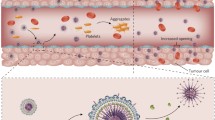
Abstract
Molecular imaging agents are extending the potential of noninvasive medical diagnosis from basic gross anatomical descriptions to complicated phenotypic characterizations based upon the recognition of unique cell-surface biochemical signatures. Although originally the purview of nuclear medicine, “molecular imaging” is now studied in conjunction with all clinically relevant imaging modalities. Of the myriad of particles that have emerged as prospective candidates for clinical translation, perfluorocarbon nanoparticles offer great potential for combining targeted imaging with drug delivery, much like the “magic bullet” envisioned by Paul Ehrlich 100 years ago. Perfluorocarbon nanoparticles, once studied in Phase III clinical trials as blood substitutes, have found new life for molecular imaging and drug delivery. The particles have been adapted for use with all clinically relevant modalities and for targeted drug delivery. In particular, their intravascular constraint due to particle size provides a distinct advantage for angiogenesis imaging and antiangiogenesis therapy. As perfluorocarbon nanoparticles have recently entered Phase I clinical study, this review provides a timely focus on the development of this platform technology and its application for angiogenesis-related pathologies.






Similar content being viewed by others
References
Folkman J (1971) Tumor angiogenesis: therapeutic implications. N Engl J Med 285:1182–1186
Ferrara N et al (2005) Bevacizumab (Avastin), a humanized anti-VEGF monoclonal antibody for cancer therapy. Biochem Biophys Res Commun 333:328–335
Cohen MH et al (2007) FDA drug approval summary: bevacizumab (Avastin) plus carboplatin and paclitaxel as first-line treatment of advanced/metastatic recurrent nonsquamous non-small cell lung cancer. Oncologist 12:713–718
Cohen MH et al (2007) FDA drug approval summary: bevacizumab plus FOLFOX4 as second-line treatment of colorectal cancer. Oncologist 12:356–361
Lang L (2008) FDA approves sorafenib for patients with inoperable liver cancer. Gastroenterology 134:379
Rock EP et al (2007) Food and Drug Administration drug approval summary: sunitinib malate for the treatment of gastrointestinal stromal tumor and advanced renal cell carcinoma. Oncologist 12:107–113
Eskens FA, Verweij J (2006) The clinical toxicity profile of vascular endothelial growth factor (VEGF) and vascular endothelial growth factor receptor (VEGFR) targeting angiogenesis inhibitors; a review. Eur J Cancer 42:3127–3139
Verheul HM, Pinedo HM (2007) Possible molecular mechanisms involved in the toxicity of angiogenesis inhibition. Nat Rev Cancer 7:475–485
Chen X et al (2004) MicroPET and autoradiographic imaging of breast cancer alpha v-integrin expression using 18F- and 64Cu-labeled RGD peptide. Bioconjug Chem 15:41–49
Liu S et al (2007) Evaluation of a 99mTc-labeled cyclic RGD tetramer for noninvasive imaging integrin alpha(v)beta3-positive breast cancer. Bioconjug Chem 18:438–446
Wilmes LJ et al (2007) AG-013736, a novel inhibitor of VEGF receptor tyrosine kinases, inhibits breast cancer growth and decreases vascular permeability as detected by dynamic contrast-enhanced magnetic resonance imaging. Magn Reson Imaging 25:319–327
Liu G et al (2005) Dynamic contrast-enhanced magnetic resonance imaging as a pharmacodynamic measure of response after acute dosing of AG-013736, an oral angiogenesis inhibitor, in patients with advanced solid tumors: results from a phase I study. J Clin Oncol 23:5464–5473
Leach MO et al (2005) The assessment of antiangiogenic and antivascular therapies in early-stage clinical trials using magnetic resonance imaging: issues and recommendations. Br J Cancer 92:1599–1610
Hylton N (2006) Dynamic contrast-enhanced magnetic resonance imaging as an imaging biomarker. J Clin Oncol 24:3293–3298
Flacke S et al (2001) A novel MRI contrast agent for molecular imaging of fibrin: implications for detecting vulnerable plaques. Circulation 104:1280–1285
Winter PM et al (2003) Molecular imaging of angiogenesis in nascent Vx-2 rabbit tumors using a novel alpha(nu)beta3-targeted nanoparticle and 1.5 Tesla magnetic resonance imaging. Cancer Res 63:5838–5843
Schmieder A et al (2005) MR molecular imaging of melanoma angiogenesis with αvβ3-targeted paramagnetic nanoparticles. Magn Reson Med 53:621–627
Sipkins DA et al (1998) Detection of tumor angiogenesis in vivo by alpha v beta 3-targeted magnetic resonance imaging. Nat Med 4:623–626
Mulder WJ et al (2005) MR molecular imaging and fluorescence microscopy for identification of activated tumor endothelium using a bimodal lipidic nanoparticle. FASEB J 19:2008–2010
Mulder WJ et al (2006) Lipid-based nanoparticles for contrast-enhanced MRI and molecular imaging. NMR Biomed 19:142–164
Mulder WJ et al (2007) Early in vivo assessment of angiostatic therapy efficacy by molecular MRI. FASEB J 21:378–383
Frias JC et al (2004) Recombinant HDL-like nanoparticles: a specific contrast agent for MRI of atherosclerotic plaques. J Am Chem Soc 126:16316–16317
Krafft M (2001) Fluorocarbons and fluorinated amphiphiles in drug delivery and biomedical research. Adv Drug Del Rev 47:209–228
De Nichilo M et al (1993) Granulocyte-macrophage and macrophage colony-stimulating factors differentially regulate alpha v integrin expression on cultured human macrophages. Proc Natl Acad Sci USA 90:2517–2521
Helluin O et al (2000) The activation state of alpha v beta 3 regulates platelet and lymphocyte adhesion to intact and thrombin-cleaved osteopontin. J Biol Chem 275:18337–18343
Itoh H et al (1997) The role of integrins in saphenous vein vascular smooth muscle cell migration. J Vasc Surg 25:1061–1069
Carreiras F et al (1996) Expression and localization of alpha v integrins and their ligand vitronectin in normal ovarian epithelium and in ovarian carcinoma. Gynecol Oncol 62:260–267
Kageshita T et al (2000) Differential clinical significance of alpha(v)Beta(3) expression in primary lesions of acral lentiginous melanoma and of other melanoma histotypes. Int J Cancer 89:153–159
Weissleder R et al (2001) Size optimization of synthetic graft copolymers for in vivo angiogenesis imaging. Bioconjug Chem 12:213–219
Ruoslahti E (1988) Fibronectin and its receptors. Annu Rev Biochem 57:375–413
Sadeghi MM et al (2004) Detection of injury-induced vascular remodeling by targeting activated alphavbeta3 integrin in vivo. Circulation 110:84–90
Mattrey RF (1994) The potential role of perfluorochemicals (PFCs) in diagnostic imaging. Artif Cells Blood Substit Immobil Biotechnol 22:295–313
Mattrey R et al (1982) Perfluoroctylbromide: a liver/spleen = specific and tumor-imaging ultrasound contrast material. Radiology 145:759–762
Lanza G et al (1996) A novel site-targeted ultrasonic contrast agent with broad biomedical application. Circulation 94:3334–3340
Lanza G et al (1998) In vitro characterization of a novel, tissue-targeted ultrasonic contrast system with acoustic microscopy. J Acoust Soc Am 104:3665–3672
Hall CS et al (2000) Experimental determination of phase velocity of perfluorocarbons: applications to targeted contrast agents. IEEE Trans Ultrason Ferroelec Freq Contr 47:75–84
Marsh JN et al (2002) Improvements in the ultrasonic contrast of targeted perfluorocarbon nanoparticles using an acoustic transmission line model. IEEE Trans Ultrason Ferroelectr Freq Control 49:29–38
Hall C et al (2001) Temperature dependence of ultrasonic enhancement with a site-targeted contrast agent. J Acous Soc AM 110:1677–1684
Hughes MS et al (2007) Properties of an entropy-based signal receiver with an application to ultrasonic molecular imaging. J Acoust Soc Am 121:3542–3557
Hughes MS et al (2009) Application of Renyi entropy for ultrasonic molecular imaging. J Acoust Soc Am 125:3141–3145
Winter PM et al (2003) Improved molecular imaging contrast agent for detection of human thrombus. Magn Reson Med 50:411–416
Winter P et al (2005) Improved paramagnetic chelate for molecular imaging with MRI. J Magn Magn Mater 293:540–545
Bachert P (1998) Pharmacokinetics using fluorine NMR in vivo. Prog Nucl Magn Reson Spectrosc 33:1–56
Wolf W et al (2000) 19F-MRS studies of fluorinated drugs in humans. Adv Drug Deliv Rev 41:55–74
Kaneda MM et al (2009) Perfluorocarbon nanoemulsions for quantitative molecular imaging and targeted therapeutics. Ann Biomed Eng 37:1922–1933
Lanza GM et al (1996) A novel site-targeted ultrasonic contrast agent with broad biomedical application. Circulation 95:3334–3340
Yu X et al. (2000) Molecular characterization of thrombus using bimodal 1H/19F MR imaging with a novel fibrin-targeted nanoparticulate contrast agent. Proc Int Soc Magn Reson Med 8:465
Morawski AM et al (2004) Quantitative “magnetic resonance immunohistochemistry” with ligand-targeted 19F nanoparticles. Magn Reson Med 52:1255–1262
Caruthers SD et al (2006) In vitro demonstration using 19F magnetic resonance to augment molecular imaging with paramagnetic perfluorocarbon nanoparticles at 1.5 Tesla. Invest Radiol 41:305–312
Neubauer AM et al (2007) Fluorine cardiovascular magnetic resonance angiography in vivo at 1.5 T with perfluorocarbon nanoparticle contrast agents. J Cardiovasc Magn Reson 9:565–573
Waters EA et al (2008) Detection and quantification of angiogenesis in experimental valve disease with integrin-targeted nanoparticles and 19-fluorine MRI/MRS. J Cardiovasc Magn Reson 10:43
Waters EA et al (2008) Detection of targeted perfluorocarbon nanoparticle binding using 19F diffusion weighted MR spectroscopy. Magn Reson Med 60:1232–1236
Haubner R et al (2001) Glycosylated RGD-containing peptides: tracer for tumor targeting and angiogenesis imaging with improved biokinetics. J Nucl Med 42:326–336
Haubner R et al (1999) Radiolabeled alpha(v)beta3 integrin antagonists: a new class of tracers for tumor targeting. J Nucl Med 40:1061–1071
Janssen ML et al (2002) Tumor targeting with radiolabeled alpha(v)beta(3) integrin binding peptides in a nude mouse model. Cancer Res 62:6146–6151
McQuade P et al (2004) Evaluation of 64Cu- and 125I-radiolabeled bitistatin as potential agents for targeting alpha v beta 3 integrins in tumor angiogenesis. Bioconjug Chem 15:988–996
Chen X et al (2004) MicroPET imaging of brain tumor angiogenesis with 18F-labeled PEGylated RGD peptide. Eur J Nucl Med Mol Imaging 31:1081–1089
Chen X et al (2004) Pharmacokinetics and tumor retention of 125I-labeled RGD peptide are improved by PEGylation. Nucl Med Biol 31:11–19
Chen X et al (2004) 18F-labeled RGD peptide: initial evaluation for imaging brain tumor angiogenesis. Nucl Med Biol 31:179–189
Onthank DC et al (2004) 90Y and 111In complexes of a DOTA-conjugated integrin alpha v beta 3 receptor antagonist: different but biologically equivalent. Bioconjug Chem 15:235–241
Kong G et al (2000) Hyperthermia enables tumor-specific nanoparticle delivery: effect of particle size. Cancer Res 60:4440–4445
Winter PM et al (2003) Molecular imaging of angiogenesis in early-stage atherosclerosis with alpha(v)beta3-integrin-targeted nanoparticles. Circulation 108:2270–2274
Winter PM et al (2008) Minute dosages of alpha(nu)beta3-targeted fumagillin nanoparticles impair Vx-2 tumor angiogenesis and development in rabbits. FASEB J 22:2758–2767
Winter P et al (2008) Antiangiogenic synergism of integrin-targeted fumagillin nanoparticles and atorvastatin in atherosclerosis. J Am Coll Cardiol Img 1:624–634
Schmieder AH et al (2008) Three-dimensional MR mapping of angiogenesis with {alpha}5{beta}1({alpha}{nu}{beta}3)-targeted theragnostic nanoparticles in the MDA-MB-435 xenograft mouse model. FASEB J 22:4179–4189
Hu G et al (2007) Imaging of Vx-2 rabbit tumors with alpha(nu)beta3-integrin-targeted 111In nanoparticles. Int J Cancer 120:1951–1957
Lijowski M et al (2008) High sensitivity: high-resolution SPECT-CT/MR molecular imaging of angiogenesis in the Vx2 model. Invest Radiol 43:100–111
Lanza GM et al (2002) Targeted antiproliferative drug delivery to vascular smooth muscle cells with a magnetic resonance imaging nanoparticle contrast agent: implications for rational therapy of restenosis. Circulation 106:2842–2847
Soman N et al (2008) Synthesis and characterization of stable fluorocarbon nanostructures as drug delivery vehicles for cytolytic peptides. Nano Lett 8:1131–1136
Crowder KC et al (2005) Sonic activation of molecularly-targeted nanoparticles accelerates transmembrane lipid delivery to cancer cells through contact-mediated mechanisms: implications for enhanced local drug delivery. Ultrasound Med Biol 31:1693–1700
Cyrus T et al (2008) Intramural delivery of rapamycin with alphavbeta3-targeted paramagnetic nanoparticles inhibits stenosis after balloon injury. Arterioscler Thromb Vasc Biol 28:820–826
Winter P et al (2006) Endothelial alpha(nu)beta(3)-Integrin targeted fumagillin nanoparticles inhibit angiogenesis in atherosclerosis. Arterioscler Thromb Vasc Biol 26:2103–2109
Soman N et al (2009) A platform of molecularly targeted nanostructures for anticancer therapy with cytolytic peptides. J Clin Invest 119:2830–2842
Marsh J et al (2007) Fibrin-targeted perfluorocarbon nanoparticles for targeted thrombolysis. Nanomedicine 2:533–543
Liu S et al (1998) Structure of human methionine aminopeptidase-2 complexed with fumagillin. Science 282:1324–1327
Sin N et al (1997) The anti-angiogenic agent fumagillin covalently binds and inhibits the methionine aminopeptidase, MetAP-2. Proc Natl Acad Sci USA 94:6099–6103
Bergers G et al (1999) Effects of angiogenesis inhibitors on multistage carcinogenesis in mice. Science 284:808–812
Castronovo V, Belotti D (1996) TNP-470 (AGM-1470): mechanisms of action and early clinical development. Eur J Cancer 32A:2520–2527
Konno H et al (1996) Efficacy of an angiogenesis inhibitor, TNP-470, in xenotransplanted human colorectal cancer with high metastatic potential. Cancer 77:1736–1740
Shusterman S et al (2001) The angiogenesis inhibitor tnp-470 effectively inhibits human neuroblastoma xenograft growth, especially in the setting of subclinical disease. Clin Cancer Res 7:977–984
Bhargava P et al (1999) A Phase I and pharmacokinetic study of TNP-470 administered weekly to patients with advanced cancer. Clin Cancer Res 5:1989–1995
Kudelka AP et al (1998) Complete remission of metastatic cervical cancer with the angiogenesis inhibitor TNP-470. N Engl J Med 338:991–992
Kudelka AP et al (1997) A phase I study of TNP-470 administered to patients with advanced squamous cell cancer of the cervix. Clin Cancer Res 3:1501–1505
Logothetis CJ et al (2001) Phase I trial of the angiogenesis inhibitor TNP-470 for progressive androgen-independent prostate cancer. Clin Cancer Res 7:1198–1203
Offodile R et al (1999) Regression of metastatic breast cancer in a patient treated with the anti-angiogenic drug TNP-470. Tumori 85:51–53
Schmieder AH et al (2005) Molecular MR imaging of melanoma angiogenesis with alpha (v) beta (3)-targeted paramagnetic nanoparticles. Magn Reson Med 53:621–627
Kim S et al (2000) Regulation of integrin alpha v beta 3-mediated endothelial cell migration and angiogenesis by integrin alpha 5 beta1 and protein kinase A. J Biol Chem 275:33920–33928
Boudreau N, Varner J (2004) The homeobox transcription factor Hox D3 promotes integrin alpha 5 beta 1 expression and function during angiogenesis. J Biol Chem 279:4862–4868
Moreno PR et al (2004) Plaque neovascularization is increased in ruptured atherosclerotic lesions of human aorta: implications for plaque vulnerability. Circulation 110:2032–2038
Moreno PR et al (2006) Neovascularization in human atherosclerosis. Circulation 113:2245–2252
Virmani R et al (2005) Atherosclerotic plaque progression and vulnerability to rupture: angiogenesis as a source of intraplaque hemorrhage. Arterioscler Thromb Vasc Biol 25:2054–2061
Bjornheden T et al (1999) Evidence of hypoxic areas within the arterial wall in vivo. Arterioscler Thromb Vasc Biol 19:870–876
Boyle JJ et al (2000) Expression of angiogenic factor thymidine phosphorylase and angiogenesis in human atherosclerosis. J Pathol 192:234–242
Khatri JJ et al (2004) Vascular oxidant stress enhances progression and angiogenesis of experimental atheroma. Circulation 109:520–525
de Boer OJ et al (1999) Leucocyte recruitment in rupture prone regions of lipid-rich plaques: a prominent role for neovascularization? Cardiovas Res 41:443–449
Kolodgi F et al (2007) Eliminating plaque angiogenesis: reply. J Am Coll Cardiol 50:1521
Khurana R et al (2005) Role of angiogenesis in cardiovascular disease: a critical appraisal. Circulation 112:1813–1824
Jain RK et al (2007) Antiangiogenic therapy for normalization of atherosclerotic plaque vasculature: a potential strategy for plaque stabilization. Nat Clin Pract Cardiovasc Med 4:491–502
Pasternak RC et al (2002) ACC/AHA/NHLBI clinical advisory on the use and safety of statins. Circulation 106:1024–1028
Girona J et al (1999) Simvastatin decreases aldehyde production derived from lipoprotein oxidation. Am J Cardiol 83:846–851
Kimura M et al (1997) Effects of fluvastatin on leukocyte-endothelial cell adhesion in hypercholesterolemic rats. Arterioscler Thromb Vasc Biol 17:1521–1526
Verhoeven BA et al (2006) Statin treatment is not associated with consistent alterations in inflammatory status of carotid atherosclerotic plaques: a retrospective study in 378 patients undergoing carotid endarterectomy. Stroke 37:2054–2060
Laufs U et al (1998) Upregulation of endothelial nitric oxide synthase by HMG CoA reductase inhibitors. Circulation 97:1129–1135
Sukhova GK et al (2002) Statins reduce inflammation in atheroma of nonhuman primates independent of effects on serum cholesterol. Arterioscler Thromb Vasc Biol 22:1452–1458
Koutouzis M et al (2007) Statin treated patients have reduced intraplaque angiogenesis in carotid endarterectomy specimens. Atherosclerosis 192:457–463
Acknowledgments
Financial, equipment, or material support: AHS, PMW, SAW, SCD, and GL are supported by NCI, NHLBI, NIBIB (HL-78631, HL-73646, HL094470, and CA-119342) and Philips Healthcare. SAW and GML are scientific cofounders of Kereos, Inc.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lanza, G.M., Winter, P.M., Caruthers, S.D. et al. Theragnostics for tumor and plaque angiogenesis with perfluorocarbon nanoemulsions. Angiogenesis 13, 189–202 (2010). https://doi.org/10.1007/s10456-010-9166-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10456-010-9166-0