Abstract
Diarrhea and weight loss are common after pancreaticoduodenectomy, and arise from varying etiologies. An uncommon but important cause for these symptoms is the postoperative activation of silent celiac disease. We sought to describe the clinical presentation, diagnosis, treatment, and follow-up of a series of patients with silent celiac disease unmasked after pancreaticoduodenectomy, and to summarize the existing case reports on this association. A search of the electronic medical record at our institution was performed cross-referencing terms associated with celiac disease and pancreaticoduodenectomy for the years 1976–2004. Cases were then reviewed to ensure that no signs or symptoms attributable to celiac disease were present preoperatively. Seven patients were identified; five were male, and the median age was 56. All patients underwent surgery for a presumed pancreatic or ampullary malignancy. Six patients developed symptoms ultimately attributable to celiac disease immediately after pancreaticoduodenectomy, most commonly diarrhea and weight loss. A single patient had silent celiac disease incidentally diagnosed at pancreaticoduodenectomy that remained silent postoperatively on an unrestricted diet. Symptoms completely resolved in 4 of 6 patients after initiation of a gluten-free diet, with partial improvement in the remaining 2 patients. The median delay from pancreaticoduodenectomy to diagnosis of celiac disease in the 6 symptomatic patients was 6 months. Clinicians should consider celiac disease as a potential diagnosis in patients with failure to thrive and diarrhea after pancreaticoduodenectomy. This entity is uncommon, but may be underrecognized. The underlying mechanism may relate to an increased antigenic load secondary to postsurgical changes in intestinal physiology.


Similar content being viewed by others
References
Nguyen TC, Sohn TA, Cameron JL, Lillemoe KD, Campbell KA, Coleman J, Sauter PK, Abrams RA, Hruban RH, Yeo CJ (2003) Standard vs. radical pancreaticoduodenectomy for periampullary adenocarcinoma: a prospective, randomized trial evaluating quality of life in pancreaticoduodenectomy survivors. J Gastrointest Surg 7:1–11
MacGowan DJL, Hourihane DO, Tanner WA, O’Morain C (1996) Duodeno-jejunal adenocarcinoma as a first presentation of coeliac disease. J Clin Pathol 49:602–604
Kagnoff MF (2005) Overview and pathogenesis of celiac disease. Gastroenterology 128:S10–S18
Murray JA, Van Dyke C, Plevak MF, Dierkhising RA, Zinsmeister AR, Melton LJ (2003) Trends in the identification and clinical features of celiac disease in a North American community, 1950–2001. Clin Gastroenterol Hepatol 1:19–27
Dewar DH, Ciclitira PJ (2005) Clinical features and diagnosis of celiac disease. Gastroenterology 128:S19–S24
Malnick SD, Atali M, Lurie Y, Fraser G, Geltner D (1998) Celiac disease presenting during the puerperium: a report of three cases and a review of the literature. J Clin Gastroenterol 26:164–166
Bai J, Moran C, Martinez C, Niveloni S, Crosetti E, Sambuelli A, Boerr L (1991) Celiac sprue after surgery of the upper gastrointestinal tract. Report of 10 cases with special attention to diagnosis, clinical behavior, and follow-up. J Clin Gastroenterol 13:521–524
Hedberg CA, Melnyk CS, Johnson CF (1966) Gluten enteropathy appearing after gastric surgery. Gastroenterology 50:796–804
Mason CH, Dunk AA (1997) Duodeno-jejunal adenocarcinoma and coeliac disease. J Clin Pathol 50:619
Gebrayel N, Conlon K, Shike M (2000) Coeliac disease diagnosed after pancreaticoduodenectomy. Eur J Surg 166:742–743
Boggi U, Bellini R, Rosetti E, Pietrabissi A, Mosca F (2001) Untractable diarrhea due to late onset celiac disease of the adult following pancreaticoduodenectomy. Hepatogastroenterology 48:1030–1032
Stone CD, Klein S, McDoniel K, Davidson NO, Prakash C, Strasberg SM (2005) Celiac disease unmasked after pancreaticoduodenectomy. JPEN J Parenter Enteral Nutr 29:270–271
Chedid AD, Kruel CRP, Chedid MF, Torresini RJS, Geyer GR (2005) Development of clinical celiac disease after pancreaticoduodenectomy: a potential complication of major upper abdominal surgery. Langenbecks Arch Surg 390:39–41
Tran KTC, Smeenk HG, van Eijck CHJ, et al (2004) Pylorus preserving pancreaticoduodenectomy versus standard Whipple procedure: a prospective, randomized, multicenter analysis of 170 patients with pancreatic and periampullary tumors. Ann Surg 240:738–745
Fort JM, Azpiroz F, Casellas F, Andreu J, Malagelada JR (1996) Bowel habit after cholecystectomy: physiological changes and clinical implications. Gastroenterology 111:617–622
Chang EB, Fedorak RN, Field M (1986) Experimental diabetic diarrhea in rats. Intestinal mucosal denervation hypersensitivity and treatment with clonidine. Gastroenterology 91:564–569
Carroccio A, Cavataio F, Montalto G, Paparo F, Troncone R, Iacono G (2001) Treatment of giardiasis reverses “active coeliac disease to “latent” celiac disease. Eur J Gastroenterol Hepatol 13:1101–1105
Askling J, Linet M, Gridley G, Halstensen TS, Ekstrom K, Ekbom A (2002) Cancer incidence in a population-based cohort of individuals hospitalized with celiac disease or dermatitis herpetiformis. Gastroenterology 123:1428–1435
West J, Logan RF, Smith CJ, Hubbard RB, Card TR (2004) Malignancy and mortality in people with coeliac disease: population based cohort study. BMJ 329:716–719
Matejovic M, Krouzecky A, Rokyta R, Treska V, Spidlen V, Novak I (2004) Effects of intestinal surgery on pulmonary, glomerular, and intestinal permeability, and its relation to the hemodynamics and oxidative stress. Surg Today 34:24–31
Holland J, Carey M, Hughes N, Sweeney K, Byrne PJ, Healy M, Ravi N, Reynolds JV (2005) Intraoperative splanchnic hypoperfusion, increased intestinal permeability, down-regulation of monocyte class II major histocompatibility complex expression, exaggerated acute phase response, and sepsis. Am J Surg 190:393–400
Fasano A, Uzzau S, Fiore C, Margaretten K (1997) The enterotoxic effect of zonula occludens toxin (Zot) on rabbit small intestine involves the paracellular pathway. Gastroenterology 112:839–846
Fasano A, Not T, Wang W, Uzzau S, Berti I, Tommasini A, Goldblum SE (2000) Zonulin, a newly discovered modulator of intestinal permeability, and its expression in coeliac disease. Lancet 355:1518–1519
Ciccocioppo R, Di Sabatino A, Corazza GR (2005) The immune recognition of gluten in coeliac disease. Clin Exp Immunol 140:408–416
Acknowledgments
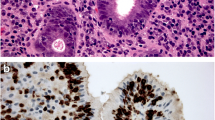
We thank Dr Schuyler Sanderson from the Division of Anatomic Pathology at Mayo Clinic College of Medicine for his assistance in preparing the figure used in this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Maple, J.T., Pearson, R.K., Murray, J.A. et al. Silent Celiac Disease Activated by Pancreaticoduodenectomy. Dig Dis Sci 52, 2140–2144 (2007). https://doi.org/10.1007/s10620-006-9598-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-006-9598-y