Abstract
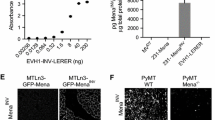
We have studied the gene expression pattern of invasive primary mammary tumor cells using a unique in vivo invasion assay that isolates the invasive tumor cells by chemotaxis. One of the genes upregulated in the invasive tumor cells is Mena, an actin binding protein involved in the regulation of cell motility. There are multiple known splice variants of Mena accounted for by four alternatively included exons, +, ++, +++ and 11a. Using the in vivo invasion assay in rats and mice with mammary tumors we observed that two isoforms of Mena, ++ and +++, are upregulated in the invasive tumor cells and one isoform, 11a, is downregulated. The Mena isoform switching pattern described here may provide a new biomarker for the presence of metastatic cancer cells and for prognosis.




Similar content being viewed by others
References
Condeelis J, Singer RH, Segall JE (2005) The great escape: when cancer cells hijack the genes for chemotaxis and motility. Annu Rev Cell Dev Biol 21:695–718. doi:10.1146/annurev.cellbio.21.122303.120306
Wyckoff J, Wang W, Lin EY et al (2004) A paracrine loop between tumor cells and macrophages is required for tumor cell migration in mammary tumors. Cancer Res 64(19):7022–7029. doi:10.1158/0008-5472.CAN-04-1449
Wang W, Goswami S, Lapidus K et al (2004) Identification and testing of a gene expression signature of invasive carcinoma cells within primary mammary tumors. Cancer Res 64(23):8585–8594. doi:10.1158/0008-5472.CAN-04-1136
Wang W, Wyckoff JB, Goswami S et al (2007) Coordinated regulation of pathways for enhanced cell motility and chemotaxis is conserved in rat and mouse mammary tumors. Cancer Res 67(8):3505–3511. doi:10.1158/0008-5472.CAN-06-3714
Goswami S, Wang W, Wyckoff JB, Condeelis JS (2004) Breast cancer cells isolated by chemotaxis from primary tumors show increased survival and resistance to chemotherapy. Cancer Res 64(21):7664–7667. doi:10.1158/0008-5472.CAN-04-2027
Wang W, Mouneimne G, Sidani M et al (2006) The activity status of cofilin is directly related to invasion, intravasation, and metastasis of mammary tumors. J Cell Biol 173(3):395–404. doi:10.1083/jcb.200510115
Gertler FB, Niebuhr K, Reinhard M, Wehland J, Soriano P (1996) Mena, a relative of VASP and Drosophila Enabled, is implicated in the control of microfilament dynamics. Cell 87:227–239. doi:10.1016/S0092-8674(00)81341-0
Barzik M, Kotova TI, Higgs HN et al (2005) Ena/VASP proteins enhance actin polymerization in the presence of barbed end capping proteins. J Biol Chem 280(31):28653–28662. doi:10.1074/jbc.M503957200
Bear JE, Svitkina TM, Krause M et al (2002) Antagonism between Ena/VASP Proteins and actin filament capping regulates fibroblast motility. Cell 109:509–521. doi:10.1016/S0092-8674(02)00731-6
Philippar U, Roussos ET, Oser M, Yamaguchi H, Kim H, Giampieri S, Wang Y, Goswami S, Wyckoff JB, Sahai E, Condeelis JS, Gertler FB (2008) A Mena invasion isoform potentiates EGF-induced carcinoma cell invasion and metastasis. Dev Cell (in press)
Scott JA, Shewan AM, den Elzen NR et al (2006) Ena/VASP proteins can regulate distinct modes of actin organization at cadherin-adhesive contacts. Mol Biol Cell 17(3):1085–1095. doi:10.1091/mbc.E05-07-0644
Prehoda KE, Lee DJ, Lim WA (1999) Structure of the enabled/VASP homology 1 domain-peptide complex: a key component in the spatial control of actin assembly. Cell 97(4):471–480. doi:10.1016/S0092-8674(00)80757-6
Boeda B et al (2007) Tes, a specific Mena interacting protein, breaks the rules for EVH1 binding. Mol Cell 28:1071–1082. doi:10.1016/j.molcel.2007.10.033
Kuhnel K, Jarchau T, Wolf E et al (2004) The VASP tetramerization domain is a right-handed coiled coil based on a 15-residue repeat. Proc Natl Acad Sci USA 101(49):17027–17032. doi:10.1073/pnas.0403069101
Loureiro JJ, Rubinson DA, Bear JE et al (2002) Critical roles of phosphorylation and actin binding motifs, but not the central proline-rich region, for Ena/vasodilator-stimulated phosphoprotein (VASP) function during cell migration. Mol Biol Cell 13(7):2533–2546. doi:10.1091/mbc.E01-10-0102
Di Modugno F, Bronzi G, Scanlan MJ et al (2004) Human Mena protein, a serex-defined antigen overexpressed in breast cancer eliciting both humoral and CD8 + T-cell immune response. Int J Cancer 109(6):909–918. doi:10.1002/ijc.20094
Di Modugno F, DeMonte L, Balsamo M et al (2007) Molecular cloning of hMena (ENAH) and its splice-variant hMena + 11a. Epidermal growth factor (EGF) increases their expression and stimulates hMena + 11a phosphorylation in breast cancer cell lines. Cancer Res 67:2657–2665. doi:10.1158/0008-5472.CAN-06-1997
Di Modugno F, Mottolese M, Di Benedetto A et al (2006) The cytoskeleton regulatory protein hMena (ENAH) is overexpressed in human benign breast lesions with high risk of transformation and human epidermal growth factor receptor-2-positive/hormonal receptor-negative tumors. Clin Cancer Res 12(5):1470–1478. doi:10.1158/1078-0432.CCR-05-2027
Urbanelli L, Massini C, Emiliani C et al (2006) Characterization of human Enah gene. Biochim Biophys Acta 1759(1–2):99–107
Brinkman BM (2004) Splice variants as cancer biomarkers. Clin Biochem 37(7):584–594. doi:10.1016/j.clinbiochem.2004.05.015
Venables JP (2006) Unbalanced alternative splicing and its significance in cancer. BioEssays 28(4):378–386. doi:10.1002/bies.20390
Wyckoff J, Segall J, Condeelis J (2000) The collection of the motile population of cells from a living tumor. Cancer Res 60:5401–5404
Wyckoff J, Segall JE, Condeelis J (2005) Single-cell imaging in animal tumors in vivo. In: Spector D, Goldman R (eds) Live cell imaging, a laboratory manual, chap 22. Cold Spring Harbor Laboratory Press, pp 409–422
Pino MS, Balsamo M, Di Modugno F et al (2008) Human Mena + 11a isoform serves as a marker of epithelial phenotype and sensitivity to epidermal growth factor receptor inhibition in human pancreatic cancer cell lines. Clin Cancer Res 14(15):4943–4950. doi:10.1158/1078-0432.CCR-08-0436
Acknowledgements
The authors wish to acknowledge Andrew Freidman for technical help, Drs Erik Sahai and Jeffrey Segall for their help in discussions. U.P. was supported by the Anna Fuller Molecular Oncology Fund and the Ludwig Center for Molecular Oncology. This work is supported by NIH CA 100324 (SG and JC) and CA113395 (JC), NIH grant # GM58801 and ICBP grant # 1-U54-CA112967 for FBG, Lega Italiana per la Lotta Contro i Tumori and Associazione Italiana per la Ricerca sul Cancro (AIRC) for PN. SG is the recipient of the Young Investigator Award from Breast Cancer Alliance Inc.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Goswami, S., Philippar, U., Sun, D. et al. Identification of invasion specific splice variants of the cytoskeletal protein Mena present in mammary tumor cells during invasion in vivo. Clin Exp Metastasis 26, 153–159 (2009). https://doi.org/10.1007/s10585-008-9225-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10585-008-9225-8