Abstract
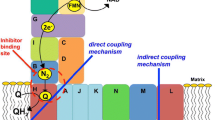
The present investigation utilized the site-directed spin labeling method of electron paramagnetic resonance (EPR) spectroscopy to identify the effect of citrate, the natural ligand, and transport inhibitors on the conformation of the yeast mitochondrial citrate transport protein (CTP) reconstituted in liposomal vesicles. Spin label was placed at six different locations within the CTP in order to monitor conformational changes that occurred near each of the transporter’s two substrate binding sites, as well as at more distant domains within the CTP architecture. We observed that citrate caused little change in the EPR spectra. In contrast the transport inhibitors 1,2,3-benzenetricarboxylate (BTC), pyridoxal 5′-phosphate (PLP), and compound 792949 resulted in spectral changes that indicated a decrease in the flexibility of the attached spin label at each of the six locations tested. The rank order of the immobilizing effect was compound 792949 > PLP > BTC. The four spin-label locations that report on the CTP substrate binding sites displayed the greatest changes in the EPR spectra upon addition of inhibitor. Furthermore, we found that when compound 792949 was added vectorially (i.e., extra- and/or intra-liposomally), the immobilizing effect was mediated nearly exclusively by external reagent. In contrast, upon addition of PLP vectorially, the effect was mediated to a similar extent from both the external and the internal compartments. In combination our data indicate that: i) citrate binding to the CTP substrate binding sites does not alter side-chain and/or backbone mobility in a global manner and is consistent with our expectation that both in the absence and presence of substrate the CTP displays the flexibility required of a membrane transporter; and ii) binding of each of the transport inhibitors tested locked multiple CTP domains into more rigid conformations, thereby exhibiting long-range inter-domain conformational communication. The differential vectorial effects of compound 792949 and PLP are discussed in the context of the CTP homology-modeled structure and potential mechanistic molecular explanations are given.
Similar content being viewed by others
References
Aluvila S, Sun J, Harrison DHT, Walters DE, Kaplan RS (2010) Mol Pharm 77:26–34
Barnes JP, Liang Z, Mchaourab HS, Freed JH, Hubbell WL (1999) Biophys J 76:3298–3306
Berliner LJ, Grunwald J, Hankovszky HO, Hideg K (1982) Anal Biochem 119:450–455
Brunengraber H, Lowenstein JM (1973) FEBS Lett 36:130–132
Budil DE, Lee S, Saxena S, Freed JH (1996) J Magn Resn. A 120:155–189
Columbus L, Hubbell WL (2002) Trends Biochem Sci 27:288–295
Conover TE (1987) Trends Biochem Sci 12:88–89
DeSensi SC, Rangel DP, Beth AH, Lybrand TP, Hustedt EJ (2008) Biophys J 94:3798–3809
Endemann G, Goetz PG, Edmond J, Brunengraber H (1982) J Biol Chem 257:3434–3440
Feix JB, Klug CS (1998) In: Berliner LJ (ed) Biological magnetic resonance, vol. 14, Spin labeling: The next millennium. Plenum Press, New York, pp 251–281
Heisterkamp N, Mulder MP, Langeveld A, Hoeve JT, Wang Z, Roe BA, Groffen J (1995) Genomics 29:451–456
Hubbell WL, Gross A, Langen R, Lietzow MA (1998) Curr Opin Struct Biol 8:649–656
Hubbell WL, Cafiso DS, Altenbach C (2000) Nat Struct Biol 7:735–739
Jensen MV, Joseph JW, Ronnebaum SM, Burgess SC, Sherry AD, Newgard CB (2008) Am J Physiol Endocrinol Metab 295:E1287–E1297
Joseph JW, Jensen MV, Ilkayeva O, Palmieri F, Alarcon C, Rhodes CJ, Newgard CB (2006) J Biol Chem 281:35624–35632
Kaplan RS, Pedersen PL (1985) Anal Biochem 150:97–104
Kaplan RS, Morris HP, Coleman PS (1982) Cancer Res 42:4399–4407
Kaplan RS, Mayor JA, Johnston N, Oliveira DL (1990a) J Biol Chem 265:13379–13385
Kaplan RS, Oliveira DL, Wilson GL (1990b) Arch Biochem Biophys 280:181–191
Kaplan RS, Mayor JA, Wood DO (1993) J Biol Chem 268:13682–13690
Kaplan RS, Mayor JA, Gremse DA, Wood DO (1995) J Biol Chem 270:4108–4114
Kaplan RS, Mayor JA, Brauer D, Kotaria R, Walters DE, Dean AM (2000a) J Biol Chem 275:12009–12016
Kaplan RS, Mayor JA, Kotaria R, Walters DE, Mchaourab HS (2000b) Biochemistry 39:9157–9163
Klare JP, Steinhoff H-J (2009) Photosynth Res 102:377–390
Klug CS, Feix JB (2008) Methods and applications of site-directed spin labeling EPR spectroscopy. In: Correia JJ, Detrich HW (eds) Methods in cell biology. Biophysical tools for biologists, volume one: in vitro techniques. Academic Press, New York, pp 617–658
Kotaria R, Mayor JA, Walters DE, Kaplan RS (1999) J Bioenerg Biomemb 31:543–549
Langen R, Oh KJ, Cascio D, Hubbell WL (2000) Biochemistry 39:8396–8405
Lundblad RL (1991) Chemical reagents for protein modification. CRC, Boca Raton
Ma C, Kotaria R, Mayor JA, Eriks LR, Dean AM, Walters DE, Kaplan RS (2004) J Biol Chem 279:1533–1540
Ma C, Kotaria R, Mayor JA, Remani S, Walters DE, Kaplan RS (2005) J Biol Chem 280:2331–2340
Ma C, Remani S, Kotaria R, Mayor JA, Walters DE, Kaplan RS (2006) Biochim Biophys Acta 1757:1271–1276
Ma C, Remani S, Sun J, Kotaria R, Mayor JA, Walters DE, Kaplan RS (2007) J Biol Chem 282:17210–17220
Mchaourab HS, Lietzow MA, Hideg MA, Hubbell WL (1996) Biochemistry 35:7692–7704
Palmieri F, Stipani I, Quagliariello E, Klingenberg M (1972) Eur J Biochem 26:587–594
Pebay-Peyroula E, Dahout-Gonzalez C, Kahn R, Trézéguet V, Lauquin GJ, Brandolin G (2003) Nature 426:39–44
Remani S, Sun J, Kotaria R, Mayor JA, Brownlee JM, Harrison DHT, Walters DE, Kaplan RS (2008) J Bioenerg Biomembr 40:577–585
Robinson BH, Williams GR, Halperin ML, Leznoff CC (1971) J Biol Chem 246:5280–5286
Walters DE, Kaplan RS (2004) Biophys J 87:907–911
Watson JA, Lowenstein JM (1970) J Biol Chem 245:5993–6002
Xu Y, Mayor JA, Gremse D, Wood DO, Kaplan RS (1995) Biochem Biophys Res Commun 207:783–789
Xu Y, Kakhniashvili DA, Gremse DA, Wood DO, Mayor JA, Walters DE, Kaplan RS (2000) J Biol Chem 275:7117–7124
Author information
Authors and Affiliations
Corresponding authors
Additional information
June A. Mayor and Jiakang Sun contributed equally to this work.
Rights and permissions
About this article
Cite this article
Mayor, J.A., Sun, J., Kotaria, R. et al. Probing the effect of transport inhibitors on the conformation of the mitochondrial citrate transport protein via a site-directed spin labeling approach. J Bioenerg Biomembr 42, 99–109 (2010). https://doi.org/10.1007/s10863-010-9280-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10863-010-9280-0