Abstract
Background
Although sunitinib malate has shown significant clinical effect on imatinib-resistant gastrointestinal stromal tumors, with acceptable tolerability and improved prognosis for the patients, the mechanism of resistance to the drug is still under investigation.
Methods
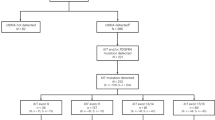
We analyzed findings in 8 patients (seven men and one woman, median age, 59 years) out of 17 patients with imatinib-resistant gastrointestinal stromal tumors who had been treated with sunitinib. Sunitinib was orally administered once a day at a starting dose of 37.5 mg/day, 50 mg/day, or 75 mg/day, with 4 weeks on and 2 weeks off.
Results
All imatinib- as well as sunitinib-resistant lesions showed viable tumor cells strongly re-expressing the KIT protein. Pre-imatinib samples had heterogeneous KIT mutations either in exon 9 (n = 1) or exon 11 (n = 7), and seven imatinib-resistant tumors carried a secondary mutation either in the ATP-binding domain or in the activation loop in the same allele as the primary mutation. Most patients with imatinib-resistant tumors carrying secondary mutations in the ATP-binding domain obtained clinical benefits from sunitinib, whereas some tumors with mutations in the activation loop showed resistance to the drug. A tumor with mutations in exon 11 and 13 of the KIT gene, and showing partial response to sunitinib, harbored a third mutation in the activation loop when sunitinib resistance was shown. All additional secondary and tertiary mutations were located on the same allele as the primary mutation (cis-mutation).
Conclusion
These findings indicate that an additional cis-mutation in the activation loop of the KIT gene could be a potential cause of sunitinib resistance in gastrointestinal stromal tumors.
Similar content being viewed by others
References
Hirota S, Isozaki K, Moriyama Y, et al. (1998) Gain-of-Function mutations of c-kit in human gastrointestinal stromal tumors. Science 279:577–580
Heinrich MC, Corless CL, Duencing A, et al. (2003) PDGFRA activating mutations in gastrointestinal stromal tumors. Science 299:708–710
Nishida T, Hirota S, Taniguchi M, et al. (1998) Familial gastrointestinal stromal tumors with germ line mutation of the KIT gene. Nat Genet 19:323–324
Rubin BP, Heinrich MC, Corless CL (2007) Gastrointestinal stromal tumor. Lancet 369:1731–1741
Demetri GD, von Mehren M, Blanke CD, et al. (2002) Efficacy and safety of imatinib mesylate in advanced gastrointestinal stromal tumors. N Engl J Med 347:472–480
Demetri GD, van Oosterom AT, Garrett CR, et al. (2006) Efficacy and safety of sunitinib in patients with advanced gastrointestinal stromal tumour after failure of imatinib: a randomised controlled trial. Lancet 368:1329–1338
Wakai T, Kanda T, Hirota S, et al. (2004) Late resistance to imatinib therapy in a metastatic gastrointestinal stromal tumour is associated with a second KIT mutation. Br. J Cancer 90:2059–2061
Chen LL, Trent JC, Wu EF, et al. (2004) A missense mutation in KIT kinase domain 1 correlates with imatinib resistance in gastrointestinal stromal tumors. Cancer Res 64:5913–5919
Debiec-Rychter M, Cools J, Dumez H, et al. (2005) Mechanism of resistance to imatinib mesylate in gastrointestinal stromal tumors and activity of the PKC412 inhibitor against imatinib-resistant mutants. Gastroenterology 128:270–279
Antonescu CA, Besmar P, Tianhua G, et al. (2005) Acquired resistance to imatinib in gastrointestinal stromal tumor occurs through secondary gene mutation. Clin Cancer Res 11:4182–4190
Wardelmann E, Thomas N, Merkelbach-Bruse S et al. (2005) Acquired resistance to imatinib in gastrointestinal stromal tumours caused by multiple KIT mutations. Lancet Oncol 6:249–251
Heinrich MC, Corless CL, Blanke CD et al. (2006) Molecular correlates of imatinib resistance in gastrointestinal stromal tumors. J Clin Oncol 24:4764–4774
Wardelmann E, Merkelbach-Bruse S, Pauls K, et al. (2006) Polyclonal evolution of multiple secondary KIT mutations in gastrointestinal stromal tumors under treatment with imatinib mesylate. Clin Cancer Res 12:1743–1749
Bello CL, Sherman L, Zhou J, et al. (2006) Effect of food on the pharmacokinetics of sunitinib malate (SU11248), a multi-targeted receptor tyrosine kinase inhibitor: results from a phase I study in healthy subjects. Anticancer Drugs 17:353–358
Nishitani A, Hirota S, Nishida T, et al. (2005) Different expression of connexin 43 in gastrointestinal stromal tumours between gastric and small intestinal origin. J Pathol 206:377–382
Hirota S, Ohashi A, Nishida T, et al. (2003) Gain-of-function mutations of platelet-derived growth factor receptor alpha gene in gastrointestinal stromal tumors. Gastroenterology 125:660–667
Gorre ME, Mohammed M, Ellwood K, et al. (2001) Clinical resistance to STI-571 cancer therapy caused by BCR-ABL gene mutation or amplification. Science 293:876–880
Cools J, DeAngelo DJ, Gotlib J, et al. (2003) A tyrosine kinase created by fusion of the PDGFRA and FIP1L1 genes as a therapeutic target of imatinib in idiopathic hypereosinophilic syndrome. N Engl J Med 348:1201–1214
Kobayashi S, Boggon TJ, Dayaram T, et al. (2005) EGFR mutation and resistance of non-small-cell lung cancer to gefitinib. N Engl J Med 352:786–792
Prenen H, Cools J, Mentens N, et al. (2006) Efficacy of the kinase inhibitor SU11248 against gastrointestinal stromal tumor mutants refractory to imatinib mesylate. Clin Cancer Res 12: 2622–2627
Demetri GD, Gajiwala K, Christensen J, et al. (2008) Novel mechanisms of resistance to imatinib or sunitinib in KIT mutants from patients with gastrointestinal stromal tumors: structural biology and functional enzymology studies of wild-type and mutated proteins. Proceedings of the American Association for Cancer Research Annual Meeting: 12–16 April 2008; San Diego, CA. AACR 2008 Abstract No. 3184
Heinrich MC, Corless CL, Demetri GD, et al. (2003) Kinase mutations and imatinib response in patients with metastatic gastrointestinal stromal tumor. J Clin Oncol 21:4342–4349
Guo T, Agaram NP, Wong GC, et al. (2007) Sorafenib inhibits the imatinib-resistant KITT670I gatekeeper mutation in gastrointestinal stromal tumor. Clin Cancer Res 13:4874–4881
Nishida T, Kanda T, Nishitani A, et al. (2008) Secondary mutations in the kinase domain of the KIT gene are predominant in imatinibresistant gastrointestinal stromal tumor. Int J Clin Oncol 99:799–804.
Nagar B, Hantschel O, Young MA, et al. (2003) Structural basis for the autoinhibition of c-Abl tyrosine kinase. Cell 112:859–871
Author information
Authors and Affiliations
Consortia
Corresponding author
About this article
Cite this article
Nishida, T., Takahashi, T., Nishitani, A. et al. Sunitinib-resistant gastrointestinal stromal tumors harbor cis-mutations in the activation loop of the KIT gene. Int J Clin Oncol 14, 143–149 (2009). https://doi.org/10.1007/s10147-008-0822-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-008-0822-y