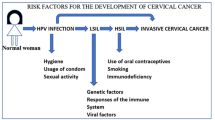
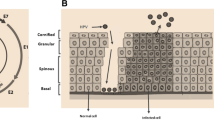
Abstract
Cervical cancer is a virus-induced disease that is caused by the integration of high-risk infecting human papillomaviruses (HPV) in the host genome. For this reason, the carcinogenesis process of cervical cancer is associated to the expression of the viral oncogenic proteins E6 and E7. These proteins are capable of inactivating p53 and pRb, which induces a continuous cell proliferation with the increasing risk of accumulation of DNA damage that eventually leads to cancer. Moreover, cervical cancer can be prevented by prophylactic HPV vaccines; their molecular characteristics and mechanism of action are reviewed. Ultimately, new molecular targets for cervical cancer like proteasome, the EGFR family and IGF family are exposed.
Similar content being viewed by others
References
WHO (2006) World Health Organization Report: Comprehensive cervical cancer control: a guide to essential practice. Available on WWW at www.who.int/reproductive-health/publications/cervical_cancer_gep/index.htm
Parkin M, Bray F (2006) The burden of HPV-related cancers. Vaccine 24[Suppl 3]:S11–S25
Clifford GM, Smith JS, Plummer M et al (2003) Human papillomavirus types in invasive cervical cancer worldwide: a meta-analysis. Br J Cancer 88:63–73
Persson G, Andersson K, Krantz I (1996) Symptomatic genital papillomavirus infection in a community. Incidence and clinical picture. Acta Obstet Gynecol Scand 75:287–290
Woodman CB, Collins S, Winter H et al (2001) Natural history of cervical human papillomavirus infection in young women: a longitudinal cohort study. Lancet 357:1831–1836
Richardson H, Kelsall G, Tellier P et al (2003) The natural history of type-specific human papillomavirus infections in female university students. Cancer Epidemiol Biomarkers Prev 12:485–490
Clifford GM, Rana RK, Franceschi S et al (2005) Human papillomavirus genotype distribution in low-grade cervical lesions: comparison by geographic region and with cervical cancer. Cancer Epidemiol Biomarkers Prev 14:1157–1164
Parkin DM, Bray F, Ferlay J, Pisani P (2005) Global cancer statistics, 2002. CA Cancer J Clin 55:74–108
zur Hausen H (2002) Papillomaviruses and cancer: from basic studies to clinical application. Nat Rev Cancer 2:342–350
Baker CC, Phelps WC, Lindgren V et al (1987) Structural and transcriptional analysis of human papillomavirus type 16 sequences in cervical carcinoma cell lines. J Virol 61:962–967
Shafti-Keramat S, Handisurya A, Kriehuber E et al (2003) Different heparan sulfate proteoglycans serve as cellular receptors for human papillomaviruses. J Virol 77:13125–13135
Summerford C, Bartlett JS, Samulski RJ (1999) ?-5-integrin: a co-receptor for adeno-associated virus type 2 infection. Nat Med 5:78–82
Culp TD, Christensen ND (2004) Kinetics of in vitro adsorption and entry of papillomavirus virions. Virology 319:152–161
Bousarghin L, Touze A, Sizaret PY, Coursaget P (2003) Human papillomavirus types 16, 31, and 58 use different endocytosis pathways to enter cells. J Virol 77:3846–3850
You J, Croyle JL, Nishimura A et al (2004) Interaction of the bovine papillomavirus E2 protein with Brd4 tethers the viral DNA to host mitotic chromosomes. Cell 117:349–360
Brehm A, Nielsen SJ, Miska EA et al (1999) The E7 oncoprotein associates with Mi2 and histone deacetylase activity to promote cell growth. EMBO J 18:2449–2458
Antinore MJ, Birrer MJ, Patel D et al (1996) The human papillomavirus type 16 E7 gene product interacts with and trans-activates the AP1 family of transcription factors. EMBO J 15:1950–1960
Funk JO, Waga S, Harry JB et al (1997) Inhibition of CDK activity and PCNA-dependent DNA replication by p21 is blocked by interaction with the HPV16 E7 oncoprotein. Genes Dev 11:2090–2100
Thomas M, Pim D, Banks L (1999) The role of the E6-p53 interaction in the molecular pathogenesis of HPV. Oncogene 18:7690–7700
Thomas M, Banks L (1998) Inhibition of Bakinduced apoptosis by HPV-18 E6. Oncogene 17:2943–2954
Li B, Dou QP (2000) Bax degradation by the ubiquitin/proteasome-dependent pathway: involvement in tumor survival and progression. Proc Natl Acad Sci USA 97:3850–3855
Munger K, Baldwin A, Edwards KM et al (2004) Mechanism of human papillomavirus-induced oncogenesis. J Virol 78:11451–11460
Middleton K, Peh W, Southern SA et al (2003) Organisation of the human papillomavirus productive cycle during neoplastic progression provides a basis for the selection of diagnostic markers. J Virol 77:10186–10201
Florin L, Sapp C, Streeck RE, Sapp M (2002) Assembly and translocation of papillomavirus capsid proteins. J Virol 76:10009–10014
Day PM, Roden RBS, Lowy DR, Schiller JT (1998) The papillomavirus minor capsid protein, L2, induces localization of the major capsid protein, L1 and the viral transcription/replication protein, E2, to PML oncogenic domains. J Virol 72:142–150
Moscicki AB, Schiffman M, Kjaer S, Villa L (2006) Updating the natural history of HPV anogenital cancer. Vaccine 24[Suppl 3]:S42–51
Fujii T, Masumoto N, Saito M et al (2005) Comparison between in situ hybridization and real-time PCR technique as a means of detecting the integrated form of human papillomavirus 16 in cervical neoplasia. Diagn Mol Pathol 14:103–108
Yu T, Ferber MJ, Cheung TH et al (2005) The role of viral integration in the development of cervical cancer. Cancer Genet Cytogenet 158: 27–34
Frattini MG, Hurst SD, Lim HB et al (1997) Abrogation of a mitotic checkpoint by E2 proteins from oncogenic human papillomaviruses correlates with increased turnover of the p53 tumour suppressor protein. EMBO J 16:318–331
Duensing S, Munger K (2002) The human papillomavirus type 16 E6 and E7 oncoproteins independently induce numerical and structural chromosome instability. Cancer Res 62:7075–7082
Duensing S, Duensing A, Flores ER et al (2001) Centrosome abnormalities and genomic instability by episomal expression of human papillomavirus type 16 in raft cultures of human keratinocytes. J Virol 75:7712–7716
Schaeffer AJ, Nguyen M, Liem A et al (2004) E6 and E7 oncoproteins induce distinct patterns of chromosomal aneuploidy in skin tumors from transgenic mice. Cancer Res 64:538–546
Duensing S, Munger K (2004) Mechanisms of genomic instability in human cancer: insights from studies with human papillomavirus oncoproteins. Int J Cancer 109:157–162
Helt AM, Galloway DA (2003) Mechanisms by which DNA tumor virus oncoproteins target the Rb family of pocket proteins. Carcinogenesis 24:159–169
Klingelhutz AJ, Foster SA, McDougall JK (1996) Telomerase activation by the E6 gene product of human papillomavirus type 16. Nature 380:79–82
Hellberg D, Stendahl U (2005) The biological role of smoking, oral contraceptive use and endogenous sexual steroid hormones in invasive squamous epithelial cervical cancer. Anticancer Res 25:3041–3046
Haverkos HW (2003) Viruses, chemicals and cocarcinogenesis. Oncogene 23:6492–6499
Moodley J (2004) Combined oral contraceptives and cervical cancer. Curr Opin Obstet Gynecol 16:27–29
Muñoz N, Catellsague X, Berrington A, Gissmann L (2006) HPV in the etiology of human cancer. Vaccine 24[Suppl 3]:S1–S10
Dueñas-González A, Lizano M, Candelaria M et al (2005) Epigenetics of cervical cancer. An overview and therapeutic perspectives. Mol Cancer 4:38
Zambrano P, Segura-Pacheco B, Pérez-Cárdenas E et al (2005) A phase I study of hydralazine to demethylate and reactivate the expression of tumor suppressor genes. BMC Cancer 5:44
Stanley M, Lowy DR, Frazer I (2006) Prophylactic HPV vaccines: underlying mechanisms. Vaccine 24[Suppl 3]:S106–S113
Harper DM, Franco EL, Wheeler CM et al (2006) Sustained efficacy up to 4, 5 years of a bivalent L1 virus-like particle vaccine against human papillomavirus types 16 and 18: follow-up from a randomised control trial. Lancet 367:1247–1255
Villa LL, Costa RL, Petta CA et al (2005) Prophylactic quadrivalent human papillomavirus (types 6, 11, 16, and 18) L1 virus-like particle vaccine in young woman: a randomised double-blind placebo-controlled multicentre phase II efficacy trial. Lancet Oncol 6:271–278
Giroglou T, Sapp M, Lane C et al (2001) Immunological analyses of human papillomavirus capsids. Vaccine 19:1783–1793
Fleury MJ, Touze A, Alvarez E et al (2006) Identification of type-specific and cross-reactive neutralizing conformational epitopes on the major capsid protein of human papillomavirus type 31. Arch Virol 151:1511–1523
Combita AL, Touze A, Bousarghin L et al (2002) Identification of two cross-neutralizing linear epitopes within the L1 major capsid protein of human papillomaviruses. J Virol 76:6480–6486
Adams J (2004) The proteasome: a suitable antineoplastic target. Nat Rev Cancer 4:349–360
Sunwoo JB, Chen Z, Dong G et al (2001) Novel proteasome inhibitor PS-341 inhibits activation of nuclear factor-kappa B, cell survival, tumor growth, and angiogenesis in squamous cell carcinoma. Clin Cancer Res 7:1419–1428
Russo SM, Tepper JE, Baldwin AS Jr et al (2001) Enhancement of radiosensitivity by proteasome inhibition: implications for a role of NF-kB. Int J Radiat Oncol Biol Phys 50:183–193
Mathur SP, Mathur RS, Young RC (2000) Cervical epidermal growth factor-receptor (EGF-R) and serum insulin-like growth factor II (IGF-II) levels are potential markers for cervical cancer. Am J Reprod Immunol 44:222–230
Kersemaekers AM, Fleuren GJ, Kenter GG et al (1999) Oncogene alterations in carcinomas of the uterine cervix: overexpression of the epidermal growth factor receptor is associated with poor prognosis. Clin Cancer Res 5:577–586
Vergote I, Humblet Y, van Cutsem E et al (2005) A multicenter Phase II trial of gefitinib 500 mg/day in 193 patients with advanced epidermal growth factor receptor-positive solid tumors who had failed previous chemotherapy: interim data. J Clin Oncol (2005) ASCO Annual Meeting Proceedings) 23[June 1 Supplement]:3162
Ferreira GG, Salgado M, Lima R et al (2006) A phase I trial of erlotinib (E), cisplatin (C) and radiotherapy (RT) for patients with locally advanced squamous cell cervical cancer. J Clin Oncol (2006 ASCO Annual Meeting Proceedings Part I) 24[June 20 Supplement]:13081
LeRoith D, Roberts CT Jr (2003) The insulin-like growth factor system and cancer. Cancer Lett 195:127–137
Steller MA, Delgado CH, Bartels CJ et al (1996) Overexpression of the insulin-like growth factor-1 receptor and autocrine stimulation in human cervical cancer cells. Cancer Res 56:1761–1765
Mathur SP, Mathur RS, Underwood PB et al (2004) Circulating levels of insulin-like growth factor-II and IGF-binding protein 3 in cervical cancer. Gynecol Oncol 91:486–493
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by an unrestricted educational grant from Sanofi-Aventis.
Rights and permissions
About this article
Cite this article
González Martín, A. Molecular biology of cervical cancer. Clin Transl Oncol 9, 347–354 (2007). https://doi.org/10.1007/s12094-007-0066-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-007-0066-8