Abstract
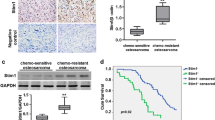
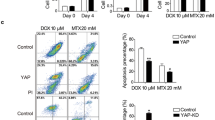
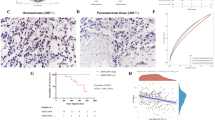
To identify apoptosis genes involved in the multidrug resistance of osteosarcoma, a multidrug resistant human osteosarcoma cell line (U-2 OS MR) was established. Apoptosis gene microarray analysis demonstrated that GADD45α was significantly induced in U-2 OS cells after exposure to paclitaxel (P < 0.0001). However, the induction of GADD45α did not occur in U-2 OS MR cells. Subsequent analysis by Western blot confirmed that the expression of GADD45α could be significantly induced by paclitaxel and doxorubicin in U-2 OS cells but not in U-2 OS MR cells. Furthermore, the paclitaxel or doxorubicin treated U-2 OS and KH OS cells have a higher percentage of apoptotic cells when compared with U-2 OS MR and KH OS R2 cells treated with the same drugs. When GADD45α was transfected into U-2 OS MR or KH OS R2 and expressed, the cells became more sensitive to chemotherapeutic drugs. These results suggest that GADD45α may play a role in drug-induced apoptosis, as well as multidrug resistance in osteosarcoma cells.









Similar content being viewed by others
References
Schwartz CL, Gorlick R, Teot L, Krailo M, Chen Z, Goorin A et al (2007) Multiple drug resistance in osteogenic sarcoma: INT0133 from the Children’s Oncology Group. J Clin Oncol 25:2057–2062. doi:10.1200/JCO.2006.07.7776
Provisor AJ, Ettinger LJ, Nachman JB, Krailo MD, Makley JT, Yunis EJ et al (1997) Treatment of nonmetastatic osteosarcoma of the extremity with preoperative and postoperative chemotherapy: a report from the Children’s Cancer Group. J Clin Oncol 15:76–84
Marcove RC, Mike V, Hajek JV, Levin AG, Hutter RV (1970) Osteogenic sarcoma under the age of twenty-one. A review of one hundred and forty-five operative cases. J Bone Joint Surg Am 52:411–423
Chou AJ, Gorlick R (2006) Chemotherapy resistance in osteosarcoma: current challenges and future directions. Expert Rev Anticancer Ther 6:1075–1085. doi:10.1586/14737140.6.7.1075
Mimeault M, Hauke R, Batra SK (2008) Recent advances on the molecular mechanisms involved in the drug resistance of cancer cells and novel targeting therapies. Clin Pharmacol Ther 83:673–691. doi:10.1038/sj.clpt.6100296
Hunter AM, LaCasse EC, Korneluk RG (2007) The inhibitors of apoptosis (IAPs) as cancer targets. Apoptosis 12:1543–1568. doi:10.1007/s10495-007-0087-3
Prabhudesai SG, Rekhraj S, Roberts G, Darzi AW, Ziprin P (2007) Apoptosis and chemo-resistance in colorectal cancer. J Surg Oncol 96:77–88. doi:10.1002/jso.20785
Johnstone RW, Ruefli AA, Lowe SW (2002) Apoptosis: a link between cancer genetics and chemotherapy. Cell 108:153–164. doi:10.1016/S0092-8674(02)00625-6
Makin G, Hickman JA (2000) Apoptosis and cancer chemotherapy. Cell Tissue Res 301:143–152. doi:10.1007/s004419900160
Duan Z, Feller AJ, Penson RT, Chabner BA, Seiden MV (1999) Discovery of differentially expressed genes associated with paclitaxel resistance using cDNA array technology: analysis of interleukin (IL) 6, IL-8, and monocyte chemotactic protein 1 in the paclitaxel-resistant phenotype. Clin Cancer Res 5:3445–3453
Lourda M, Trougakos IP, Gonos ES (2007) Development of resistance to chemotherapeutic drugs in human osteosarcoma cell lines largely depends on up-regulation of Clusterin/Apolipoprotein J. Int J Cancer 120:611–622. doi:10.1002/ijc.22327
Duan Z, Duan Y, Lamendola DE, Yusuf RZ, Naeem R, Penson RT et al (2003) Overexpression of MAGE/GAGE genes in paclitaxel/doxorubicin-resistant human cancer cell lines. Clin Cancer Res 9:2778–2785
Elstein KH, Zucker RM (1994) Comparison of cellular and nuclear flow cytometric techniques for discriminating apoptotic subpopulations. Exp Cell Res 211:322–331. doi:10.1006/excr.1994.1094
Darzynkiewicz Z, Juan G, Li X, Gorczyca W, Murakami T, Traganos F (1997) Cytometry in cell necrobiology: analysis of apoptosis and accidental cell death (necrosis). Cytometry 27:1–20. doi:10.1002/(SICI)1097-0320(19970101)27:1<1::AID-CYTO2>3.0.CO;2-L
Belloc F, Dumain P, Boisseau MR, Jalloustre C, Reiffers J, Bernard P et al (1994) A flow cytometric method using Hoechst 33342 and propidium iodide for simultaneous cell cycle analysis and apoptosis determination in unfixed cells. Cytometry 17:59–65. doi:10.1002/cyto.990170108
Jiang T, Soprano DR, Soprano KJ (2007) GADD45A is a mediator of CD437 induced apoptosis in ovarian carcinoma cells. J Cell Physiol 212:771–779. doi:10.1002/jcp.21073
Hsu CA, Rishi AK, Su-Li X, Gerald TM, Dawson MI, Schiffer C et al (1997) Retinoid induced apoptosis in leukemia cells through a retinoic acid nuclear receptor-independent pathway. Blood 89:4470–4479
Rishi AK, Sun RJ, Gao Y, Hsu CK, Gerald TM, Sheikh MS et al (1999) Post-transcriptional regulation of the DNA damage-inducible gadd45 gene in human breast carcinoma cells exposed to a novel retinoid CD437. Nucleic Acids Res 27:3111–3119. doi:10.1093/nar/27.15.3111
Lievre A, Bachet JB, Boige V, Cayre A, Le Corre D, Buc E et al (2008) KRAS mutations as an independent prognostic factor in patients with advanced colorectal cancer treated with cetuximab. J Clin Oncol 26:374–379. doi:10.1200/JCO.2007.12.5906
Smith GB, Mocarski ES (2005) Contribution of GADD45 family members to cell death suppression by cellular Bcl-xL and cytomegalovirus vMIA. J Virol 79:14923–14932. doi:10.1128/JVI.79.23.14923-14932.2005
Takekawa M, Saito H (1998) A family of stress-inducible GADD45-like proteins mediate activation of the stress-responsive MTK1/MEKK4 MAPKKK. Cell 95:521–530. doi:10.1016/S0092-8674(00)81619-0
Miyake Z, Takekawa M, Ge Q, Saito H (2007) Activation of MTK1/MEKK4 by GADD45 through induced N-C dissociation and dimerization-mediated trans autophosphorylation of the MTK1 kinase domain. Mol Cell Biol 27:2765–2776. doi:10.1128/MCB.01435-06
Tront JS, Hoffman B, Liebermann DA (2006) Gadd45a suppresses Ras-driven mammary tumorigenesis by activation of c-Jun NH2-terminal kinase and p38 stress signaling resulting in apoptosis and senescence. Cancer Res 66:8448–8454. doi:10.1158/0008-5472.CAN-06-2013
Fayette J, Blay JY (2005) Genetic predictors for drug resistance in soft tissue sarcoma: a review of publications in 2004. Curr Opin Oncol 17:370–375. doi:10.1097/01.cco.0000166650.30793.f2
Sherr CJ (2004) Principles of tumor suppression. Cell 116:235–246. doi:10.1016/S0092-8674(03)01075-4
Weinstein JN, Myers TG, O’Connor PM, Friend SH, Fornace AJ Jr, Kohn KW et al (1997) An information-intensive approach to the molecular pharmacology of cancer. Science 275:343–349. doi:10.1126/science.275.5298.343
Wong RP, Tsang WP, Chau PY, Co NN, Tsang TY, Kwok TT (2007) p53–R273H gains new function in induction of drug resistance through down-regulation of procaspase-3. Mol Cancer Ther 6:1054–1061. doi:10.1158/1535-7163.MCT-06-0336
Pakos EE, Kyzas PA, Ioannidis JP (2004) Prognostic significance of TP53 tumor suppressor gene expression and mutations in human osteosarcoma: a meta-analysis. Clin Cancer Res 10:6208–6214. doi:10.1158/1078-0432.CCR-04-0246
Harkin DP, Bean JM, Miklos D, Song YH, Truong VB, Englert C et al (1999) Induction of GADD45 and JNK/SAPK-dependent apoptosis following inducible expression of BRCA1. Cell 97:575–586. doi:10.1016/S0092-8674(00)80769-2
Chen F, Zhang Z, Leonard SS, Shi X (2001) Contrasting roles of NF-kappaB and JNK in arsenite-induced p53-independent expression of GADD45alpha. Oncogene 20:3585–3589. doi:10.1038/sj.onc.1204442
Liu F, Xie ZH, Cai GP, Jiang YY (2007) The effect of survivin on multidrug resistance mediated by P-glycoprotein in MCF-7 and its adriamycin resistant cells. Biol Pharm Bull 30:2279–2283. doi:10.1248/bpb.30.2279
Sturm I, Petrowsky H, Volz R, Lorenz M, Radetzki S, Hillebrand T et al (2001) Analysis of p53/BAX/p16(ink4a/CDKN2) in esophageal squamous cell carcinoma: high BAX and p16(ink4a/CDKN2) identifies patients with good prognosis. J Clin Oncol 19:2272–2281
Violette S, Poulain L, Dussaulx E, Pepin D, Faussat AM, Chambaz J et al (2002) Resistance of colon cancer cells to long-term 5-fluorouracil exposure is correlated to the relative level of Bcl-2 and Bcl-X(L) in addition to Bax and p53 status. Int J Cancer 98:498–504. doi:10.1002/ijc.10146
Li YC, Tzeng CC, Song JH, Tsia FJ, Hsieh LJ, Liao SJ et al (2006) Genomic alterations in human malignant glioma cells associate with the cell resistance to the combination treatment with tumor necrosis factor-related apoptosis-inducing ligand and chemotherapy. Clin Cancer Res 12:2716–2729. doi:10.1158/1078-0432.CCR-05-1980
Matsuyama R, Togo S, Shimizu D, Momiyama N, Ishikawa T, Ichikawa Y et al (2006) Predicting 5-fluorouracil chemosensitivity of liver metastases from colorectal cancer using primary tumor specimens: three-gene expression model predicts clinical response. Int J Cancer 119:406–413. doi:10.1002/ijc.21843
Zhang M, Guo R, Zhai Y, Yang D (2003) LIGHT sensitizes IFNgamma-mediated apoptosis of MDA-MB-231 breast cancer cells leading to down-regulation of anti-apoptosis Bcl-2 family members. Cancer Lett 195:201–210
Acknowledgments
Dr. C. Yang is supported through a grant from the Chinese Council of Scholarship. Dr. Z. Duan is supported, in part, through a grant from the National Cancer Institute, NIH (Nanotechnology Platform Partnership), R01-CA119617. Support has also been provided by the Gaetagno and Wechsler funds.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, C., Yang, S., Wood, K.B. et al. Multidrug resistant osteosarcoma cell lines exhibit deficiency of GADD45α expression. Apoptosis 14, 124–133 (2009). https://doi.org/10.1007/s10495-008-0282-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-008-0282-x