Abstract
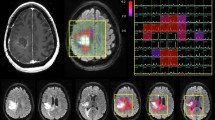
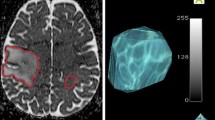
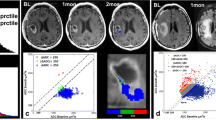
Glioblastoma Multiforme (GBM) are heterogeneous lesions, both in terms of their appearance on anatomic images and their response to therapy. The goal of this study was to evaluate the prognostic value of parameters derived from physiological and metabolic images of these lesions. Fifty-six patients with GBM were scanned immediately before surgical resection using conventional anatomical MR imaging and, where possible, perfusion-weighted imaging, diffusion-weighted imaging, and proton MR spectroscopic imaging. The median survival time was 517 days, with 15 patients censored. Absolute anatomic lesion volumes were not associated with survival but patients for whom the combined volume of contrast enhancement and necrosis was a large percentage of the T2 hyperintense lesion had relatively poor survival. Other volumetric parameters linked with less favorable survival were the volume of the region with elevated choline to N-acetylaspartate index (CNI) and the volume within the T2 lesion that had apparent diffusion coefficient (ADC) less than 1.5 times that in white matter. Intensity parameters associated with survival were the maximum and the sum of levels of lactate and of lipid within the CNI lesion, as well as the magnitude of the 10th percentile of the normalized ADC within the contrast-enhancing lesion. Patients whose imaging parameters indicating that lesions with a relatively large percentage with breakdown of the blood brain barrier or necrosis, large regions with abnormal metabolism or areas with restricted diffusion have relatively poor survival. These parameters may provide useful information for predicting outcome and for the stratification of patients into high or low risk groups for clinical trials.








Similar content being viewed by others
References
Oh J, Henry RG, Pirzkall A, Lu Y, Li X, Catalaa I et al (2004) Survival analysis in patients with glioblastoma multiforme: predictive value of choline-to-N-acetylaspartate index, apparent diffusion coefficient, and relative cerebral blood volume. J Magn Reson Imaging 19:546–554. doi:10.1002/jmri.20039
Salvati M, Cervoni L, Artico M, Caruso R, Gagliardi FM (1998) Long-term survival in patients with supratentorial glioblastoma. J Neurooncol 36:61–64. doi:10.1023/A:1017926603341
Simpson JR, Horton J, Scott C, Curran WJ, Rubin P, Fischbach J et al (1993) Influence of location and extent of surgical resection on survival of patients with glioblastoma multiforme: results of three consecutive Radiation Therapy Oncology Group (RTOG) clinical trials. Int J Radiat Oncol Biol Phys 26:239–244
Kowalczuk A, Macdonald RL, Amidei C, Dohrmann G 3rd, Erickson RK, Hekmatpanah J et al (1997) Quantitative imaging study of extent of surgical resection and prognosis of malignant astrocytomas. Neurosurgery 41:1028–1036. doi:10.1097/00006123-199711000-00004 (discussion 1036–1038)
Lacroix M, Abi-Said D, Fourney DR, Gokaslan ZL, Shi W, De Monte F et al (2001) A multivariate analysis of 416 patients with glioblastoma multiforme: prognosis, extent of resection, and survival. J Neurosurg 95:190–198
Lamborn KR, Chang SM, Prados MD (2004) Prognostic factors for survival of patients with glioblastoma: recursive partitioning analysis. Neuro-Oncology 6:227–235. doi:10.1215/S1152851703000620
McLendon RE, Halperin EC (2003) Is the long-term survival of patients with intracranial glioblastoma multiforme overstated? Cancer 98:1745–1748. doi:10.1002/cncr.11666
Croteau D, Mikkelsen T (2001) Adults with newly diagnosed high-grade gliomas. Curr Treat Options Oncol 2:507–515. doi:10.1007/s11864-001-0072-y
Pirzkall A, McKnight TR, Graves EE, Carol MP, Sneed PK, Wara WW et al (2001) MR-spectroscopy guided target delineation for high-grade gliomas. Int J Radiat Oncol Biol Phys 50:915–928. doi:10.1016/S0360-3016(01)01548-6
Giese A, Westphal M (2001) Treatment of malignant glioma: a problem beyond the margins of resection. J Cancer Res Clin Oncol 127:217–225. doi:10.1007/s004320000188
McKnight TR, von dem Bussche MH, Vigneron DB, Lu Y, Berger MS, McDermott MW et al (2002) Histopathological validation of a three-dimensional magnetic resonance spectroscopy index as a predictor of tumor presence. J Neurosurg 97:794–802
Pirzkall A, Li X, Oh J, Chang S, Berger MS, Larson DA et al (2004) 3D MRSI for resected high-grade gliomas before RT: tumor extent according to metabolic activity in relation to MRI. Int J Radiat Oncol Biol Phys 59:126–137. doi:10.1016/j.ijrobp.2003.08.023
Chen J, Xia J, Zhou YC, Xia LM, Zhu WZ, Zou ML et al (2005) Correlation between magnetic resonance diffusion weighted imaging and cell density in astrocytoma. Zhonghua Zhong Liu Za Zhi 27:309–311
Fuss M, Wenz F, Essig M, Muenter M, Debus J, Herman TS et al (2001) Tumor angiogenesis of low-grade astrocytomas measured by dynamic susceptibility contrast-enhanced MRI (DSC-MRI) is predictive of local tumor control after radiation therapy. Int J Radiat Oncol Biol Phys 51:478–482. doi:10.1016/S0360-3016(01)01691-1
Rosen BR, Belliveau JW, Vevea JM, Brady TJ (1990) Perfusion imaging with NMR contrast agents. Magn Reson Med 14:249–265. doi:10.1002/mrm.1910140211
Chenevert TL, Stegman LD, Taylor JM, Robertson PL, Greenberg HS, Rehemtulla A et al (2000) Diffusion magnetic resonance imaging: an early surrogate marker of therapeutic efficacy in brain tumors. J Natl Cancer Inst 92:2029–2036. doi:10.1093/jnci/92.24.2029
Kono K, Inoue Y, Nakayama K, Shakudo M, Morino M, Ohata K et al (2001) The role of diffusion-weighted imaging in patients with brain tumors. AJNR Am J Neuroradiol 22:1081–1088
Sugahara T, Korogi Y, Kochi M, Ikushima I, Shigematu Y, Hirai T et al (1999) Usefulness of diffusion-weighted MRI with echo-planar technique in the evaluation of cellularity in gliomas. J Magn Reson Imaging 9:53–60. doi:10.1002/(SICI)1522-2586(199901)9:1<53::AID-JMRI7>3.0.CO;2-2
Mardor Y, Pfeffer R, Spiegelmann R, Roth Y, Maier SE, Nissim O et al (2003) Early detection of response to radiation therapy in patients with brain malignancies using conventional and high b-value diffusion-weighted magnetic resonance imaging. J Clin Oncol 21:1094–1100. doi:10.1200/JCO.2003.05.069
Kuznetsov YE, Caramanos Z, Antel SB, Preul MC, Leblanc R, Villemure JG et al (2003) Proton magnetic resonance spectroscopic imaging can predict length of survival in patients with supratentorial gliomas. Neurosurgery 53:565–574. doi:10.1227/01.NEU.0000079331.21178.8E (discussion 574–576)
Tarnawski R, Sokol M, Pieniazek P, Maciejewski B, Walecki J, Miszczyk L et al (2002) 1H-MRS in vivo predicts the early treatment outcome of postoperative radiotherapy for malignant gliomas. Int J Radiat Oncol Biol Phys 52:1271–1276. doi:10.1016/S0360-3016(01)02769-9
Li X, Jin H, Lu Y, Oh J, Chang S, Nelson SJ (2004) Identification of MRI and 1H MRSI parameters that may predict survival for patients with malignant gliomas. NMR Biomed 17:10–20. doi:10.1002/nbm.858
Barker FG 2nd, Prados MD, Chang SM, Gutin PH, Lamborn KR, Larson DA et al (1996) Radiation response and survival time in patients with glioblastoma multiforme. J Neurosurg 84:442–448
Gamburg ES, Regine WF, Patchell RA, Strottmann JM, Mohiuddin M, Young AB (2000) The prognostic significance of midline shift at presentation on survival in patients with glioblastoma multiforme. Int J Radiat Oncol Biol Phys 48:1359–1362. doi:10.1016/S0360-3016(00)01410-3
Keles GE, Lamborn KR, Chang SM, Prados MD, Berger MS (2004) Volume of residual disease as a predictor of outcome in adult patients with recurrent supratentorial glioblastomas multiforme who are undergoing chemotherapy. J Neurosurg 100:41–46
Kreth FW, Berlis A, Spiropoulou V, Faist M, Scheremet R, Rossner R et al (1999) The role of tumor resection in the treatment of glioblastoma multiforme in adults. Cancer 86:2117–2123. doi:10.1002/(SICI)1097-0142(19991115)86:10<2117::AID-CNCR33>3.0.CO;2-8
Keles GE, Anderson B, Berger MS (1999) The effect of extent of resection on time to tumor progression and survival in patients with glioblastoma multiforme of the cerebral hemisphere. Surg Neurol 52:371–379. doi:10.1016/S0090-3019(99)00103-2
Nelson SJ, Nalbandian AB, Proctor E, Vigneron DB (1994) Registration of images from sequential MR studies of the brain. J Magn Reson Imaging 4:877–883. doi:10.1002/jmri.1880040621
Saraswathy S (2006) Semi-automated segmentation of brain tumor lesions in MR images [abstract]. International Society of Magnetic Resonance Imaging, Seattle
Zhang Y, Brady M, Smith S (2001) Segmentation of brain MR images through a hidden Markov random field model and the expectation-maximization algorithm. IEEE Trans Med Imaging 20:45–57. doi:10.1109/42.906424
Chan AA, Nelson SJ (2004) Simplified gamma-variate fitting of perfusion curves. In: Biomedical imaging: macro to nano. IEEE international symposium, vol 2, pp 1067–1070
Johnson G, Wetzel SG, Cha S, Babb J, Tofts PS (2004) Measuring blood volume and vascular transfer constant from dynamic, T(2)*-weighted contrast-enhanced MRI. Magn Reson Med 51:961–968. doi:10.1002/mrm.20049
Lee MC, Cha S, Chang SM, Nelson SJ (2005) Dynamic susceptibility contrast perfusion imaging of radiation effects in normal-appearing brain tissue: changes in the first-pass and recirculation phases. J Magn Reson Imaging 21:683–693. doi:10.1002/jmri.20298
Hartkens T, Rueckert D, Schnabel JA, Hawkes DJ, Hill DLG (2002) VTK CISG registration toolkit: an open source software package for affine and non-rigid registration of single- and multimodal 3D images. [abstract] BVM 2002. Springer-Verlag, Leipzig
Moonen CT, von Kienlin M, van Zijl PC, Cohen J, Gillen J, Daly P et al (1989) Comparison of single-shot localization methods (STEAM and PRESS) for in vivo proton NMR spectroscopy. NMR Biomed 2:201–208. doi:10.1002/nbm.1940020506
Tran TK, Vigneron DB, Sailasuta N, Tropp J, Le Roux P, Kurhanewicz J et al (2000) Very selective suppression pulses for clinical MRSI studies of brain and prostate cancer. Magn Reson Med 43:23–33. doi:10.1002/(SICI)1522-2594(200001)43:1<23::AID-MRM4>3.0.CO;2-E
Star-Lack J, Spielman D, Adalsteinsson E, Kurhanewicz J, Terris DJ, Vigneron DB (1998) In vivo lactate editing with simultaneous detection of choline, creatine, NAA, and lipid singlets at 1.5 T using PRESS excitation with applications to the study of brain and head and neck tumors. J Magn Reson 133:243–254. doi:10.1006/jmre.1998.1458
Nelson SJ (2001) Analysis of volume MRI and MR spectroscopic imaging data for the evaluation of patients with brain tumors. Magn Reson Med 46:228–239. doi:10.1002/mrm.1183
Nelson SJ, Cha S (2003) Imaging glioblastoma multiforme. Cancer J 9:134–145. doi:10.1097/00130404-200303000-00009
McKnight TR, Noworolski SM, Vigneron DB, Nelson SJ (2001) An automated technique for the quantitative assessment of 3D-MRSI data from patients with glioma. J Magn Reson Imaging 13:167–177. doi:10.1002/1522-2586(200102)13:2<167::AID-JMRI1026>3.0.CO;2-K
Catalaa I, Henry R, Dillon WP, Graves EE, McKnight TR, Lu Y et al (2006) Perfusion, diffusion and spectroscopy values in newly diagnosed cerebral gliomas. NMR Biomed 19:463–475. doi:10.1002/nbm.1059
Law M, Yang S, Wang H, Babb JS, Johnson G, Cha S et al (2003) Glioma grading: sensitivity, specificity, and predictive values of perfusion MR imaging and proton MR spectroscopic imaging compared with conventional MR imaging. AJNR Am J Neuroradiol 24:1989–1998
Lupo JM, Cha S, Chang SM, Nelson SJ (2005) Dynamic susceptibility-weighted perfusion imaging of high-grade gliomas: characterization of spatial heterogeneity. AJNR Am J Neuroradiol 26:1446–1454
Cha S, Lupo JM, Chen MH, Lamborn KR, McDermott MW, Berger MS et al (2007) Differentiation of glioblastoma multiforme and single brain metastasis by peak height and percentage of signal intensity recovery derived from dynamic susceptibility-weighted contrast-enhanced perfusion MR imaging. AJNR Am J Neuroradiol 28:1078–1084. doi:10.3174/ajnr.A0484
Lupo JM, Lee MC, Han ET, Cha S, Chang SM, Berger MS et al (2006) Feasibility of dynamic susceptibility contrast perfusion MR imaging at 3T using a standard quadrature head coil and eight-channel phased-array coil with and without SENSE reconstruction. J Magn Reson Imaging 24:520–529. doi:10.1002/jmri.20673
Rana AK, Wardlaw JM, Armitage PA, Bastin ME (2003) Apparent diffusion coefficient (ADC) measurements may be more reliable and reproducible than lesion volume on diffusion-weighted images from patients with acute ischaemic stroke-implications for study design. Magn Reson Imaging 21:617–624. doi:10.1016/S0730-725X(03)00087-0
Sinha S, Bastin ME, Whittle IR, Wardlaw JM (2002) Diffusion tensor MR imaging of high-grade cerebral gliomas. AJNR Am J Neuroradiol 23:520–527
Henkelman RM (1990) Diffusion-weighted MR imaging: a useful adjunct to clinical diagnosis or a scientific curiosity? AJNR Am J Neuroradiol 11:932–934
Le Bihan D, Douek P, Argyropoulou M, Turner R, Patronas N, Fulham M (1993) Diffusion and perfusion magnetic resonance imaging in brain tumors. Top Magn Reson Imaging 5:25–31. doi:10.1097/00002142-199300520-00005
Tien RD, Felsberg GJ, Friedman H, Brown M, MacFall J (1994) MR imaging of high-grade cerebral gliomas: value of diffusion-weighted echoplanar pulse sequences. AJR Am J Roentgenol 162:671–677
Gauvain KM, McKinstry RC, Mukherjee P, Perry A, Neil JJ, Kaufman BA et al (2001) Evaluating pediatric brain tumor cellularity with diffusion-tensor imaging. AJR Am J Roentgenol 177:449–454
Li Y, Osorio JA, Ozturk-Isik E, Chen AP, Xu D, Crane JC et al (2006) Considerations in applying 3D PRESS H-1 brain MRSI with an eight-channel phased-array coil at 3 T. Magn Reson Imaging 24:1295–1302. doi:10.1016/j.mri.2006.07.012
Ozturk E, Banerjee S, Majumdar S, Nelson SJ (2006) Partially parallel MR spectroscopic imaging of gliomas at 3T. Conf Proc IEEE Eng Med Biol Soc 1:493–496
Acknowledgements
This study was supported by UC Discovery grants LSIT01-10107 and ITL-BIO04-10148 funded in conjunction with GE Healthcare, and NIH grants R01 CA059880 and P50 CA97257.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Crawford, F.W., Khayal, I.S., McGue, C. et al. Relationship of pre-surgery metabolic and physiological MR imaging parameters to survival for patients with untreated GBM. J Neurooncol 91, 337–351 (2009). https://doi.org/10.1007/s11060-008-9719-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-008-9719-x