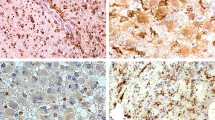
Abstract
The growth pattern of pilocytic astrocytoma (PAs) is unpredictable. Gene expression profiling has recently demonstrated an inverse relationship between myelin basic protein (MBP) expression and progression free survival (PFS) in PAs. We present here the pattern of expression of oligodendroglial differentiation markers (ODMs) in PAs by immunohistochemistry and their correlation with PI and PFS. Sixty-four cases of PA were reviewed and representative sections were stained for Ki-67 and ODMs, including MBP, platelet-derived growth factor receptor-α (PDGFR-α), Olig-1, and Olig-2. Sections were graded semi-quantitatively for intensity (I: 0–3+) and extent (E: 0–4+) of staining. PI was expressed as a percentage of Ki-67 positive cells. Immunoreactivity of MBP, PDGFR-α, Olig-1, and Olig-2 was observed in 84, 56, 97, and 75% of cases, respectively. There was a statistically significant inverse correlation between MBP expression and PI (r 2 = .696, p = .014). A positive correlation was observed between PDGFR-α and PI (r 2 = .727, p = .011). Further analysis showed a significant difference in PFS between low expressors [I + E score ≤ 3] and high expressors (I + E score ≥ 4) for PDGFR-α with p < .001. Notably, there was a significant difference in PFS between high expressors of MBP and high expressors of PDGFR-α with p < .001. These results suggest that expression of ODMs, especially MBP and PDGFR-α, may identify two clinical subsets of PA. In addition, we have shown the expression of 4 different ODMs in PAs, which may support the possibility that PAs arise from oligodendrocyte progenitor/precursor cells probably similar to the O2A progenitor cells in the mouse.




Similar content being viewed by others
Abbreviations
- CNS:
-
Central nervous system
- E:
-
Extent
- I:
-
Intensity
- IHC:
-
Immunohistochemistry
- LI:
-
Labeling index
- MBP:
-
Myelin basic protein
- ODM:
-
Oligodendroglial differentiation marker
- PA:
-
Pilocytic astrocytoma
- PDGFR:
-
Platelet-derived growth factor receptor
- PFS:
-
Progression free survival
- PI:
-
Proliferation index
- WHO:
-
World Health Organization
References
Burger PC, Scheithauer BW, Paulus W, Szymas J, Giannini C, Kleihues P (2000) Pilocytic astrocytoma. In: Kleihues P, Cavenee WK (ed) World Health Organization classification of tumours. Pathology and genetics of tumours of the nervous system. IARC Press, Lyon, pp 45–51
Central Brain Tumor Registry of the United States (CBTRUS). http://www.cbtrus.org/2005–2006/2005–2006.html. Cited 16 March 2007
Gajjar A, Sanford RA, Heideman R, Jenkins JJ, Walter A, Li Y, Langston JW, Muhlbauer M, Boyett JM, Kun LE (1997) Low grade astrocytoma: a decade of experience at St. Jude Children’s Research Hospital. J Clin Oncol 15:2792–2799
Wong KK, Chang YM, Tsang YT, Perlaky L, Su J, Adesina A, Armstrong DL, Bhattacharjee M, Dauser R, Blaney SM, Chintagumpala M, Lau CC (2005) Expression analysis of juvenile pilocytic astrocytomas by oligonucleotide microarray reveals two potential subgroups. Cancer Res 65:76–84
Bowers DC, Gargan L, Kapur P, Reisch JS, Mulne AF, Shapiro KN, Elterman RD, Winick NJ, Margraf LR (2003) Study of the MIB-1 labeling index as a predictor of tumor progression in pilocytic astrocytomas in children and adolescents. J Clin Oncol 21:2968–2973
Roessler K, Bertalanffy A, Jezan H, Ba-Ssalamah A, Slavc I, Czech T, Budka H (2002) Proliferative activity as measured by MIB-1 labeling index and long-term outcome of cerebellar juvenile pilocytic astrocytomas. J Neuro-Oncol 58:141–146
Haapasalo H, Sallinen S-L, Sallinen P, Helen P, Jaaskelainen J, Salmi TT, Paetau A, Paljarvi L, Visakorpi T, Kalimo H (1999) Clinicopathological correlation of cell proliferation, apoptosis and p53 in cerebellar pilocytic astrocytomas. Neuropathol Appl Neurobiol 25:134–142
Machen SK, Prayson RA (1998) Cyclin D1 and MIB-1 immunohistochemistry in pilocytic astrocytomas: a study of 48 cases. Hum Pathol 29:1511–1516
Palma L, Celli P, Mariottini A (2004) Long-term follow-up of childhood cerebellar astrocytomas after incomplete resection with particular reference to arrested growth or spontaneous tumour regression. Acta Neurochir (Wien) 146:581–588
Dirven CM, Koudstaal J, Mooij JJ, Molenaar WM (1998) The proliferative potential of the pilocytic astrocytoma: the relation between MIB-1 labeling and clinical and neuro-radiological follow-up. J Neurooncol 37:9–16
Distelmaier F, Janssen G, Mayatepek E, Schaper J, Gobel U, Rosenbaum T (2006) Disseminated pilocytic astrocytoma involving brain stem and diencephalons: a history of atypical eating disorder and diagnostic delay. J Neurooncol 79:197–201
Aryan HE, Meltzer HS, Lu DC, Ozgur BM, Levy ML, Bruce DA (2005) Management of pilocytic astrocytoma with diffuse leptomeningeal spread: two cases and review of the literature. Childs Nerv Syst 21:477–481
Komotar RJ, Burger PC, Carson BS, Brem H, Olivi A, Goldthwaite PT, Tihan T (2004) Pilocytic and pilomyxoid hypothalamic/chiasmatic astrocytomas. Neurosurgery 54:72–80
Fernandez C, Figarella-Branger D. Girard N, Bouvier-Labit C, Gouvernet J, Paz Paredes A, Lena G. (2003) Pilocytic astrocytomas in children: prognostic factors–a retrospective study of 80 cases. Neurosurgery 53:544–555
Shih AH, Holland EC (2006) Platelet-derived growth factor (PDGF) and glial tumorigenesis. Cancer Lett 232:139–147
Hart IK, Richardson WD, Bolsover SR, Raff MC (1989) PDGF and intracellular signaling in the timing of oligodendrocyte differentiation. J Cell Biol 109:3411–3417
Pringle N, Collarini EJ, Mosley MJ, Heldin CH, Westermark B, Richardson WD (1989) PDGF A chain homodimers drive proliferation of bipotential (O-2A) glial progenitor cells in the developing rat optic nerve. EMBO J 8:1049–1056
Richardson WD, Pringle N, Mosley MJ, Westermark B, Dubois-Dalcq M (1988) A role for platelet-derived growth factor in normal gliogenesis in the central nervous system. Cell 53:309–319
Butt AM, Hornby MF, Ibrahim M, Kirvell S, Graham A, Berry M (1997) PDGF-alpha receptor and myelin basic protein mRNA are not coexpressed by oligodendrocytes in vivo: a double in situ hybridization study in the anterior medullary velum of the neonatal rat. Mol Cell Neurosci 8:311–322
Varela M, Ranuncolo SM, Morandi A, Lastiri J, De Kier Joffe EB, Puricelli LI, Pallotta MG. (2004) EGF-R and PDGF-R, but not bcl-2, overexpression predict overall survival in patients with low-grade astrocytomas. J Surg Oncol 86:34–40
Wegner M (2001) Expression of transcription factors during oligodendroglial development. Microsc Res Tech 52:746–752
Lu QR, Yuk D, Alberta JA, Zhu Z, Pawlitzky I, Chan J, McMahon AP, Stiles CD, Rowitch DH (2000) Sonic hedgehog-regulated oligodendrocytes lineage genes encoding bHLH proteins in the mammalian central nervous system. Neuron 25:317–329
Ohnishi A, Sawa H, Tsuda M, Sawamura Y, Itoh T, Iwasaki Y, Nagashima K (2003) Expression of the oligodendroglial lineage-associated markers Olig1 and Olig2 in different types of human gliomas. J Neuropathol Exp Neurol 62:1052–1059
Shoshan Y, Nishiyama A, Chang A, Mork S, Barnett GH, Cowell JK, Trapp BD, Staugaitis SM (1999) Expression of oligodendrocyte progenitor cell antigens by gliomas: implications for the histogenesis of brain tumors. Proc Natl Acad Sci USA 96:10361–10366
Raff MC, Miller RH, Noble M (1983) A glial progenitor cell that develops in vitro into an astrocyte or an oligodendrocyte depending on culture medium. Nature 303:390–396
Figarella-Branger D, Daniel L, Andre P, Guia S, Renaud W, Monti G, Vivier E, Rougon G (1999) The PEN5 epitope identifies an oligodendrocyte precursor cell population and pilocytic astrocytomas. Am J Pathol 155:1261–1269
Sung CC, Collins R, Li J, Pearl DK, Coons SW, Scheithauer BW, Johnson PC, Yates AJ (1996) Glycolipids and myelin proteins in human oligodendrogliomas. Glycoconj J 13:433–443
Kamitani H, Masuzawa H, Sato J, Kanazawa I (1988) Mixed oligodendroglioma and astrocytoma: fine structural and immunohistochemical studies of four cases. J Neurol Sci 83:219–225
Acknowledgement
The authors thank Pediatric Brain Tumor Foundation (KKW) for their support.
Author information
Authors and Affiliations
Corresponding author
Additional information
This paper was presented, in part, at the 12th International Symposium on Pediatric Neuro-Oncology 2006, Nara, Japan, June 6–9, 2006.
Rights and permissions
About this article
Cite this article
Takei, H., Yogeswaren, S.T., Wong, KK. et al. Expression of oligodendroglial differentiation markers in pilocytic astrocytomas identifies two clinical subsets and shows a significant correlation with proliferation index and progression free survival. J Neurooncol 86, 183–190 (2008). https://doi.org/10.1007/s11060-007-9455-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-007-9455-7