Abstract
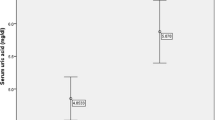
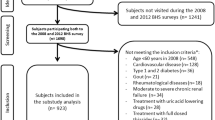
Research on the importance of serum uric acid (SUA) as a contributing metabolic factor to cardiovascular diseases has conducted to conflicting results, with most studies assuming a cross-sectional design. The aim of this study was to evaluate the association of SUA and metabolic syndrome (MetS) and its features. A representative sample of 2,485 individuals aged ≥18 years was randomly selected from the non-institutionalized resident population of Porto, Portugal. A total of 1,054 eligible subjects were included for the longitudinal analyses. Hyperuricemia was defined as SUA ≥70 mg/L in men and ≥60 mg/L in women. MetS was defined according the Joint Interim (2009) criteria. Associations were estimated using Poison regression and binomial models. In the cross-sectional analysis, subjects with hyperuricemia had a 2.10-fold increased risk of MetS as compared with normouricemic subjects (PR = 2.10, 95% CI: 1.68–2.63). Among MetS features, high triglycerides presented the strongest association with hyperuricemia (PR = 2.32, 95% CI: 1.84–2.91). The MetS crude incidence rate was 4.5/100 person-year (95% CI: 3.9–5.2) in normal uricemic and 13.0/100 person-year (95% CI: 8.5–20.0) in hyperuricemic participants. Using a multivariate longitudinal approach, hyperuricemia was positively associated with MetS incidence rate ratios (IRR = 1.73, 95% CI: 1.08–2.76). One standard deviation increase of SUA concentration was associated with a 1.22-fold increase in MetS risk (IRR = 1.22, 95% CI: 1.05–1.42). Elevated SUA presented the strongest association with high-triglycerides concentration (IRR = 1.44, 95%: 1.22–1.71) and waist circumference (IRR = 1.25, 95%: 1.05–1.49). The independent positive association between SUA and MetS suggested by this longitudinal study supports that SUA might be a risk factor for MetS.
Similar content being viewed by others
References
D. Conen, V. Wietlisbach, P. Bovet, C. Shamlaye, W. Riesen, F. Paccaud, M. Burnier, Prevalence of hyperuricemia and relation of serum uric acid with cardiovascular risk factors in a developing country. BMC Public Health 4, 9 (2004). doi:10.1186/1471-2458-4-9
A.C. Gagliardi, M.H. Miname, R.D. Santos, Uric acid: a marker of increased cardiovascular risk. Atherosclerosis 202(1), 11–17 (2009). doi:10.1016/j.atherosclerosis.2008.05.022
T. Montalcini, G. Gorgone, C. Gazzaruso, G. Sesti, F. Perticone, A. Pujia, Relation between serum uric acid and carotid intima-media thickness in healthy postmenopausal women. Intern. Emerg. Med. 2(1), 19–23 (2007). doi:10.1007/s11739-007-0004-3
T. Nakagawa, P. Cirillo, W. Sato, M. Gersch, Y. Sautin, C. Roncal, W. Mu, L.G. Sanchez-Lozada, R.J. Johnson, The conundrum of hyperuricemia, metabolic syndrome, and renal disease. Intern. Emerg. Med. 3(4), 313–318 (2008). doi:10.1007/s11739-008-0141-3
J. Lee, D. Sparrow, P.S. Vokonas, L. Landsberg, S.T. Weiss, Uric acid and coronary heart disease risk: evidence for a role of uric acid in the obesity-insulin resistance syndrome. The Normative Aging Study. Am. J. Epidemiol. 142(3), 288–294 (1995)
W. Rathmann, B. Haastert, A. Icks, G. Giani, J.M. Roseman, Ten-year change in serum uric acid and its relation to changes in other metabolic risk factors in young black and white adults: the CARDIA study. Eur. J. Epidemiol. 22(7), 439–445 (2007). doi:10.1007/s10654-007-9132-3
D.I. Feig, D.H. Kang, R.J. Johnson, Uric acid and cardiovascular risk. N. Engl. J. Med. 359(17), 1811–1821 (2008). doi:10.1056/NEJMra0800885
R.J. Johnson, D.I. Feig, J. Herrera-Acosta, D.H. Kang, Resurrection of uric acid as a causal risk factor in essential hypertension. Hypertension 45(1), 18–20 (2005). doi:10.1161/01.HYP.0000150785.39055.e8
M. Cigolini, G. Targher, M. Tonoli, F. Manara, M. Muggeo, G. De Sandre, Hyperuricaemia: relationships to body fat distribution and other components of the insulin resistance syndrome in 38-year-old healthy men and women. Int. J. Obes. Relat. Metab. Disord. 19(2), 92–96 (1995)
T. Nakagawa, K.R. Tuttle, R.A. Short, R.J. Johnson, Hypothesis: fructose-induced hyperuricemia as a causal mechanism for the epidemic of the metabolic syndrome. Nat Clin Pract Nephrol 1(2), 80–86 (2005). doi:10.1038/ncpneph0019
K. Masuo, H. Kawaguchi, H. Mikami, T. Ogihara, M.L. Tuck, Serum uric acid and plasma norepinephrine concentrations predict subsequent weight gain and blood pressure elevation. Hypertension 42(4), 474–480 (2003)
A. Dehghan, M. van Hoek, E.J. Sijbrands, A. Hofman, J.C. Witteman, High serum uric acid as a novel risk factor for type 2 diabetes. Diabetes Care 31(2), 361–362 (2008). doi:10.2337/dc07-1276
T. Wang, Y. Bi, M. Xu, Y. Huang, Y. Xu, X. Li, W. Wang, G. Ning, Serum uric acid associates with the incidence of type 2 diabetes in a prospective cohort of middle-aged and elderly Chinese. Endocrine 40(1), 109–116 (2011). doi:10.1007/s12020-011-9449-2
M. Alderman, K.J. Aiyer, Uric acid: role in cardiovascular disease and effects of losartan. Curr. Med. Res. Opin. 20(3), 369–379 (2004). doi:10.1185/030079904125002982
M. Schachter, Uric acid and hypertension. Curr. Pharm. Des. 11(32), 4139–4143 (2005)
Y.Y. Sautin, T. Nakagawa, S. Zharikov, R.J. Johnson, Adverse effects of the classic antioxidant uric acid in adipocytes: NADPH oxidase-mediated oxidative/nitrosative stress. Am J Physiol Cell Physiol 293(2), C584–C596 (2007). doi:10.1152/ajpcell.00600.2006
K.J. Cheung, I. Tzameli, P. Pissios, I. Rovira, O. Gavrilova, T. Ohtsubo, Z. Chen, T. Finkel, J.S. Flier, J.M. Friedman, Xanthine oxidoreductase is a regulator of adipogenesis and PPARgamma activity. Cell Metab. 5(2), 115–128 (2007). doi:10.1016/j.cmet.2007.01.005
T. Nakagawa, H. Hu, S. Zharikov, K.R. Tuttle, R.A. Short, O. Glushakova, X. Ouyang, D.I. Feig, E.R. Block, J. Herrera-Acosta, J.M. Patel, R.J. Johnson, A causal role for uric acid in fructose-induced metabolic syndrome. Am J Physiol Renal Physiol 290(3), F625–F631 (2006). doi:10.1152/ajprenal.00140.2005
W.Y. Lin, C.S. Liu, T.C. Li, T. Lin, W. Chen, C.C. Chen, C.I. Li, C.C. Lin, In addition to insulin resistance and obesity, hyperuricemia is strongly associated with metabolic syndrome using different definitions in Chinese populations: a population-based study (Taichung Community Health Study). Ann. Rheum. Dis. 67(3), 432–433 (2008). doi:10.1136/ard.2007.073601
N. Ishizaka, Y. Ishizaka, E. Toda, R. Nagai, M. Yamakado, Association between serum uric acid, metabolic syndrome, and carotid atherosclerosis in Japanese individuals. Arterioscler. Thromb. Vasc. Biol. 25(5), 1038–1044 (2005). doi:10.1161/01.ATV.0000161274.87407.26
T. Wilsgaard, B.K. Jacobsen, Lifestyle factors and incident metabolic syndrome. The Tromso Study 1979–2001. Diabetes Res. Clin. Pract. 78(2), 217–224 (2007). doi:10.1016/j.diabres.2007.03.006
S. Ryu, J. Song, B.Y. Choi, S.J. Lee, W.S. Kim, Y. Chang, D.I. Kim, B.S. Suh, K.C. Sung, Incidence and risk factors for metabolic syndrome in Korean male workers, ages 30 to 39. Ann. Epidemiol. 17(4), 245–252 (2007). doi:10.1016/j.annepidem.2006.10.001
J. Tong, E.J. Boyko, K.M. Utzschneider, M.J. McNeely, T. Hayashi, D.B. Carr, T.M. Wallace, S. Zraika, F. Gerchman, D.L. Leonetti, W.Y. Fujimoto, S.E. Kahn, Intra-abdominal fat accumulation predicts the development of the metabolic syndrome in non-diabetic Japanese-Americans. Diabetologia 50(6), 1156–1160 (2007). doi:10.1007/s00125-007-0651-y
A.C. Santos, M. Severo, H. Barros, Incidence and risk factors for the metabolic syndrome in an urban South European population. Prev. Med. 50(3), 99–105 (2010). doi:10.1016/j.ypmed.2009.11.011
E. Ramos, C. Lopes, H. Barros, Investigating the effect of nonparticipation using a population-based case-control study on myocardial infarction. Ann. Epidemiol. 14(6), 437–441 (2004). doi:10.1016/j.annepidem.2003.09.013
Organization, W.H.: Guidelines for ATC classification. Collaborating centre for drug statistics methodology. Nordic Council on Medicines. WHO. (1990)
M.C. Horchberg, J.S. Smolen et al., Rheumatology, 3rd edn. (Mosby, New York, 2003)
C. Lopes, A. Aro, A. Azevedo, E. Ramos, H. Barros, Intake and adipose tissue composition of fatty acids and risk of myocardial infarction in a male Portuguese community sample. J. Am. Diet. Assoc. 107(2), 276–286 (2007). doi:10.1016/j.jada.2006.11.008
Lopes, C.: Reproducibility and validity of a semi-quantitative food frequency questionnaire. In Diet and acute myocardial infarction: a population based case-control study. PhD thesis. University of Porto, Porto, 79–115 2000
M. Camoes, M. Severo, A.C. Santos, H. Barros, C. Lopes, Testing an adaptation of the EPIC physical activity questionnaire in Portuguese adults: a validation study that assesses the seasonal bias of self-report. Ann. Hum. Biol. 37(2), 185–197 (2010). doi:10.3109/03014460903341836
S.M. Grundy, J.I. Cleeman, S.R. Daniels, K.A. Donato, R.H. Eckel, B.A. Franklin, D.J. Gordon, R.M. Krauss, P.J. Savage, S.C. Smith Jr, J.A. Spertus, F. Costa, Diagnosis and management of the metabolic syndrome: an American Heart Association/National Heart, Lung, and Blood Institute Scientific Statement. Circulation 112(17), 2735–2752 (2005). doi:10.1161/CIRCULATIONAHA.105.169404
K.G. Alberti, R.H. Eckel, S.M. Grundy, P.Z. Zimmet, J.I. Cleeman, K.A. Donato, J.C. Fruchart, W.P. James, C.M. Loria, S.C. Smith Jr, Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation 120(16), 1640–1645 (2009). doi:10.1161/CIRCULATIONAHA.109.192644
L.Y. Chen, W.H. Zhu, Z.W. Chen, H.L. Dai, J.J. Ren, J.H. Chen, L.Q. Chen, L.Z. Fang, Relationship between hyperuricemia and metabolic syndrome. J Zhejiang Univ Sci B 8(8), 593–598 (2007). doi:10.1631/jzus.2007.B0593
W. Rathmann, E. Funkhouser, A.R. Dyer, J.M. Roseman, Relations of hyperuricemia with the various components of the insulin resistance syndrome in young black and white adults: the CARDIA study. Coronary Artery Risk Development in Young Adults. Ann. Epidemiol. 8(4), 250–261 (1998)
H. Vuorinen-Markkola, H. Yki-Jarvinen, Hyperuricemia and insulin resistance. J. Clin. Endocrinol. Metab. 78(1), 25–29 (1994)
C. Russo, O. Olivieri, D. Girelli, P. Guarini, R. Corrocher, Relationships between serum uric acid and lipids in healthy subjects. Prev. Med. 25(5), 611–616 (1996). doi:10.1006/pmed.1996.0096
Y. Moriwaki, T. Yamamoto, S. Takahashi, Z. Tsutsumi, K. Higashino, Apolipoprotein E phenotypes in patients with gout: relation with hypertriglyceridaemia. Ann. Rheum. Dis. 54(5), 351–354 (1995)
F. Cardona, F.J. Tinahones, E. Collantes, A. Escudero, E. Garcia-Fuentes, F.J. Soriguer, The elevated prevalence of apolipoprotein E2 in patients with gout is associated with reduced renal excretion of urates. Rheumatology (Oxford) 42(3), 468–472 (2003)
J.M. Boer, C. Ehnholm, H.J. Menzel, L.M. Havekes, M. Rosseneu, D.S. O’Reilly, L. Tiret, Interactions between lifestyle-related factors and the ApoE polymorphism on plasma lipids and apolipoproteins. The EARS Study. European Atherosclerosis Research Study. Arterioscler. Thromb. Vasc. Biol. 17(9), 1675–1681 (1997)
A. Bedir, M. Topbas, F. Tanyeri, M. Alvur, N. Arik, Leptin might be a regulator of serum uric acid concentrations in humans. Jpn. Heart J. 44(4), 527–536 (2003)
O. Bosello, M. Zamboni, Visceral obesity and metabolic syndrome. Obes. Rev. 1(1), 47–56 (2000)
J.P. Forman, H. Choi, G.C. Curhan, Plasma uric acid level and risk for incident hypertension among men. J. Am. Soc. Nephrol. 18(1), 287–292 (2007). doi:10.1681/ASN.2006080865
P.B. Mellen, A.J. Bleyer, T.P. Erlinger, G.W. Evans, F.J. Nieto, L.E. Wagenknecht, M.R. Wofford, D.M. Herrington, Serum uric acid predicts incident hypertension in a biethnic cohort: the atherosclerosis risk in communities study. Hypertension 48(6), 1037–1042 (2006). doi:10.1161/01.HYP.0000249768.26560.66
A. Shankar, R. Klein, B.E. Klein, F.J. Nieto, The association between serum uric acid level and long-term incidence of hypertension: population-based cohort study. J. Hum. Hypertens. 20(12), 937–945 (2006). doi:10.1038/sj.jhh.1002095
J. Lee, Odds ratio or relative risk for cross-sectional data? Int. J. Epidemiol. 23(1), 201–203 (1994)
O. Axelson, M. Fredriksson, K. Ekberg, Use of the prevalence ratio v the prevalence odds ratio in view of confounding in cross sectional studies. Occup. Environ. Med. 52(7), 494 (1995)
P. Neil, Effect measures in prevalence studies. Environ. Health Perspect. 112(10), 1047–1050 (2004)
M.L. Thompson, J.E. Myers, D. Kriebel, Prevalence odds ratio or prevalence ratio in the analysis of cross sectional data: what is to be done? Occup. Environ. Med. 55(4), 272–277 (1998)
Acknowledgment
This study used information from the EPIPorto cohort study, funded by the Fundacão para a Ciência e a Tecnologia (FCT, Portugal): POCTI/ESP/42361/2001; POCTI/SAU-ESP/61160/2004; PTDC/SAU-ESA/108315/2008.
Conflicts of interest
We don’t have any financial interests or affiliations with institutions, organizations, or companies that are mentioned in the manuscript or whose products or services are discussed.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gonçalves, J.P., Oliveira, A., Severo, M. et al. Cross-sectional and longitudinal associations between serum uric acid and metabolic syndrome. Endocrine 41, 450–457 (2012). https://doi.org/10.1007/s12020-012-9629-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-012-9629-8