Abstract
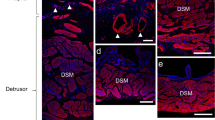
We have examined structures that may operate by using nitric oxide (NO)/soluble guanylyl cyclase (sGC) signalling in the lamina propria of the guinea pig bladder. Cells on the luminal surface of the urothelium and sub-urothelial interstitial cells (SU-ICs) responded to NO with a rise in cGMP. The distribution of these different cells varied between the base, lateral wall and dome. In the base, two regions were identified: areas with sparse surface urothelial cells and areas with a complete covering. A layer of cGMP-positive (cGMP+) cells (up to 10 cells deep) was found in the base. cGMP+/SU-ICs were also observed in the lateral wall. However, here, the cGMP+ cells were confined to a layer of only 1–2 cells immediately below the basal urothelial layer (basal cGMP+/SU-ICs). Below these cGMP+/SU-ICs lay cells that had a similar structure but that showed little cGMP accumulation (deep cGMP–/SU-ICs). Both basal and deep SU-ICs expressed the β1 subunit of sGC and the cGMP-dependent protein kinase I (cGKI), suggesting that the deep SU-ICs can sense NO and signal via cGMP. By using BAY 41-2272, a sensor of endogenous NO production, NO-dependent cGMP synthesis was observed primarily in the basal SU-ICs. A third population of cGKI+/cGMP− cells was seen to lie immediately below the basal urothelial layer. These cells (“necklace” cells) were less numerous than SU-ICs and extended linking processes suggesting a network. The specific functions of these structures are not known but they may contribute to the emerging multiple roles of the urothelium associated with the generation of bladder sensation.







Similar content being viewed by others
Abbreviations
- β1-sGC:
-
β1 subunit of soluble guanylyl cyclase
- BUCs:
-
Basal urothelial cells
- BV:
-
Blood vessel
- cGKI:
-
cGMP-dependent protein kinase I
- cGMP:
-
Cyclic guanosine mono-phosphate
- DEANO:
-
Diethylamine-NONOate
- IBMX:
-
Isobutyl-methyl-xanthine
- IM-ICs:
-
Intra-muscular interstitial cells
- IUCs:
-
Intermediate urothelial cells
- NO:
-
Nitric oxide
- nNOS:
-
Neuronal nitric oxide synthase
- MC-ICs:
-
Muscle-coat interstitial cells
- sGC:
-
Soluble guanylyl cyclase
- SM-ICs:
-
Surface muscle interstitial cells
- SUCs:
-
Surface urothelial cells
- SU-ICs:
-
Sub-urothelial interstitial cells
References
Andersson KE, Arner A (2004) Urinary bladder contraction and relaxation: physiology and pathophysiology. Physiol Rev 84:935–986
Becker EM, Alonso-Alija C, Apeler H, Gerzer R, Minuth T, Pleiss U, Schmidt P, Schramm M, Schröder H, Schroeder W, Steinke W, Straub A, Stasch JP (2001) NO-independent regulatory site of direct sGC stimulators like YC-1 and BAY 41-2272. BMC Pharmacol 1:1–12
Behrends S, Kempfert J, Mietens A, Koglin M, Scholz H, Middendorf R (2001) Developmental changes of nitric oxide-sensitive guanylyl cyclase expression in pulmonary arteries. Biochem Biophys Res Commun 283:883–887
Bellamy TC, Wood J, Goodwin DA, Garthwaite J (2000) Rapid desensitization of the nitric oxide receptor, soluble guanylyl cyclase, underlies diversity of cellular cGMP responses. Proc Natl Acad Sci USA 97:2928–2933
Birder LA, Apodaca G, Groat WC de, Kanai AJ (1998) Adrenergic- and capsaicin-evoked nitric oxide release from urothelium and afferent nerves in urinary bladder. Am J Physiol 275:F226–F229
Birder LA, Kanai AJ, Groat WC de, Kiss S, Nealen ML, Burke NE, Dineley KE, Watkins S, Reynolds IJ, Caterina MJ (2001) Vanilloid receptor expression suggests a sensory role for urinary bladder epithelial cells. Proc Natl Acad Sci USA 98:13396–13401
Birder LA, Nealen ML, Kiss S, Groat WC de, Caterina MJ, Wang E, Apodaca G, Kanai AJ (2002) Beta-adrenoceptor agonists stimulate endothelial nitric oxide synthase in rat urinary bladder urothelial cells. J Neurosci 22:8063–8070
Burnstock G (2001) Purinergic signalling in the lower urinary tract. In: Abbracchio MP, Williams M (eds) Handbook of experimental pharmacology, vol 151/I. Purinergic and pyrimidinergic signalling in molecular, nervous and urogenitary system function. Springer, Berlin Heidelberg New York, pp 10-100
Davidson RA, McCloskey KD (2005) Morphology and localization of interstitial cells in the guinea pig bladder: structural relationships with smooth muscle and neurons. J Urol 173:1385–1390
De Vente J, Steinbusch HWM (1999) Immunocytochemical analysis of cyclic nucleotides. In: Maines MD (ed) Current protocols of toxicology. Wiley, New York, pp 10.7.1–10.7.17
De Vente J, Hopkins DA, Markerink-van Ittersum M, Steinbusch HWM (1998a) Nitric oxide-mediated cGMP production in the islands of Calleja in the rat. Brain Res 789:175–178
De Vente J, Hopkins DA, Markerink-van Ittersum M, Emson PC, Schmidt HHHW, Steinbusch HWM (1998b) Distribution of nitric oxide synthase and nitric oxide-receptive, cyclic GMP-producing structures in the rat brain. Neuroscience 87:207–241
Drake MJ, Harvey IJ, Gillespie JI (2003) Autonomous activity in the isolated guinea pig bladder. Exp Physiol 88:19–30
Ferguson DR (1999) Urothelial function. BJU Int 84:235–242
Ferguson DR, Kennedy I, Burton TJ (1997) ATP is released from rabbit urinary bladder epithelial cells by hydrostatic pressure changes—a possible sensory mechanism? J Physiol (Lond) 505:503–511
Fry CH, Ikeda Y, Harvey R, Wu C, Sui GP (2004) Control of bladder function by peripheral nerves: avenues for novel drug targets.Urology 63(3 Suppl 1):24-31
Gillespie JI (2004a) The autonomous bladder: a view of the origin of bladder overactivity. BJU Int 93:478–483
Gillespie JI (2004b) Noradrenaline inhibits autonomous activity in the isolated guinea pig bladder. BJU Int 93:401–409
Gillespie JI (2004c) Modulation of autonomous contractile activity in the isolated bladder of the guinea pig. BJU Int 93:393–400
Gillespie JI (2005a) Inhibitory actions of calcitonin gene related peptide and capsaicin: evidence for local axonal reflexes in the bladder wall. BJU Int 95:149–156
Gillespie JI (2005b) A developing view of the origins of urgency: the importance of animal models.BJU Int 96 (Suppl 1):22–28
Gillespie JI, Harvey IJ, Drake MJ (2003) Agonist and nerve induced phasic activity in the isolated whole bladder of the guinea pig: evidence for two types of bladder activity. Exp Physiol 88:343–357
Gillespie JI, Markerink-van Ittersum M, De Vente J (2004) cGMP generating cells in the bladder wall: identification of distinct networks of interstitial cells. BJU Int 94:1114–1124
Gillespie JI, Markerink-van Ittersum M, De Vente J (2005) The expression of neuronal nitric oxide synthase (nNOS) and nitric oxide induced changes in cGMP in the urothelial layer of the guinea pig bladder. Cell Tissue Res 321:341–351
Hashitani H, Yanai Y, Suzuki H (2004) Role of interstitial cells and gap junctions in the transmission of spontaneous Ca2+ signals in detrusor smooth muscles of the guinea-pig urinary bladder. J Physiol (Lond) 559:567–581
Ko FN, Wu CC, Kuo SC, Lee FY, Teng CM (1994) YC-1, a novel activator of platelet guanylate cyclase. Blood 84:4226–4233
Lagou M, Drake MJ, Gillespie JI (2005) Volume-induced effects on the isolated bladder: a possible local reflex. BJU Int 94:1356–1365
Lev-Ram V, Jiang T, Wood J, Lawrence DS, Tsien RY (1997) Synergies and coincidence requirements between NO, cGMP and Ca2+ in the induction of cerebellar long-term depression. Neuron 18:1025–1038
Markert T, Vaandrager AB, Gambaryan S, Pohler D, Hausler C, Walter U, De Jonge HR, Jarchau T, Lohmann SM (1995) Endogenous expression of type II cGMP-dependent protein kinase mRNA and protein in rat intestine. Implications for cystic fibrosis transmembrane conductance regulator. J Clin Invest 96:822–830
McCloskey KD, Gurney AM (2002) kit-positive cells in the guinea pig bladder. J Urol 168:832–836
Ott SR, Delago A, Elphick MR (2004) An evolutionarily conserved mechanism for sensitization of soluble guanylyl cyclase reveals extensive nitric oxide-mediated upregulation of cyclic GMP in insect brain. Eur J Neurosci 20:1231–1244
Persson K, Igawa Y, Mattiasson A, Andersson KE (1992) Effects of inhibition of the L-arginine/nitric oxide pathway in the rat lower urinary tract in vivo and in vitro. Br J Pharmacol 107:178–184
Poehler D, Butt E, Meissner J, Mueller S, Lohse M, Walter U, Lohmann SM, Jarchau T (1995) Expression, purification, and characterization of the cGMP-dependent protein kinases I-beta and II using the baculovirus system. FEBS Lett 374:419–425
Rong W, Spyer KM, Burnstock G (2002) Activation and sensitisation of low and high threshold afferent fibres mediated by P2X receptors in the mouse urinary bladder. J Physiol (Lond) 541:591–600
Smet PJ, Jonavicius J, Marshall VR, De Vente J (1996) Distribution of nitric oxide synthase-immunoreactive nerves and identification of the cellular targets of nitric oxide in guinea-pig and human urinary bladder by cGMP immunohistochemistry. Neuroscience 71:337–348
Stasch JP, Becker EM, Alonso-Alija C, Apeler H, Dembosky K, Feurer A, Gerzer R, Minuth T, Perzborn E, Pleiss E, Schroeder H, Schroeder W, Stahl E, Steinke W, Straub A, Schram M (2001) NO-independent regulatory site on soluble guanylate cyclase. Nature 410:212–215
Sui GP, Rothery S, Dupont E, Fry CH, Severs NJ (2002) Gap junctions and connexin expression in human suburothelial interstitial cells. BJU Int 90:118–129
Van Staveren WCG, Markerink-van Ittersum M, Steinbusch HWM, Behrends S, De Vente J (2005) Localization and characterization of cGMP-immunoreactive structures in rat brain slices after NO-dependent and NO-independent stimulation of soluble guanylyl cyclase. Brain Res 1036:77–89
Wiseman OJ, Fowler CJ, Landon DN (2003) The role of the human bladder lamina propria myofibroblast. BJU Int 91:89–93
Yoshida M, Miyamae K, Iwashita H, Otani M, Inadome A (2004) Management of detrusor dysfunction in the elderly: changes in acetylcholine and adenosine triphosphate release during aging. Urology 63 (3 Suppl 1):17–23
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gillespie, J., Markerink-van Ittersum, M. & De Vente, J. Endogenous nitric oxide/cGMP signalling in the guinea pig bladder: evidence for distinct populations of sub-urothelial interstitial cells. Cell Tissue Res 325, 325–332 (2006). https://doi.org/10.1007/s00441-005-0146-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-005-0146-4