Abstract
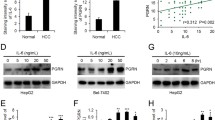
Overproduction of hepcidin by interleukin-6 (IL-6) is considered to be the main factor responsible for the development of anemia in inflammatory conditions. Since oncostatin M (OSM), a member of the IL-6 family, plays an important role in immune and inflammatory responses, we assessed the effect of OSM on hepcidin expression, as well as that of leukemia inhibitory factor (LIF), another member of the IL-6 family. We found that hepcidin expression was markedly induced by OSM and LIF in a time- and dose-dependent manner in hepatoma cell lines, and this expression was induced independent of IL-6/IL-6 receptor signaling. Luciferase assay revealed that OSM and LIF stimulated a −1.3-kb hepcidin promoter. This effect was markedly reduced when the signal transducer and activator of transcription (STAT) site of the promoter was mutated, and was almost completely abolished in the presence of AG-490, a Janus kinase (JAK) inhibitor. Hence, the JAK/STAT pathway plays a major role in OSM- and LIF-induced activation of the hepcidin promoter. In conclusion, we demonstrated that OSM and LIF can induce hepcidin expression mainly through the JAK/STAT pathways. Further studies are warranted to evaluate the clinical significance of OSM and LIF in the development of anemia in various inflammatory diseases.






Similar content being viewed by others
References
Krause A, Neitz S, Magert HJ, Schulz A, Forssmann WG, Schulz-Knappe P, et al. LEAP-1, a novel highly disulfide-bonded human peptide, exhibits antimicrobial activity. FEBS Lett. 2000;480:147–50.
Park CH, Valore EV, Waring AJ, Ganz T. Hepcidin, a urinary antimicrobial peptide synthesized in the liver. J Biol Chem. 2001;276:7806–10.
Andrews NC. Forging a field: the golden age of iron biology. Blood. 2008;112:219–30.
Kohgo Y, Ikuta K, Ohtake T, Torimoto Y, Kato J. Body iron metabolism and pathophysiology of iron overload. Int J Hematol. 2008;88:7–15.
Sow FB, Florence WC, Satoskar AR, Schlesinger LS, Zwilling BS, Lafuse WP. Expression and localization of hepcidin in macrophages: a role in host defense against tuberculosis. J Leukoc Biol. 2007;82:934–45.
Nemeth E, Tuttle MS, Powelson J, Vaughn MB, Donovan A, Ward DM, et al. Hepcidin regulates cellular iron efflux by binding to ferroportin and inducing its internalization. Science. 2004;306:2090–3.
Ganz T. Hepcidin: a regulator of intestinal iron absorption and iron recycling by macrophages. Best Pract Res Clin Haematol. 2005;18:171–82.
Pigeon C, Ilyin G, Courselaud B, Leroyer P, Turlin B, Brissot P, et al. A new mouse liver-specific gene, encoding a protein homologous to human antimicrobial peptide hepcidin, is overexpressed during iron overload. J Biol Chem. 2001;276:7811–9.
Nemeth E, Rivera S, Gabayan V, Keller C, Taudorf S, Pedersen BK, et al. IL-6 mediates hypoferremia of inflammation by inducing the synthesis of the iron-regulatory hormone hepcidin. J Clin Invest. 2004;113:1271–6.
Beutler E. Iron storage disease: facts, fiction and progress. Blood Cells Mol Dis. 2007;39:140–7.
Anderson GJ, Powell LW. HFE and non-HFE hemochromatosis. Int J Hematol. 2002;76:203–7.
Lee P, Peng H, Gelbart T, Wang L, Beutler E. Regulation of hepcidin transcription by interleukin-1 and interleukin-6. Proc Natl Acad Sci USA. 2005;102:1906–10.
Inamura J, Ikuta K, Jimbo J, Shindo M, Sato K, Torimoto Y, et al. Upregulation of hepcidin by interleukin-1 beta in human hepatoma cell lines. Hepatol Res. 2005;33:198–205.
Collins HL. Withholding iron as a cellular defence mechanism: friend or foe? Eur J Immunol. 2008;38:1803–6.
Nemeth E, Valore EV, Territo M, Schiller G, Lichtenstein A, Ganz T. Hepcidin, a putative mediator of anemia of inflammation, is a type II acute-phase protein. Blood. 2003;101:2461–3.
Kawabata H, Tomosugi N, Kanda J, Tanaka Y, Yoshizaki K, Uchiyama T. Anti-interleukin 6 receptor antibody tocilizumab reduces the level of serum hepcidin in patients with multicentric Castleman’s disease. Haematologica. 2007;92:857–8.
Heinrich PC, Behrmann I, Haan S, Hermanns HM, Muller-Newen G, Schaper F. Principles of interleukin (IL)-6-type cytokine signalling and its regulation. Biochem J. 2003;374:1–20.
Malik N, Kallestad JC, Gunderson NL, Austin SD, Neubauer MG, Ochs V, et al. Molecular cloning, sequence analysis, and functional expression of a novel growth regulator, oncostatin M. Mol Cell Biol. 1989;9:2847–53.
Linsley PS, Kallestad J, Ochs V, Neubauer M. Cleavage of a hydrophilic C-terminal domain increases growth-inhibitory activity of oncostatin M. Mol Cell Biol. 1990;10:1882–90.
Grenier A, Dehoux M, Boutten A, Arce-Vicioso M, Durand G, Gougerot-Pocidalo MA, et al. Oncostatin M production and regulation by human polymorphonuclear neutrophils. Blood. 1999;93:1413–21.
Brown TJ, Lioubin MN, Marquardt H. Purification and characterization of cytostatic lymphokines produced by activated human T lymphocytes. Synergistic antiproliferative activity of transforming growth factor beta 1, interferon-gamma, and oncostatin M for human melanoma cells. J Immunol. 1987;139:2977–83.
Znoyko I, Sohara N, Spicer SS, Trojanowska M, Reuben A. Expression of oncostatin M and its receptors in normal and cirrhotic human liver. J Hepatol. 2005;43:893–900.
Suda T, Chida K, Todate A, Ide K, Asada K, Nakamura Y, et al. Oncostatin M production by human dendritic cells in response to bacterial products. Cytokine. 2002;17:335–40.
Richards CD, Brown TJ, Shoyab M, Baumann H, Gauldie J. Recombinant oncostatin M stimulates the production of acute phase proteins in HepG2 cells and rat primary hepatocytes in vitro. J Immunol. 1992;148:1731–6.
Baumann H, Gauldie J. The acute phase response. Immunol Today. 1994;15:74–80.
Tanaka M, Miyajima A. Oncostatin M, a multifunctional cytokine. Rev Physiol Biochem Pharmacol. 2003;149:39–52.
Nakamura K, Nonaka H, Saito H, Tanaka M, Miyajima A. Hepatocyte proliferation and tissue remodeling is impaired after liver injury in oncostatin M receptor knockout mice. Hepatology. 2004;39:635–44.
Murao N, Ishigai M, Yasuno H, Shimonaka Y, Aso Y. Simple and sensitive quantification of bioactive peptides in biological matrices using liquid chromatography/selected reaction monitoring mass spectrometry coupled with trichloroacetic acid clean-up. Rapid Commun Mass Spectrom. 2007;21:4033–8.
Wrighting DM, Andrews NC. Interleukin-6 induces hepcidin expression through STAT3. Blood. 2006;108:3204–9.
Kemna E, Pickkers P, Nemeth E, van der Hoeven H, Swinkels D. Time-course analysis of hepcidin, serum iron, and plasma cytokine levels in humans injected with LPS. Blood. 2005;106:1864–6.
Tomosugi N, Kawabata H, Wakatabe R, Higuchi M, Yamaya H, Umehara H, et al. Detection of serum hepcidin in renal failure and inflammation by using ProteinChip System. Blood. 2006;108:1381–7.
Waterston A, Bower M. Fifty years of multicentric Castleman’s disease. Acta Oncol. 2004;43:698–704.
Lee P, Peng H, Gelbart T, Beutler E. The IL-6- and lipopolysaccharide-induced transcription of hepcidin in HFE-, transferrin receptor 2-, and beta 2-microglobulin-deficient hepatocytes. Proc Natl Acad Sci USA. 2004;101:9263–5.
Urbanska-Rys H, Wiersbowska A, Stepien H, Robak T. Relationship between circulating interleukin-10 (IL-10) with interleukin-6 (IL-6) type cytokines (IL-6, interleukin-11 (IL-11), oncostatin M (OSM)) and soluble interleukin-6 (IL-6) receptor (sIL-6R) in patients with multiple myeloma. Eur Cytokine Netw. 2000;11:443–51.
Koskela K, Pelliniemi TT, Rajamaki A, Pulkki K, Remes K. Serum oncostatin M in multiple myeloma: impact on disease severity and prognosis. Eur J Haematol. 2000;65:52–6.
Sharma S, Nemeth E, Chen YH, Goodnough J, Huston A, Roodman GD, et al. Involvement of hepcidin in the anemia of multiple myeloma. Clin Cancer Res. 2008;14:3262–7.
Ganz T, Olbina G, Girelli D, Nemeth E, Westerman M. Immunoassay for human serum hepcidin. Blood. 2008;112:4292–7.
Gearing DP, Gough NM, King JA, Hilton DJ, Nicola NA, Simpson RJ, et al. Molecular cloning and expression of cDNA encoding a murine myeloid leukaemia inhibitory factor (LIF). EMBO J. 1987;6:3995–4002.
Gough NM. Molecular genetics of leukemia inhibitory factor (LIF) and its receptor. Growth Factors. 1992;7:175–9.
Brown MA, Metcalf D, Gough NM. Leukaemia inhibitory factor and interleukin 6 are expressed at very low levels in the normal adult mouse and are induced by inflammation. Cytokine. 1994;6:300–9.
Gearing DP. The leukemia inhibitory factor and its receptor. Adv Immunol. 1993;53:31–58.
Lorgeot V, Praloran V, Turlure P, Denizot Y. Concentrations of serum leukemia inhibitory factor (LIF) in patients with hematologic malignancies. Leukemia. 1997;11:311–2.
Koliaraki V, Marinou M, Vassilakopoulos TP, Vavourakis E, Tsochatzis E, Pangalis GA, et al. A novel immunological assay for hepcidin quantification in human serum. PLoS One. 2009;4:e4581.
Verga Falzacappa MV, Vujic Spasic M, Kessler R, Stolte J, Hentze MW, Muckenthaler MU. STAT3 mediates hepatic hepcidin expression and its inflammatory stimulation. Blood. 2007;109:353–8.
Pietrangelo A, Dierssen U, Valli L, Garuti C, Rump A, Corradini E, et al. STAT3 is required for IL-6-gp130-dependent activation of hepcidin in vivo. Gastroenterology. 2007;132:294–300.
Acknowledgments
We thank Dr. Hideyuki Tsuchida (Division of Advanced Medicine, Medical Research Institute, Kanazawa Medical University, Ishikawa, Japan) for measuring the hepcidin-25 levels in the cell culture supernatant. This work was supported in part by a grant-in-aid for scientific research from the Ministry of Education, Culture, Sports, Science and Technology of Japan; a grant from Takeda Science Foundation; and a grant for Project Research from the High-Technology Center of Kanazawa Medical University (H2007-2).
Conflict of interest statement
N.T. declares that he is also the President of Medical Care Proteomics Biotechnology Co. Ltd. (Ishikawa-ken, Japan), a start-up company, the stocks of which are not publicly traded. Other authors declare that they have no conflicts of interest relevant to this paper.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Kanda, J., Uchiyama, T., Tomosugi, N. et al. Oncostatin M and leukemia inhibitory factor increase hepcidin expression in hepatoma cell lines. Int J Hematol 90, 545–552 (2009). https://doi.org/10.1007/s12185-009-0443-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-009-0443-x