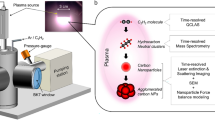
Abstract
Nanocrystals in the lower-nanometer-size range are attracting growing interest due to their unique properties. A simple and versatile atmospheric direct current mini-arc plasma source has been developed to produce nanoparticles as small as a few nanometers. The nanoparticles are formed by direct vaporization of solid precursors followed by a rapid quenching. Both semiconductor tin oxide and metallic silver nanoparticles have been produced at rates of 1–10 mg/h using the mini-arc source. Transmission electron microscopy and X-ray diffraction analyses indicate that most nanoparticles as produced are nonagglomerated and crystalline. Size distributions of nanoparticles measured with an online scanning electrical mobility spectrometer are broader than the self-preserving distribution, suggesting that the nanoparticle growth is coagulation-dominated, and that the particles experience a range of residence times. The electrical charges carried by as-produced aerosol nanoparticles facilitate the manipulation of nanoparticles. The new mini-arc plasma source hence shows promise to accelerate the exploration of nanostructured materials.
Similar content being viewed by others
References
Alivisatos A.P. (1996) Semiconductor clusters, nanocrystals, and quantum dots. Science 271: 933–937
Backman U., Jokiniemi J.K., Auvinen A., Lehtinen K.E.J. (2002) The effect of boundary conditions on gas-phase synthesised silver nanoparticles. J. Nanoparticle Res. 4: 325–335
Botti S., Celeste A., Coppola R. (1998) Particle size control and optical properties of laser-synthesized silicon nanopowders. Appl. Organomet. Chem. 12(5): 361–365
Boulaud D. (1993) Aerosol production by laser ablation. In: Marijnissen C.M., Pratsinis S.E. (eds) Synthesis and Measurement of Ultrafine Particles. Delft University Press, Delft, pp. 31–40
Camata, R.P. (1998) Aerosol synthesis and characterization of silicon nanocrystals, Ph.D. Thesis, California Institute of Technology, Pasadena, CA.
Camata R.P., Atwater H.A., Vahala K.J., Flagan R.C. (1996) Size classification of silicon nanocrystals. Appl. Phys. Lett. 68(22): 3162–3164
Chang H., Lenggoro I.W., Okuyama K., Kim T.O. (2004) Continuous single-step fabrication of nonaggregated, size-controlled and cubic nanocrystalline Y2O3:Eu3+ phosphors using flame spray pyrolysis. Jpn. J. Appl. Phys. Part 1 43(6A): 3535–3539
Chen D.R., Pui D.Y.H., Hummes D., Fissan H., Quant F.R., Sem G.J. (1998) Design and evaluation of a nanometer aerosol differential mobility analyzer (nano-DMA). J. Aerosol Sci. 29(5–6): 497–509
Chen J.H. & G.H. Lu, 2006. Controlled decoration of carbon nanotubes with nanoparticles. Nanotechnology 17(12), 2891–2894
Denbigh K. (1981) The Principles of Chemical Equilibrium 4th ed. Cambridge University Press, Cambridge
Ding Z.F., Quinn B.M., Haram S.K., Pell L.E., Korgel B.A., Bard A.J. (2002) Electrochemistry and electrogenerated chemiluminescence from silicon nanocrystal quantum dots. Science 296(5571): 1293–1297
Flagan R.C., Lunden M.M. (1995) Particle structure control in nanoparticle synthesis from the vapor phase. Mater. Sci. Eng. A 204: 113–124
Flint J.H., Haggerty J.S. (1990) A model for the growth of silicon particles from laser-heated gases. Aerosol Sci. Technol. 13(1): 72–84
Friedlander S.K. (1977) Smoke, dust, and haze: Fundamentals of aerosol behavior. Wiley, New York
Fritzsche W., Taton T.A. (2003) Metal nanoparticles as labels for heterogeneous, chip-based DNA detection. Nanotechnology 14(12): R63-R73
Girshick S.L., Chu C.P., Muno R., Wu C.Y., Yang L., Singh S.K., McMurry P.H. (1993) Thermal plasma synthesis of ultrafine iron particles. J. Aerosol Sci. 24(3): 367–382
Holunga D.M., Flagan R.C., Atwater H.A. (2005) A scalable turbulent mixing aerosol reactor for oxide-coated silicon nanoparticles. Ind. Eng. Chem. Res. 44(16): 6332–6341
Jenkins R., Snyder R.L. (1996) Introduction to X-ray Powder Diffractometry. Wiley, New York
Kennedy M.K., Kruis F.E., Fissan H. (2000) Gas phase synthesis of size selected SnO2 nanoparticles for gas sensor applications. Metastable, Mechanically Alloyed and Nanocrystalline Materials, Pts 1 and 2 343-3:949–954
Kennedy M.K., Kruis F.E., Fissan H., Mehta B.R., Stappert S., Dumpich G. (2003) Tailored nanoparticle films from monosized tin oxide nanocrystals: Particle synthesis, film formation, and size-dependent gas-sensing properties. J. Appl. Phys. 93(1): 551–560
Lee D., Yang S.S., Choi M. (2001) Controlled formation of nanoparticles utilizing laser irradiation in a flame and their characteristics. Appl. Phys. Lett. 79(15): 2459–2461
Lide D.R. (1990–1991) CRC Handbook of Chemistry and Physics. 71st Ed. CRC Press, Boca Raton, pp. 5–70
Lu Y., Liu G.L., Lee L.P. (2005) High-density silver nanoparticle film with temperature-controllable interparticle spacing for a tunable surface enhanced raman scattering substrate. Nano Lett. 5(1): 5–9
Madler L., Roessler A., Pratsinis S.E., Sahm T., Gurlo A., Barsan N., Weimar U. (2006) Direct formation of highly porous gas-sensing films by in situ thermophoretic deposition of flame-made Pt/SnO2 nanoparticles. Sens. Actuators B Chem. 114(1): 283–295
Mahoney W., Andres R.P. (1995) Aerosol synthesis of nanoscale clusters using atmospheric arc evaporator. Mater. Sci. Eng. A 204: 160–164
Makela J.M., Keskinen H., Forsblom T., Keskinen J. (2004) Generation of metal and metal oxide nanoparticles by liquid flame spray process. J. Mater. Sci. 39(8): 2783–2788
Mangolini L., Thimsen E., Kortshagen U. (2005) High-yield plasma synthesis of luminescent silicon nanocrystals. Nano Lett. 5(4): 655–659
Munz R.J., Addona T., da Cruz A.-C. (1999) Application of transferred arcs to the production of nanoparticles. Pure Appl. Chem. 71(10): 1889–1897
Murray C.B., Norris D.J., Bawendi M.G. (1993) Synthesis and characterization of nearly monodisperse CdE (E = sulfur, selenium, tellurium) semiconductor nanocrystallites. J. Am. Chem. Soc. 115(19): 8706–8715
Namiki N., Cho K., Fraundorf P., Biswas P. (2005) Tubular reactor synthesis of doped nanostructured titanium dioxide and its enhanced activation by coronas and soft X-rays. Ind. Eng. Chem. Res. 44(14): 5213–5220
Nichols W.T., Malyavanatham G., Henneke D.E., O’Brien D.T., Becker M.F., Keto J.W. (2002) Bimodal nanoparticle size distributions produced by laser ablation of microparticles in aerosols. J. Nanoparticle Res. 4(5): 423–432
Ostraat M.L., De Blauwe J.W., Green M.L., Bell L.D., Atwater H.A., Flagan R.C. (2001) Ultraclean two-stage aerosol reactor for production of oxide-passivated silicon nanoparticles for novel memory devices. J. Electrochem. Soc. 148(5): G265–G270
Ostraat M.L., De Blauwe J.W., Green M.L., Bell L.D., Brongersma M.L., Casperson J., Flagan R.C., Atwater H.A. (2001) Synthesis and characterization of aerosol silicon nanocrystal nonvolatile floating-gate memory devices. Appl. Phys. Lett. 79(3): 433–435
Park K., Lee D., Rai A., Mukherjee D., Zachariah M.R. (2005) Size-resolved kinetic measurements of aluminum nanoparticle oxidation with single particle mass spectrometry. J. Phys. Chem. B 109(15): 7290–7299
Prakash A., McCormick A.V., Zachariah M.R. (2005) Tuning the reactivity of energetic nanoparticles by creation of a core–shell nanostructure. Nano Lett. 5(7): 1357–1360
Rao N., Girshick S., Heberlein J., McMurry P., Jones S., Hansen D., Micheel B. (1995) Nanoparticle formation using a plasma expansion process. Plasma Chem. Plasma Process. 15(4): 581–606
Rao N., Micheel B., Hansen D., Fandrey C., Bench M., Girshick S., Heberlein J., McMurry P. (1995) Synthesis of nanophase silicon, carbon, and silicon-carbide powders using a plasma expansion process. J. Mater. Res. 10(8): 2073–2084
Sahm T., Madler L., Gurlo A., Barsan N., Pratsinis S.E., Weimar U. (2004) Flame spray synthesis of tin dioxide nanoparticles for gas sensing. Sens. Actuators B Chem. 98(2–3): 148–153
Sankaran R.M., Holunga D., Flagan R.C., Giapis K.P. (2005) Synthesis of blue luminescent si nanoparticles using atmospheric-pressure microdischarges. Nano Lett. 5(3): 537–541
Siegel R.W. (1993) Synthesis and properties of nanophase materials. Mater. Sci. Eng. A 168(2): 189–197
Wang S.C., Flagan R.C. (1990) Scanning electrical mobility spectrometer. Aerosol Sci. Technol. 13: 230–240
Wang X., Hafiz J., Mukherjee R., Renault T., Heberlein J., Girshick S.L., McMurry P.H. (2005) System for in situ characterization of nanoparticles synthesized in a thermal plasma process. Plasma Chem. Plasma Process. 25(5): 439–453
Wiedensohler A. (1988) An approximation of the bipolar charge distribution for particles in the submicron size range. J. Aerosol Sci. 19(3): 387–389
Wiley B., Herricks T., Sun Y.G., Xia Y.N. (2004) Polyol synthesis of silver nanoparticles: Use of chloride and oxygen to promote the formation of single-crystal, truncated cubes and tetrahedrons. Nano Lett. 4(9): 1739
Young R.M., Pfender E. (1985) Generation and behavior of fine particles in thermal plasmas—A review. Plasma Chem. Plasma Process. 5(1): 1–37
Yun C.M., Otani Y., Emi H. (1997) Development of unipolar ion generator-separation of ions in axial direction of flow. Aerosol Sci. Technol. 26: 389–397
Acknowledgments
We thank Professors Jesen Grant and Marija Gajdardziska-Josifovska for providing TEM access and Dr. Bill Tivol and Mr. Donald Robertson for technical support with TEM analyses. The financial support for this study was provided by XMX Corporation, National Science Foundation (CTS-0604079), and University of Wisconsin-Milwaukee. The TIG welder used was donated by Miller Electric Manufacturing Corporation. The work made use of X-Ray Facilities supported by the MRSEC program of the National Science Foundation (CMR-0076097) at the Materials Research Center of Northwestern University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, J., Lu, G., Zhu, L. et al. A simple and versatile mini-arc plasma source for nanocrystal synthesis. J Nanopart Res 9, 203–213 (2007). https://doi.org/10.1007/s11051-006-9168-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11051-006-9168-4