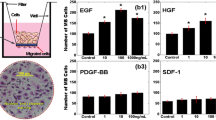
Abstract
Cell migration is essential in many diverse processes ranging from embryonic development to wound healing and immune response. Cancer cells have recently been shown to utilize chemoattraction mechanisms mediated by chemokines and their respective receptors, e.g., the CXCL12/CXCR4 pathway normally found in leukocytes. Here we show that Slit2, a secreted protein signaling through the Roundabout (Robo) receptor as a chemorepellent in axon guidance and neuronal migration, acts as a potent chemoattractant for breast cancer cells. Comparing cell lines specifically metastasizing to either brain or bone, we found significant differences in their responses to CXCL12 and Slit2 treatments, suggesting a role for Slit/Robo signaling in brain metastasis.





Similar content being viewed by others
Abbreviations
- MMP9:
-
Matrix metalloprotease 9
- VEGF:
-
Vascular endothelial growth factor
References
Ruoslahti E (1996) How cancer spreads. Sci Am 275(3):72–77
Rusciano D, Burger MM (1992) Why do cancer cells metastasize into particular organs? Bioessays 14(3):185–194
Müller A, Homey B, Soto H, Ge N, Catron D, Buchanan ME, McClanahan T, Murphy E, Yuan W, Wagner SN et al (2001) Involvement of chemokine receptors in breast cancer metastasis. Nature 410(6824):50–56
Balkwill F, Mantovani A (2001) Inflammation and cancer: back to Virchow?. Lancet 357(9255):539–545
Schmid BC, Rudas M, Rezniczek GA, Leodolter S, Zeillinger R (2004) CXCR4 is expressed in ductal carcinoma in situ of the breast and in atypical ductal hyperplasia. Breast Cancer Res Treat 84(3):247–250
Baggiolini M (1998) Chemokines and leukocyte traffic. Nature 392(6676):565–568
Campbell JJ, Butcher EC (2000) Chemokines in tissue-specific and microenvironment-specific lymphocyte homing. Curr Opin Immunol 12(3):336–341
Wu JY, Feng L, Park HT, Havlioglu N, Wen L, Tang H, Bacon KB, Jiang Z, Zhang X, Rao Y (2001) The neuronal repellent Slit inhibits leukocyte chemotaxis induced by chemotactic factors. Nature 410(6831):948–952
Wong K, Park HT, Wu JY, Rao Y (2002) Slit proteins: molecular guidance cues for cells ranging from neurons to leukocytes. Curr Opin Genet Dev 12(5):583–591
Rothberg JM, Hartley DA, Walther Z, Artavanis-Tsakonas S (1988) Slit: an EGF-homologous locus of D. melanogaster involved in the development of the embryonic central nervous system. Cell 55(6):1047–1059
Seeger M, Tear G, Ferres-Marco D, Goodman CS (1993) Mutations affecting growth cone guidance in Drosophila: genes necessary for guidance toward or away from the midline. Neuron 10(3):409–426
Hu H (1999) Chemorepulsion of neuronal migration by Slit2 in the developing mammalian forebrain. Neuron 23(4):703–711
Wu W, Wong K, Chen J, Jiang Z, Dupuis S, Wu JY, Rao Y (1999) Directional guidance of neuronal migration in the olfactory system by the protein Slit. Nature 400(6742):331–336
Holmes GP, Negus K, Burridge L, Raman S, Algar E, Yamada T, Little MH (1998) Distinct but overlapping expression patterns of two vertebrate slit homologs implies functional roles in CNS development and organogenesis. Mech Dev 79(1–2):57–72
Yoneda T, Williams PJ, Hiraga T, Niewolna M, Nishimura R (2001) A bone-seeking clone exhibits different biological properties from the MDA-MB-231 parental human breast cancer cells and a brain-seeking clone in vivo and in vitro. J Bone Miner Res 16(8):1486–1495
Li HS, Chen JH, Wu W, Fagaly T, Zhou L, Yuan W, Dupuis S, Jiang ZH, Nash W, Gick C et al (1999) Vertebrate slit, a secreted ligand for the transmembrane protein roundabout, is a repellent for olfactory bulb axons. Cell 96(6):807–818
Graham FL, van der Eb AJ (1973) Transformation of rat cells by DNA of human adenovirus 5. Virology 54(2):536–539
Kury FD, Schneeberger C, Sliutz G, Kubista E, Salzer H, Medl M, Leodolter S, Swoboda H, Zeillinger R, Spona J (1990) Determination of HER-2/neu amplification and expression in tumor tissue and cultured cells using a simple, phenol free method for nucleic acid isolation. Oncogene 5(9):1403–1408
Tong D, Czerwenka K, Sedlak J, Schneeberger C, Schiebel I, Concin N, Leodolter S, Zeillinger R (1999) Association of in vitro invasiveness and gene expression of estrogen receptor, progesterone receptor, pS2 and plasminogen activator inhibitor-1 in human breast cancer cell lines. Breast Cancer Res Treat 56(1):91–97
Rao Y, Wong K, Ward M, Jurgensen C, Wu JY (2002) Neuronal migration and molecular conservation with leukocyte chemotaxis. Genes Dev 16(23):2973–2984
Prasad A, Fernandis AZ, Rao Y, Ganju RK (2004) Slit protein-mediated inhibition of CXCR4-induced chemotactic and chemoinvasive signaling pathways in breast cancer cells. J Biol Chem 279(10):9115–9124
Kramer SG, Kidd T, Simpson JH, Goodman CS (2001) Switching repulsion to attraction: changing responses to slit during transition in mesoderm migration. Science 292(5517):737–740
Wang B, Xiao Y, Ding BB, Zhang N, Yuan X, Gui L, Qian KX, Duan S, Chen Z, Rao Y et al (2003) Induction of tumor angiogenesis by Slit-Robo signaling and inhibition of cancer growth by blocking Robo activity. Cancer Cell 4(1):19–29
Liang Y, Annan RS, Carr SA, Popp S, Mevissen M, Margolis RK, Margolis RU (1999) Mammalian homologues of the Drosophila slit protein are ligands of the heparan sulfate proteoglycan glypican-1 in brain. J Biol Chem 274(25):17885–17892
Ronca F, Andersen JS, Paech V, Margolis RU (2001) Characterization of Slit protein interactions with glypican-1. J Biol Chem 276(31):29141–29147
Mertens G, Cassiman JJ, Van den Berghe H, Vermylen J, David G (1992) Cell surface heparan sulfate proteoglycans from human vascular endothelial cells. Core protein characterization and antithrombin III binding properties. J Biol Chem 267(28):20435–20443
Rosenberg RD, Shworak NW, Liu J, Schwartz JJ, Zhang L (1997) Heparan sulfate proteoglycans of the cardiovascular system. Specific structures emerge but how is synthesis regulated? J Clin Invest 100(11 Suppl):S67–S75
Nabeshima K, Inoue T, Shimao Y, Sameshima T (2002) Matrix metalloproteinases in tumor invasion: role for cell migration. Pathol Int 52(4):255–264
Munaut C, Noel A, Hougrand O, Foidart JM, Boniver J, Deprez M (2003) Vascular endothelial growth factor expression correlates with matrix metalloproteinases MT1-MMP, MMP-2 and MMP-9 in human glioblastomas. Int J Cancer 106(6):848–855
Neufeld G, Cohen T, Gengrinovitch S, Poltorak Z (1999) Vascular endothelial growth factor (VEGF) and its receptors. Faseb J 13(1):9–22
Lee TH, Avraham HK, Jiang S, Avraham S (2003) Vascular endothelial growth factor modulates the transendothelial migration of MDA-MB-231 breast cancer cells through regulation of brain microvascular endothelial cell permeability. J Biol Chem 278(7):5277–5284
Kim LS, Huang S, Lu W, Lev DC, Price JE (2004) Vascular endothelial growth factor expression promotes the growth of breast cancer brain metastases in nude mice. Clin Exp Metastasis 21(2):107–118
Latil A, Chene L, Cochant-Priollet B, Mangin P, Fournier G, Berthon P, Cussenot O (2003) Quantification of expression of netrins, slits and their receptors in human prostate tumors. Int J Cancer 103(3):306–315
Dallol A, Da Silva NF, Viacava P, Minna JD, Bieche I, Maher ER, Latif F (2002) SLIT2, a human homologue of the Drosophila Slit2 gene, has tumor suppressor activity and is frequently inactivated in lung and breast cancers. Cancer Res 62(20):5874–5880
Dallol A, Forgacs E, Martinez A, Sekido Y, Walker R, Kishida T, Rabbitts P, Maher ER, Minna JD, Latif F (2002) Tumour specific promoter region methylation of the human homologue of the Drosophila Roundabout gene DUTT1 (ROBO1) in human cancers. Oncogene 21(19):3020–3028
Dallol A, Krex D, Hesson L, Eng C, Maher ER, Latif F (2003) Frequent epigenetic inactivation of the SLIT2 gene in gliomas. Oncogene 22(29):4611–4616
Dallol A, Morton D, Maher ER, Latif F (2003) SLIT2 axon guidance molecule is frequently inactivated in colorectal cancer and suppresses growth of colorectal carcinoma cells. Cancer Res 63(5):1054–1058
Acknowledgements
This work was supported by “Medizinisch-wissenschaftlicher Fonds des Bürgermeisters der Bundeshauptstadt Wien”, Project No. 2185,
Author information
Authors and Affiliations
Corresponding author
Additional information
Bernd C. Schmid and Günther A. Rezniczek contributed equally to this work.
Rights and permissions
About this article
Cite this article
Schmid, B.C., Rezniczek, G.A., Fabjani, G. et al. The neuronal guidance cue Slit2 induces targeted migration and may play a role in brain metastasis of breast cancer cells. Breast Cancer Res Treat 106, 333–342 (2007). https://doi.org/10.1007/s10549-007-9504-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-007-9504-0