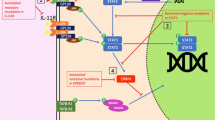
Abstract
FOXP3 is the key mediator of regulatory T-cell development in the thymus. Naturally occurring mutations of FOXP3 interfere with this process, resulting in the generation of autoaggressive lymphocyte clones that are directly responsible for the syndrome Immune Dysregulation, Polyendocrinopathy, Enteropathy, X-Linked (IPEX) in humans and scurfy in mice. Stem cell transplantation is the only cure for IPEX patients. The study of this rare disease has provided important insight into the mechanisms of immunosuppression, autoimmunity and tolerance and future studies may lead to novel strategies to treat not only patients with IPEX, but also those suffering from autoimmunity, graft-versus-host disease or cancer.


Similar content being viewed by others
References
Puck JM, Rieux-Laucat F, Le Deist F, Straus SE. Autoimmune lymphoproliferative syndrome. In: Ochs HD, Smith CIE, Puck JM, editors. Primary immunodeficiency diseases, a molecular and genetic approach. 2nd ed. New York: Oxford University Press; 2007. p. 326–341
Peltonen-Palotie L, Halonen M, Perheentupa J. Autoimmune polyendocrinopathy, candidiasis, ectodermal dystrophy. In: Ochs HD, Smith CIE, Puck JM, editors. Primary immunodeficiency diseases, a molecular and genetic approach. 2nd ed. New York: Oxford University Press; 2007. p. 342–354
Chatila TA, Blaeser F, Ho N, et al. JM2, encoding a fork head-related protein, is mutated in X-linked autoimmunity-allergic disregulation syndrome. J Clin Invest 2000;106(12):R75–81
Bennett CL, Christie J, Ramsdell F, et al. The immune dysregulation, polyendocrinopathy, enteropathy, X-linked syndrome (IPEX) is caused by mutations of FOXP3. Nat Genet 2001;27(1):20–1
Wildin RS, Ramsdell F, Peake J, et al. X-linked neonatal diabetes mellitus, enteropathy and endocrinopathy syndrome is the human equivalent of mouse scurfy. Nat Genet 2001;27(1):18–20
Bennett CL, Yoshioka R, Kiyosawa H, et al. X-Linked syndrome of polyendocrinopathy, immune dysfunction, and diarrhea maps to Xp11.23-Xq13.3. Am J Hum Genet 2000;66(2):461–8
Ferguson PJ, Blanton SH, Saulsbury FT, et al. Manifestations and linkage analysis in X-linked autoimmunity-immunodeficiency syndrome. Am J Med Genet 2000;90(5):390–7
Levy-Lahad E, Wildin RS Neonatal diabetes mellitus, enteropathy, thrombocytopenia, and endocrinopathy: Further evidence for an X-linked lethal syndrome. J Pediatr 2001;138(4):577–80
Wildin RS, Smyk-Pearson S, Filipovich AH Clinical and molecular features of the immunodysregulation, polyendocrinopathy, enteropathy, X linked (IPEX) syndrome. J Med Genet 2002;39(8):537–45
Powell BR, Buist NR, Stenzel P An X-linked syndrome of diarrhea, polyendocrinopathy, and fatal infection in infancy. J Pediatr 1982;100(5):731–7
Nieves DS, Phipps RP, Pollock SJ, et al. Dermatologic and immunologic findings in the immune dysregulation, polyendocrinopathy, enteropathy, X-linked syndrome. Arch Dermatol 2004;140(4):466–72
Savage MO, Mirakian R, Harries JT, Bottazzo GF Could protracted diarrhoea of infancy have an autoimmune pathogenesis? Lancet 1982;1(8278):966–7
Kobayashi I, Imamura K, Yamada M, et al. A 75-kD autoantigen recognized by sera from patients with X-linked autoimmune enteropathy associated with nephropathy. Clin Exp Immunol 1998;111(3):527–31
Gambineri E, Torgerson TR, Ochs HD Immune dysregulation, polyendocrinopathy, enteropathy, and X-linked inheritance (IPEX), a syndrome of systemic autoimmunity caused by mutations of FOXP3, a critical regulator of T-cell homeostasis. Curr Opin Rheumatol 2003;15(4):430–5
McGinness JL, Bivens MM, Greer KE, Patterson JW, Saulsbury FT Immune dysregulation, polyendocrinopathy, enteropathy, X-linked syndrome (IPEX) associated with pemphigoid nodularis: a case report and review of the literature. J Am Acad Dermatol 2006;55(1):143–8
Baud O, Goulet O, Canioni D, et al. Treatment of the immune dysregulation, polyendocrinopathy, enteropathy, X-linked syndrome (IPEX) by allogeneic bone marrow transplantation. N Engl J Med 2001;344(23):1758–62
Cilio CM, Bosco S, Moretti C, et al. Congenital autoimmune diabetes mellitus. N Engl J Med 2000;342(20):1529–31
Kobayashi I, Imamura K, Kubota M, et al. Identification of an autoimmune enteropathy-related 75-kilodalton antigen. Gastroenterology 1999;117(4):823–30
Bennett CL, Brunkow ME, Ramsdell F, et al. A rare polyadenylation signal mutation of the FOXP3 gene (AAUAAA–>AAUGAA) leads to the IPEX syndrome. Immunogenetics 2001;53(6):435–9
Roncador G, Brown PJ, Maestre L, et al. Analysis of FOXP3 protein expression in human CD4+CD25+ regulatory T cells at the single-cell level. Eur J Immunol 2005;35(6):1681–91
Gajiwala KS, Burley SK Winged helix proteins. Curr Opin Struct Biol 2000;10(1):110–6
Carlsson P, Mahlapuu M Forkhead transcription factors: key players in development and metabolism. Dev Biol 2002;250(1):1–23
Lopes JE, Torgerson TR, Schubert LA, et al. Analysis of FOXP3 reveals multiple domains required for its function as a transcriptional repressor. J Immunol 2006;177(5):3133–42
Schubert LA, Jeffery E, Zhang Y, Ramsdell F, Ziegler SF Scurfin (FOXP3) acts as a repressor of transcription and regulates T cell activation. J Biol Chem 2001;276(40):37672–9
Wu Y, Borde M, Heissmeyer V, et al. FOXP3 controls regulatory T cell function through cooperation with NFAT. Cell 2006;126(2):375–87
Bettelli E, Dastrange M, Oukka M Foxp3 interacts with nuclear factor of activated T cells and NF-kappa B to repress cytokine gene expression and effector functions of T helper cells. Proc Natl Acad Sci U S A 2005;102(14):5138–43
Khattri R, Kasprowicz D, Cox T, et al. The amount of scurfin protein determines peripheral T cell number and responsiveness. J Immunol 2001;167(11):6312–20
Sakaguchi S, Takahashi T, Yamazaki S, et al. Immunologic self tolerance maintained by T-cell-mediated control of self-reactive T cells: implications for autoimmunity and tumor immunity. Microbes Infect 2001;3(11):911–8
Itoh M, Takahashi T, Sakaguchi N, et al. Thymus and autoimmunity: production of CD25+CD4+ naturally anergic and suppressive T cells as a key function of the thymus in maintaining immunologic self-tolerance. J Immunol 1999;162(9):5317–26
Shevach EM CD4+CD25+ suppressor T cells: more questions than answers. Nat Rev Immunol 2002;2(6):389–400
Bacchetta R, Gregori S, Roncarolo MG CD4+ regulatory T cells: mechanisms of induction and effector function. Autoimmun Rev 2005;4(8):491–6
Randolph DA, Fathman CG Cd4+Cd25+ regulatory T cells and their therapeutic potential. Annu Rev Med 2006;57:381–402
Fontenot JD, Gavin MA, Rudensky AY Foxp3 programs the development and function of CD4+CD25+ regulatory T cells. Nat Immunol 2003;4(4):330–6
Hori S, Nomura T, Sakaguchi S Control of regulatory T cell development by the transcription factor Foxp3. Science 2003;299(5609):1057–61
Khattri R, Cox T, Yasayko SA, Ramsdell F An essential role for Scurfin in CD4+CD25+ T regulatory cells. Nat Immunol 2003;4(4):337–42
Yagi H, Nomura T, Nakamura K, et al. Crucial role of FOXP3 in the development and function of human CD25+CD4+ regulatory T cells. Int Immunol 2004;16(11):1643–56
Fontenot JD, Rudensky AY A well adapted regulatory contrivance: regulatory T cell development and the forkhead family transcription factor Foxp3. Nat Immunol 2005;6(4):331–7
Wood KJ, Sakaguchi S Regulatory T cells in transplantation tolerance. Nat Rev Immunol 2003;3(3):199–210
Zorn E, Kim HT, Lee SJ, et al. Reduced frequency of FOXP3+ CD4+CD25+ regulatory T cells in patients with chronic graft-versus-host disease. Blood 2005;106(8):2903–11
Wei WZ, Morris GP, Kong YC Anti-tumor immunity and autoimmunity: a balancing act of regulatory T cells. Cancer Immunol Immunother 2004;53(2):73–8
Loser K, Hansen W, Apelt J, Balkow S, Buer J, Beissert S In vitro-generated regulatory T cells induced by Foxp3-retrovirus infection control murine contact allergy and systemic autoimmunity. Gene Ther 2005;12(17):1294–304
Marinaki S, Neumann I, Kalsch AI, et al. Abnormalities of CD4 T cell subpopulations in ANCA-associated vasculitis. Clin Exp Immunol 2005;140(1):181–91
Yu P, Gregg RK, Bell JJ, et al. Specific T regulatory cells display broad suppressive functions against experimental allergic encephalomyelitis upon activation with cognate antigen. J Immunol 2005;174(11):6772–80
Dubois B, Chapat L, Goubier A, Papiernik M, Nicolas JF, Kaiserlian D Innate CD4+CD25+ regulatory T cells are required for oral tolerance and inhibition of CD8+ T cells mediating skin inflammation. Blood 2003;102(9):3295–301
Thorstenson KM, Khoruts A Generation of anergic and potentially immunoregulatory CD25+CD4 T cells in vivo after induction of peripheral tolerance with intravenous or oral antigen. J Immunol 2001;167(1):188–95
Hauet-Broere F, Unger WW, Garssen J, Hoijer MA, Kraal G, Samsom JN Functional CD25- and CD25+ mucosal regulatory T cells are induced in gut-draining lymphoid tissue within 48 h after oral antigen application. Eur J Immunol 2003;33(10):2801–10
Zhang X, Izikson L, Liu L, Weiner HL Activation of CD25(+)CD4(+) regulatory T cells by oral antigen administration. J Immunol 2001;167(8):4245–53
Karube K, Ohshima K, Tsuchiya T, et al. Expression of FoxP3, a key molecule in CD4CD25 regulatory T cells, in adult T-cell leukaemia/lymphoma cells. Br J Haematol 2004;126(1):81–4
Viguier M, Lemaitre F, Verola O, et al. Foxp3 expressing CD4+CD25(high) regulatory T cells are overrepresented in human metastatic melanoma lymph nodes and inhibit the function of infiltrating T cells. J Immunol 2004;173(2):1444–53
Berger CL, Tigelaar R, Cohen J, et al. Cutaneous T-cell lymphoma: malignant proliferation of T-regulatory cells. Blood 2005;105(4):1640–7
Ormandy LA, Hillemann T, Wedemeyer H, Manns MP, Greten TF, Korangy F Increased populations of regulatory T cells in peripheral blood of patients with hepatocellular carcinoma. Cancer Res 2005;65(6):2457–64
Unitt E, Rushbrook SM, Marshall A, et al. Compromised lymphocytes infiltrate hepatocellular carcinoma: the role of T-regulatory cells. Hepatology 2005;41(4):722–30
Beyer M, Kochanek M, Darabi K, et al. Reduced frequencies and suppressive function of CD4+CD25hi regulatory T cells in patients with chronic lymphocytic leukemia after therapy with fludarabine. Blood 2005;106(6):2018–25
Sharma S, Yang SC, Zhu L, et al. Tumor cyclooxygenase-2/prostaglandin E2-dependent promotion of FOXP3 expression and CD4+ CD25+ T regulatory cell activities in lung cancer. Cancer Res 2005;65(12):5211–20
Brunkow ME, Jeffery EW, Hjerrild KA, et al. Disruption of a new forkhead/winged-helix protein, scurfin, results in the fatal lymphoproliferative disorder of the scurfy mouse. Nat Genet 2001;27(1):68–73
Bindl L, Torgerson T, Perroni L, et al. Successful use of the new immune-suppressor sirolimus in IPEX (immune dysregulation, polyendocrinopathy, enteropathy, X-linked syndrome). J Pediatr 2005;147(2):256–9
Mazzolari E, Forino C, Fontana M, et al. A new case of IPEX receiving bone marrow transplantation. Bone Marrow Transplant 2005;35(10):1033–4
Rao A, Kamani N, Filipovich A, et al. Successful bone marrow transplantation for IPEX syndrome after reduced intensity conditioning. Blood 2007;109(1):383–5
Acknowledgment
Supported by National Institutes of Health Grant HD17427 and grants from the Immunodeficiency Foundation and the Jeffrey Modell Foundation (H.D.O.) and Pfizer post-doctoral fellowship and Child Health Research Center Grant HD043376-03 (T.R.T.)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ochs, H.D., Gambineri, E. & Torgerson, T.R. IPEX, FOXP3 and regulatory T-cells: a model for autoimmunity. Immunol Res 38, 112–121 (2007). https://doi.org/10.1007/s12026-007-0022-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12026-007-0022-2