERα-phospho-Ser118 were observed in proliferative phase tumors by confocal microscopy and immunoprecipitation, respectively. These data suggest that ERα-phospho-Ser118 may be important in leiomyoma growth and is possibly phosphorylated by phospho-p44/42 MAPK.








Similar content being viewed by others
References
Barbarisi A, Petillo O, Di Lieto A, Melone MA, Margarucci S, Cannas M, Peluso G (2001) 17-beta estradiol elicits an autocrine leiomyoma cell proliferation: evidence for a stimulation of protein kinase-dependent pathway. J Cell Physiol 186(3):414–424
Beinfeld MT, Bosch JL, Isaacson KB, Gazelle GS (2004) Cost-effectiveness of uterine artery embolization and hysterectomy for uterine fibroids. Radiology 230(1):207–213
Bourlev V, Pavlovitch S, Stygar D, Volkov N, Lindblom B, Olovsson M (2003) Different proliferative and apoptotic activity in peripheral versus central parts of human uterine leiomyomas. Gynecol Obstet Invest 55(4):199–204
Bunone G, Briand PA, Miksicek RJ, Picard D (1996) Activation of the unliganded estrogen receptor by EGF involves the MAP kinase pathway and direct phosphorylation. Embo J 15(9):2174–2183
Buttram VC Jr (1986) Uterine leiomyomata—aetiology, symptomatology and management. Prog Clin Biol Res 225:275–296
Chegini N, Kornberg L (2003) Gonadotropin releasing hormone analogue therapy alters signal transduction pathways involving mitogen-activated protein and focal adhesion kinases in leiomyoma. J Soc Gynecol Investig 10(1):21–26
Chen D, Washbrook E, Sarwar N, Bates GJ, Pace PE, Thirunuvakkarasu V, Taylor J, Epstein RJ, Fuller-Pace FV, Egly JM, Coombes RC, Ali S (2002) Phosphorylation of human estrogen receptor alpha at serine 118 by two distinct signal transduction pathways revealed by phosphorylation-specific antisera. Oncogene 21(32):4921–4931
Chwalisz K, DeManno D, Garg R, Larsen L, Mattia-Goldberg C, Stickler T (2004) Therapeutic potential for the selective progesterone receptor modulator asoprisnil in the treatment of leiomyomata. Semin Reprod Med 22(2):113–119
Chwalisz K, Perez MC, Demanno D, Winkel C, Schubert G, Elger W (2005) Selective progesterone receptor modulator development and use in the treatment of leiomyomata and endometriosis. Endocr Rev 26(3):423–438
Cook JD, Walker CL (2004) Treatment strategies for uterine leiomyoma: the role of hormonal modulation. Semin Reprod Med 22(2):105–111
Denton RR, Koszewski NJ, Notides AC (1992) Estrogen receptor phosphorylation. Hormonal dependence and consequence on specific DNA binding. J Biol Chem 267(11):7263–7268
Detre S, Saclani Jotti G, Dowsett M (1995) A “quickscore” method for immunohistochemical semiquantitation: validation for oestrogen receptor in breast carcinomas. J Clin Pathol 48(9):876–878
Dixon D, Flake GP, Moore AB, He H, Haseman JK, Risinger JI, Lancaster JM, Berchuck A, Barrett JC, Robboy SJ (2002) Cell proliferation and apoptosis in human uterine leiomyomas and myometria. Virchows Arch 441(1):53–62
Emembolu JO (1987) Uterine fibromyomata: presentation and management in northern Nigeria. Int J Gynaecol Obstet 25(5):413–416
Farquhar CM, Steiner CA, Sozen I, Arici A (2002) Hysterectomy rates in the United States 1990–1997. Obstet Gynecol 99(2):229–234
Hodges LC, Houston KD, Hunter DS, Fuchs-Young R, Zhang Z, Wineker RC, Walker CL (2002) Transdominant suppression of estrogen receptor signaling by progesterone receptor ligands in uterine leiomyoma cells. Mol Cell Endocrinol 196(1–2):11–20
Jasonni VM, La Marca A, Santini D (2005) Progestin effects on epidermal growth factor receptor (EGFR) endometrial expression in normal and hyperplastic endometrium. Int J Gynaecol Obstet 89(3):297–298
Kato S, Endoh H, Masuhiro Y, Kitamoto T, Uchiyama S, Sasaki H, Masushige S, Gotoh Y, Nishida E, Kawashima H, Metzger D, Chambon P (1995) Activation of the estrogen receptor through phosphorylation by mitogen-activated protein kinase. Science 270(5241):1491–1494
Katzenellenbogen BS (2000) Mechanisms of action and cross-talk between estrogen receptor and progesterone receptor pathways. J Soc Gynecol Investig 7(1 Suppl):S33–S37
Kayisli UA, Berkkanoglu M, Kizilay G, Senturk L, Arici A (2007) Expression of proliferative and preapoptotic molecules in human myometrium and leiomyoma throughout the menstrual cycle. Reprod Sci 14(7):678–686
Kim MR, Park DW, Lee JH, Choi DS, Hwang KJ, Ryu HS, Min CK (2005) Progesterone-dependent release of transforming growth factor-beta1 from epithelial cells enhances the endometrial decidualization by turning on the Smad signalling in stromal cells. Mol Hum Reprod 11(11):801–808
Kovacs KA, Oszter A, Gocze PM, Kornyei JL, Szabo I (2001) Comparative analysis of cyclin D1 and oestrogen receptor (alpha and beta) levels in human leiomyoma and adjacent myometrium. Mol Hum Reprod 7(11):1085–1091
Kraus WL, Katzenellenbogen BS (1993) Regulation of progesterone receptor gene expression and growth in the rat uterus: modulation of estrogen actions by progesterone and sex steroid hormone antagonists. Endocrinology 132(6):2371–2379
Kraus WL, Weis KE, Katzenellenbogen BS (1995) Inhibitory cross-talk between steroid hormone receptors: differential targeting of estrogen receptor in the repression of its transcriptional activity by agonist- and antagonist-occupied progestin receptors. Mol Cell Biol 15(4):1847–1857
Lannigan DA (2003) Estrogen receptor phosphorylation. Steroids 68(1):1–9
Marsh EE, Bulun SE (2006) Steroid hormones and leiomyomas. Obstet Gynecol Clin North Am 33(1):59–67
Maruo T, Ohara N, Matsuo H, Xu Q, Chen W, Sitruk-Ware R, Johansson ED (2007) Effects of levonorgestrel-releasing IUS and progesterone receptor modulator PRM CDB-2914 on uterine leiomyomas. Contraception 75(6 Suppl):S99–103
Maruo T, Ohara N, Wang J, Matsuo H (2004) Sex steroidal regulation of uterine leiomyoma growth and apoptosis. Hum Reprod Update 10(3):207–220
Matsuzaki S, Canis M, Pouly JL, Botchorishvili R, Dechelotte PJ, Mage G (2007) Both GnRH agonist and continuous oral progestin treatments reduce the expression of the tyrosine kinase receptor B and mu-opioid receptor in deep infiltrating endometriosis. Hum Reprod 22(1):124–128
Parazzini F (2006) Risk factors for clinically diagnosed uterine fibroids in women around menopause. Maturitas 55(2):174–179
Rein M (1992) Biology of uterine myomas and myometrium in vitro. In: Barbieri RL (ed) Seminars in reproductive endocrinology. Thieme, New York, pp 310–319
Reis FM, Lhullier C, Edelweiss MI, Spritzer PM (2005) In vivo assessment of the regulation of transforming growth factor alpha, epidermal growth factor (EGF), and EGF receptor in the human endometrium by medroxyprogesterone acetate. J Assist Reprod Genet 22(1):19–24
Sato F, Mori M, Nishi M, Kudo R, Miyake H (2002) Familial aggregation of uterine myomas in Japanese women. J Epidemiol 12(3):249–253
Shimomura Y, Matsuo H, Samoto T, Maruo T (1998) Up-regulation by progesterone of proliferating cell nuclear antigen and epidermal growth factor expression in human uterine leiomyoma. J Clin Endocrinol Metab 83(6):2192–2198
Sozen I, Arici A (2002) Interactions of cytokines, growth factors, and the extracellular matrix in the cellular biology of uterine leiomyomata. Fertil Steril 78(1):1–12
Stewart EA, Friedman AJ, Peck K, Nowak RA (1994) Relative overexpression of collagen type I and collagen type III messenger ribonucleic acids by uterine leiomyomas during the proliferative phase of the menstrual cycle. J Clin Endocrinol Metab 79(3):900–906
Swartz CD, Afshari CA, Yu L, Hall KE, Dixon D (2005) Estrogen-induced changes in IGF-I, Myb family and MAP kinase pathway genes in human uterine leiomyoma and normal uterine smooth muscle cell lines. Mol Hum Reprod 11(6):441–450
Vereide AB, Kaino T, Sager G, Arnes M, Orbo A (2006) Effect of levonorgestrel IUD and oral medroxyprogesterone acetate on glandular and stromal progesterone receptors (PRA and PRB), and estrogen receptors (ER-alpha and ER-beta) in human endometrial hyperplasia. Gynecol Oncol 101(2):214–223
Vollenhoven B (1998) Introduction: the epidemiology of uterine leiomyomas. Baillieres Clin Obstet Gynaecol 12(2):169–176
Wang J, Ohara N, Wang Z, Chen W, Morikawa A, Sasaki H, DeManno DA, Chwalisz K, Maruo T (2006) A novel selective progesterone receptor modulator asoprisnil (J867) down-regulates the expression of EGF, IGF-I, TGFbeta3 and their receptors in cultured uterine leiomyoma cells. Hum Reprod 21(7):1869–1877
Washburn T, Hocutt A, Brautigan DL, Korach KS (1991) Uterine estrogen receptor in vivo: phosphorylation of nuclear specific forms on serine residues. Mol Endocrinol 5(2):235–242
Yamada T, Nakago S, Kurachi O, Wang J, Takekida S, Matsuo H, Maruo T (2004) Progesterone down-regulates insulin-like growth factor-I expression in cultured human uterine leiomyoma cells. Hum Reprod 19(4):815–821
Zaslawski R, Surowiak P, Dziegiel P, Pretnik L, Zabel M (2001) Analysis of the expression of estrogen and progesterone receptors, and of PCNA and Ki67 proliferation antigens, in uterine myomata cells in relation to the phase of the menstrual cycle. Med Sci Monit 7(5):908–913
Acknowledgments
The authors would like to thank Norris Flagler, Elizabeth Ney, Paul Cacioppo, and C. Jeffrey Tucker for their technical assistance with imaging. This research was supported, in part, by the Intramural Research Program of the NIH, National Institute of Environmental Health Sciences.
Conflict of interest statement
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hermon, T.L., Moore, A.B., Yu, L. et al. Estrogen receptor alpha (ERα) phospho-serine-118 is highly expressed in human uterine leiomyomas compared to matched myometrium. Virchows Arch 453, 557–569 (2008). https://doi.org/10.1007/s00428-008-0679-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-008-0679-5