Abstract
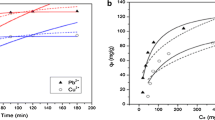
Arsenic (As) in drinking water and its related toxicology are serious concerns nowadays. Development of better techniques related to removal of As from drinking water is an urgent need. Layered double hydroxide (LDH) or hydrotalcite-like compound with the general formula [M 2+1−x M 3+ x (OH)2]x+ Ax−·nH2O can be considered as a good adsorbent for the removal of toxic As from water. Due to large surface area and high anion exchange capacity of LDH, the compound may be a good adsorbent for the removal of As from contaminated water. In this study, the removal of As in aqueous solution by adsorption method based on the calcination–rehydration reaction was investigated in batch experiment at (30 ± 1)°C. Results showed the removal of 99.99% As from a solution of 0.1 ppm of As; the adsorbent required at saturation was 0.10 g/20 ml As solution with 90 min of exposure at (30 ± 1)°C. Factors like pH, adsorbent dose and shaking time influenced the rate of As removal. Experiment showed that the adsorption process follows the Freundlich-type adsorption isotherm. The explanation of adsorption phenomenon is supported by X-ray diffraction pattern.






Similar content being viewed by others
References
Acharyya SK, Shah BA (2007) Arsenic-contaminated groundwater from parts of Damodar fan-delta and west of Bhagirathi River, West Bengal, India: influence of fluvial geomorphology and quaternary morphostratigraphy. Environ Geol 52:489–501
Ahmed J, Goldar BN, Misra S, Jakariya M (2003) Willingness to pay for arsenic free safe drinking water in Bangladesh. World Bank Water and Sanitation Programme, South Asia
Bhattacharya P, Tandukar N, Neku A, Valero AA, Mukherjee AB, Jacks G (2003) Geologic arsenic in groundwaters from Terai alluvial plain of Nepal. J Phys IV France 107:173–176
Bhattacharya P, Ahmed KM, Broms S, Fogelström J, Jacks G, Sracek O (2006) Mobility of arsenic in groundwater in a part of Brahmanbaria district, NE Bangladesh. In: Naidu R, Smith E, Owens G, Bhattacharya P, Nadebaum P (eds) Managing arsenic in the environment: from soil to human health. CSIRO, Melbourne, pp 95–115
Bhattacharya P, Welch AH, Stollenwerk KG, McLaughlin MJ, Jochen B, Panaullah G (2007) Arsenic in the environment: biology and chemistry. Sci Total Environ 379:109–120
Bissen M, Rimmel FH (2003) Arsenic-a review. Part II: oxidation of arsenic and its removal in water treatment. Acta Hydrochim Hydrobiol 31:97–107
Brindley GW, Kikkawa S (1979) A crystal-chemical study of Mg, Al and Ni, Al hydroxy-perchlorates and hydroxyl-carbonates. Am Miner 64:836–843
Bunnell JE, Finkelman RB, Centeno JA, Selinus O (2007) Medical geology: a globally emerging discipline. Geol Acta 5:273–281
Cardoso LP, Tronto J, Crepaldi EL, Valim JB (2003) Removal of benzoate anions from aqueous solution using Mg–Al layered double hydroxides. Mol Cryst Liq Crys 390:49–56
Chakraborti D, Mukherjee SC, Pati S, Sengupta MK, Rahman MM, Chowdhury UK, Lodh D, Chanda CR, Chakraborti AK, Basu OK (2003) Arsenic groundwater contamination in Middle Ganga Plain, Bihar, India. Environ Health Perspect 111:1194–1201
Chao YF, Chen PC, Wang SL (2008) Adsorption of 2,4-D on Mg/Al–NO3 layered double hydroxides with varying layer charge density. Appl Clay Sci 40:193–200
Chibwe K, Jones W (1989) Intercalation of organic and inorganic anions into layered double hydroxides. J Chem Soc Chem Commun 14:926–929
Chuang CL, Fan M, Xu M, Brown RC, Sung S, Saha B, Huang CP (2005) Adsorption of arsenic (V) by activated carbon prepared from oat hulls. Chemosphere 61(4):478–483
Das J, Patra BS, Baliarsingh N, Parida KM (2006) Adsorption of phosphate by layered double hydroxides in aqueous solutions. Appl Clay Sci 32:252–260
Das B, Rahman MM, Nayak B, Pal A, Chowdhury UK, Mukherjee SC, Saha KC, Pati S, Quamruzzaman Q, Chakraborti D (2009) Groundwater arsenic contamination, its health effects and approach for mitigation in West Bengal, India and Bangladesh. Water Qual Expo Health 1:5–21
Dousova B, Machovic V, Kolousek D, Kovanda F, Dornicak V (2003) Sorption of As (V) species from aqueous systems. Water Air Soil Pollut 149:251–267
Eatson DL, Clesceri S, Rice EW, Greenberg AE (2005) Standard methods for the examination of water and wastewater, 21st edn edn. Centennial Edition, USA, pp 4–138
Freundlich HMF (1906) Uber die adsorption in Losungen. Z fur Phys Chem (Leipzig) 57A:385–470
Gillman GP (2006) A simple technology for arsenic removal from drinking water using hydrotalcite. Sci Total Environ 366:926–931
Goh KH, Lim TT, Dong Z (2008) Application of layered double hydroxides for removal of oxyanions: a review. Water Res 42:1343–1368
Goswamee RL, Sengupta P, Bhattacharyya KG, Dutta DK (1998) Adsorption of Cr(VI) in layered double hydroxides. Appl Clay Sci 13:21–34
Jiang JQ (2001) Removing arsenic from groundwater for the developing world-a review. Water Sci Technol 44:89–98
Jie L, Waalkes MP (2008) Liver is a target of arsenic carcinogenesis. Toxicol Sci 105(1):24–32
Koritnig S, Süsse P (1975) Meixnerite, Mg6Al2(OH)18 ·4H2O, ein neues magnesium-aluminium-hydroxid-mineral. Mineral Petrol Mitt 22:79–87
Kovanda F, Kovacsova E, Kolousek D (1999) Removal of anions from solution by calcined hydrotalcite and regeneration of used sorbent in repeated calcination–rehydration–anion exchange processes. Collect Czechoslov Chem Commun 64:1517–1528
Miyata S (1980) Physico-chemical properties of synthetic hydrotalcites in relation to composition. Clays Clay Miner 28(1):50–60
Mukherjee A, Sengupta MK, Hossain MA, Ahmed S, Das B, Nayak B, Lodh D, Rahman MM, Chakraborti D (2006) Arsenic contamination in groundwater: a global perspective with emphasis on the Asian scenario. J Health Popul Nutr 24:142–163
Narita E, Yamagishi T, Tazawa K, Ichijo O, Yoshio U (1995) Uptake behaviour of chelating agents by magnesium-aluminium oxide precursors with reconstruction of hydrotalcite like layer structure. Clay Sci 9(4):187–197
Reichle WT (1986) Synthesis of anionic clay minerals (mixed metal hydroxides, hydrotalcite). Solid State Ionics 22:135–141
Mynemi SCB, Traina SJ, Wayachunas JA, Logan TJ (1998) Vibrational spectroscopy of functional group chemistry and arsenate coordination in ettringite. Geochim Cosmochim Acta 62:3499–3514
Sato T, Tezuka M, Endo T, Shimada M (1987) Removal of sulfuroxyanions by magnesium aluminium oxides and their thermal decomposition. J Chem Technol Biotechnol 39:275–285
Seida Y, Nakano Y (2002) Removal of phosphate by layered double hydroxides containing iron. Water Res 36:1306–1312
Smedley PL, Kinniburgh DG (2002) A review of the source, behaviour and distribution of arsenic in natural waters. Appl Geochem 17:517–568
Taylor HFW (1973) Crystal structures of some double hydroxide minerals. Miner Mag 39:377–389
Tchounwou PB, Patlolla AK, Centeno JA (2003) Carcinogenic and systemic health effects associated with arsenic exposure-a critical review. Toxicol Pathol 31(6):575–588
Tzou YM, Wang SL, Hsu LC, Chang RR, Lin C (2006) Deintercalation of Li/Al LDH and its application to recover adsorbed chromate from used adsorbent. Appl Clay Sci 37:107–114
Wang SC, Bekendorfer KB, Burba JL (1985) Halide removal from fluid organic materials. US Patent 4511710
Wang SL, Hseu RJ, Chang RR, Chiang PN, Chen JH, Tzou YM (2006) Adsorption and thermal desorption of Cr(VI) on Li/Al layered double hydroxide. Colloidal Surf A 277:8–14
Wang SL, Cheng CY, Tzou YM, Liaw RB, Chang TW, Chen JH (2007) Phosphate removal from water using lithium intercalated gibbsite. J Hazard Mater 147:205–212
Wang SL, Liu CH, Wang MK, Chuang YH, Chiang PN (2009) Arsenate adsorption by Mg/Al–NO3 layered double hydroxides with varying the Mg/Al ratio. Appl Clay Sci 43(1):79–85
WHO (1993) Guidelines for drinking water quality, World Health Organisation, Geneva p 41
Williams GR, Hare DO’ (2006) Towards understanding, control and application of layered double hydroxide chemistry. J Mater Chem 16:3065–3074
Xia Y, Wade TJ, Wu K, Li Y, Ning Z, Le XC, He X, Chen B, Feng Y, Mumford JL (2009) Well water arsenic exposure, arsenic induced skin lesions and self-reported morbidity in Inner Mongolia. Int J Environ Res Public Health 6:1010–1025
Yamagishi T, Narita E (1990) Adsorption of carboxylate ions by magnesium-aluminium oxide: formation of layered double hydrotalcite-like compounds. In: Farmer VC, Tardy Y (eds) Proceedings of the 9th international Clay Conference, Strassbourg. Sci Géol Mém 86:145–154
Yamagishi T, Oyanagi Y, Narita E (1993) Removal and fixation of harmful oxometallate ions by formation of layered hydrotalcite-like compounds and their heat treatment. Nippon Kagaku Kaishi 4:329–334
Yang L, Dadwhal M, Shahrivari Z, Ostwal M, Liu PKT, Sahimi M, Tsotsis TT (2006) Adsorption of arsenic on layered double hydroxides: effect of the particle size. Ind J Eng Chem Res 45:4742–4751
You Y, Vance GF, Zhao H (2001) Selenium adsorption on Mg–Al and Zn–Al layered double hydroxides. Appl Clay Sci 20:13–25
Acknowledgments
Authors wish to thank Dr P. G. Rao, Director, NEIST, Jorhat, Assam, India, for his kind consent to carry out the research study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chetia, M., Goswamee, R.L., Banerjee, S. et al. Arsenic removal from water using calcined Mg–Al layered double hydroxide. Clean Techn Environ Policy 14, 21–27 (2012). https://doi.org/10.1007/s10098-011-0365-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10098-011-0365-1