Abstract
Many patients with diabetes fail to achieve their glycaemic targets despite clear evidence that glycaemic control can prevent or delay the development of costly complications in diabetes. This article describes how insulin analogues (insulins lispro, aspart, glulisine, glargine and detemir) may have a role to play in overcoming barriers to insulin acceptance and improving adherence with therapy, and examines their cost-effectiveness as determined in published studies.
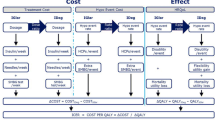
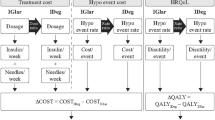
Cost-effectiveness studies attempt to assess all the costs and all the benefits of a particular therapy. Pharmacoeconomic models, which calculate the long-term effect of interventions, show that over periods such as 10 or 35 years the higher prescription costs of analogues are offset by a lower incidence of acute hypoglycaemic events and costly, chronic complications such as nephropathy. This results in costs per ‘quality-adjusted life year’ that fall well within accepted limits for good value for money.
Retrospective analyses of managed care databases show that higher prescription costs for analogues are offset by lower hypoglycaemia-related costs and/or inpatient claims. Relative to human insulins, analogues provide a better balance between glycaemic control and tolerability. Patients’ fear of hypoglycaemia is allayed; the pen devices used with analogues facilitate insulin injection; and the pharmacokinetic characteristics lead to increased flexibility and convenience. All these factors can help increase adherence with therapy, which may in itself be cost-saving. Taken overall, these results indicate that insulin analogues are a cost-effective therapy.
Similar content being viewed by others
References
American Diabetes Association. Economic costs of diabetes in the US in 2002. Diabetes Care. 2003;26:917–932.
American Diabetes Association. Standards of medical care in diabetes. Diabetes Care. 2007;30(suppl 1):S1–S41.
AACE Diabetes Mellitus Clinical Practice Guidelines Task Force. American Association of Clinical Endocrinologists medical guidelines for clinical practice for the management of diabetes mellitus. Endocr Pract. 2007;13(suppl 1):1–66.
Writing Team for the DCCT/EDIC Research Group. Effect of intensive therapy on the microvascular complications of type 1 diabetes mellitus. JAMA. 2002;287:2563–2569.
Writing Team for the DCCT/EDIC Research Group. Sustained effect of intensive treatment of type 1 diabetes mellitus on development and progression of diabetic nephropathy: the Epidemiology of Diabetes Interventions and Complications (EDIC) study. JAMA. 2003;290:2159–2167.
UKPDS. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet. 1998;352:837–853.
Stratton IM, Cull CA, Adler AI, Matthews DR, Neil HA, Holman RR. Additive effects of glycaemia and blood pressure exposure on risk of complications in type 2 diabetes: a prospective observational study (UKPDS 75). Diabetologia. 2006;49:1761–1769.
Stratton IM, Adler AI, Neil HA, et al. Association of glycaemia with macrovascular and microvascular complications of type 2 diabetes (UKPDS 35): prospective observational study. BMJ. 2000;321:405–412.
DCCT/EDIC Study Research Group. Intensive diabetes treatment and cardiovascular disease in patients with type 1 diabetes. N Engl J Med. 2005;353:2643–2653.
Klonoff DC, Schwartz DM. An economic analysis of interventions for diabetes. Diabetes Care. 2000;23:390–404.
Shetty S, Secnik K, Oglesby AK. Relationship of glycemic control to total diabetes-related costs for managed care health plan members with type 2 diabetes. J Manag Care Pharm. 2005;11:559–564.
Caro JJ, Ward AJ, O’Brien JA. Lifetime costs of complications resulting from type 2 diabetes in the US. Diabetes Care. 2002;25:476–481.
Resnick HE, Foster GL, Bardsley J, Ratner RE. Achievement of American Diabetes Association clinical practice recommendations among U.S. adults with diabetes, 1999–2002: the National Health and Nutrition Examination Survey. Diabetes Care. 2006;29:531–537.
Bond GG, Aiken LS, Somerville SC. The health belief model and adolescents with insulin-dependent diabetes mellitus. Health Psychol. 1992;11:190–198.
Tan MY. The relationship of health beliefs and complication prevention behaviors of Chinese individuals with type 2 diabetes mellitus. Diabetes Res Clin Pract. 2004;66:71–77.
Hunt LM, Valenzuela MA, Pugh JA. NIDDM patients’ fears and hopes about insulin therapy. The basis of patient reluctance. Diabetes Care. 1997;20:292–298.
Peyrot M, Rubin RR, Lauritzen T, et al. The International DAWN Advisory Panel. Resistance to insulin therapy among patients and providers: results of the crossnational Diabetes Attitudes, Wishes, and Needs (DAWN) study. Diabetes Care. 2005;28:2673–2679.
Korytkowski M. When oral agents fail: practical barriers to starting insulin. Int J Obes Relat Metab Disord. 2002;26(suppl 3):S18–S24.
Davis S, Alonso MD. Hypoglycemia as a barrier to glycemic control. J Diabetes Complications. 2004;18:60–68.
Cramer J. A systematic review of adherence with medications for diabetes. Diabetes Care. 2004;27:1218–1224.
Polonsky WH, Anderson BJ, Lohrer PA, Aponte JE, Jacobson AM, Cole CF. Insulin omission in women with IDDM. Diabetes Care. 1994;17:1178–1185.
Lee WC, Balu S, Cobden D, Joshi AV, Pashos CL. Prevalence and economic consequences of medication adherence in diabetes: a systematic literature review. Manag Care Interface. 2006;19:31–41.
Wooldridge KL, Wallston KA, Graber AL, Brown AW, Davidson P. The relationship between health beliefs, adherence, and metabolic control of diabetes. Diabetes Educ. 1992;18:495–500.
Heisler M, Piette JD, Spencer M, Kieffer E, Vijan S. The relationship between knowledge of recent HbA1c values and diabetes care understanding and self-management. Diabetes Care. 2005;28:816–822.
Rolla A. Pharmacokinetic and pharmacodynamic advantages of insulin analogs and premixed insulin analogs over human insulins: impact on efficacy and safety. Am J Med. 2008;121(suppl). In press.
Anderson JH Jr, Brunelle RL, Koivisto VA, et al. Reduction of postprandial hyperglycemia and frequency of hypoglycemia in IDDM patients on insulin-analog treatment. Multicenter Insulin Lispro Study Group. Diabetes. 1997;46:265–270.
Lindholm A, McEwen J, Riis AP. Improved postprandial glycaemic control with insulin aspart. A randomized double-blind crossover trial in type 1 diabetes. Diabetes Care. 1999;22:801–805.
Garg SK, Ellis SL, Ulrich H. Insulin glulisine: a new rapid-acting insulin analogue for the treatment of diabetes. Expert Opin Pharmacother. 2005;6:643–651.
Ross SA, Zinman B, Campos RV, Strack T. Canadian Lispro Study Group. A comparative study of insulin lispro and human regular insulin in patients with type 2 diabetes mellitus and secondary failure of oral hypoglycemic agents. Clin Invest Med. 2001;24:292–298.
Perriello G, Pampanelli S, Porcellati F, et al. Insulin aspart improves meal time glycaemic control in patients with type 2 diabetes: a randomized, stratified, doubleblind and cross-over trial. Diabet Med. 2005;22:606–611.
Chatterjee S, Tringham JR, Davies MJ. Insulin glargine and its place in the treatment of types 1 and 2 diabetes mellitus. Expert Opin Pharmacother. 2006;7:1357–1371.
Home P, Kurtzhals P. Insulin detemir: from concept to clinical experience. Expert Opin Pharmacother. 2006;7:325–343.
Heise T, Nosek L, Ronn BB, et al. Lower within-subject variability of insulin detemir in comparison to NPH insulin and insulin glargine in people with type 1 diabetes. Diabetes. 2004;53:1614–1620.
Klein O, Lynge J, Endahl L, Damholt B, Nosek L, Heise T. Albumin-bound basal insulin analogues (insulin detemir and NN344): comparable time-action profiles but less variability than insulin glargine in type 2 diabetes. Diabetes Obes Metab. 2007;9:290–299.
Gough S. A review of human and analogue insulin trials. Diabetes Res Clin Pract. 2007;77:1–15.
Hermansen K, Davies M. Does insulin detemir have a role in reducing risk of insulin-associated weight gain? Diabetes Obes Metab. 2007;9:209–217.
Korytkowski M, Bell D, Jacobsen C, Suwannasari R. FlexPen Study Team. A multicenter, randomized, open-label, comparative, two-period crossover trial of preference, efficacy, and safety profiles of a prefilled, disposable pen and conventional vial/syringe for insulin injection in patients with type 1 or 2 diabetes mellitus. Clin Ther. 2003;25:2836–2848.
Da Costa S, Brackenridge B, Hicks D. A comparison of insulin pen use in the United States and the United Kingdom. Diabetes Educ. 2002;28:52–60.
Bohannon NJ. Insulin delivery using pen devices. Simple-to-use tools may help young and old alike. Postgrad Med. 1999;106:57–68.
Ashkenazy R, Abrahamson MJ. Medicare coverage for patients with diabetes. J Gen Intern Med. 2006;21:386–392.
Reimbursement information. Paying for NovoLog® Mix 70/30. Novo Nordisk web site. Available at: http://www.novologmix70-30.com/reimbursement.asp. Accessed 10 January 2008.
Kotsanos JG, Vignati L, Huster W, et al. Health-related quality-of-life results from multinational clinical trials of insulin lispro. Assessing benefits of a new diabetes therapy. Diabetes Care. 1997;20:948–958.
Bott U, Ebrahim S, Hirschberger S, Skovlund SE. Effect of the insulin analogue insulin aspart on quality-of-life and treatment satisfaction in type 1 diabetic patients. Diabet Med. 2003;20:626–634.
Manini R, Forlani G, Moscatiello S, Zannoni C, Marzocchi R, Marchesini G. Insulin glargine improves glycemic control and health-related quality of life in type 1 diabetes. Nutr Metab Cardiovasc Dis. 2006;17:493–498.
Palmer AJ, Roze S, Valentine WJ, et al. The CORE diabetes model: projecting longterm clinical outcomes, costs and costeffectiveness of interventions in diabetes mellitus (types 1 and 2) to support clinical and reimbursement decision-making. Curr Med Res Opin. 2004;20(suppl 1):S5–S26.
Valentine WJ, Palmer AJ, Erny-Albrecht KM, et al. Cost-effectiveness of basal insulin from a US health system perspective: comparative analyses of detemir, glargine, and NPH. Adv Ther. 2006;23:191–207.
Palmer A, Cobden D, Koenen C, et al. Projecting the economic and health outcome effects of basal insulin among type 2 patients in clinical practice settings. Diabet Med. 2006;23(suppl 4):368.
Bullano MF, Al-Zakwani IS, Fisher MD, Menditto L, Willey VJ. Differences in hypoglycemia event rates and associated cost-consequence in patients initiated on long-acting and intermediate-acting insulin products. Curr Med Res Opin. 2005;21:291–298.
Bullano MF, Fisher MD, Grochulski WD, Menditto L, Willey VJ. Hypoglycemic events and glycosylated hemoglobin values in patients with type 2 diabetes mellitus newly initiated on insulin glargine or premixed insulin combination products. Am J Health Syst Pharm. 2006;63:2473–2482.
Zhang Q, Menditto L. Incremental cost savings 6 months following initiation of insulin glargine in a Medicaid fee-for-service sample. Am J Ther. 2005;12:337–343.
Orsini L, Huse DM. Total health care costs in medicaid patients receiving NPH, lente, or insulin glargine. Diabetes. 2005;54(suppl 1):A144.
Miller DM, Gardner J, Hendricks A, Fincke G, Zhang Q. Changes in health care utilization and expenditures associated with insulin glargine use. Diabetes. 2005;54(suppl 1):A302.
Ory C, Cyprien L, Menditto LA, Chang E. Health economic outcomes associated with insulin therapies: an evaluation of insulin glargine. Diabetes. 2005;54(suppl 1):A612.
Hall JA, Summers KH, Obenchain RL. Cost and utilization comparisons among propensity score-matched insulin lispro and regular insulin users. J Manag Care Pharm. 2003;9:2638.
Chen K, Chang EY, Summers KH, Obenchain RL, Yu-Isenberg KS, Sun P. Comparison of costs and utilization between users of insulin lispro versus users of regular insulin in a managed care setting. J Manag Care Pharm. 2005;11:376–382.
Valentine WJ, Erny-Albrecht KM, Ray JA, Roze S, Cobden D, Palmer AJ. Therapy conversion to insulin detemir among patients with type 2 diabetes treated with oral agents: a modeling study of costeffectiveness in the United States. Adv Ther. 2007;24:273–290
Minshall M, Ray J, Lammert M, et al. Estimating the long-term cost-effectiveness of biphasic insulin aspart plus metformin versus optimization of oral hypoglycemic agents in insulin-naïve patients with type 2 diabetes in a US cost setting. Diabetologia. 2005;48(suppl 1):A335.
Valentine W, Cobden D, Braceras R, et al. Health economic evaluation of improved glycemic control: initiating biphasic analog insulin. Diabet Med. 2006;23(suppl 4):367.
Nicklasson L, Palmer AJ, Cobden D, et al. Long-term cost-effectiveness analysis of patients with type 2 diabetes mellitus inadequately controlled on thiazolidenedione therapy and either biphasic insulin aspart 30 or sulfonylurea. Diabetologia. 2005;48(suppl 1):A334.
Grima DT, Thompson MF, Sauriol L. Modelling cost effectiveness of insulin glargine for the treatment of type 1 and 2 diabetes in Canada. Pharmacoeconomics. 2007;25:253–266.
Palmer AJ, Valentine WJ, Ray JA, et al. An economic assessment of analogue basal-bolus insulin versus human basal-bolus insulin in subjects with type 1 diabetes in the UK. Curr Med Res Opin. 2007;23:895–901.
Lammert M, Plamer A, Roze S, et al. Decreased rates of major hypoglycemic events lead to improved long-term costeffectiveness of biphasic insulin aspart 30/70 versus biphasic human insulin 30 in type 2 diabetic subjects in Danish, Finnish, German, Norwegian, Spanish, Swedish and UK settings. Value Health. 2004;7:736.
Express Scripts. 2004 Drug Trend Report. Express Scripts: Maryland Heights, Missouri, USA; 2005. Available at: http://express-scripts.com/industryresearch/industryreports/drugtrendreport/2004/dtrFinal.pdf. Accessed 25 February 2008.
IMS Health. Data on file, 2000 and 2007.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Leichter, S. Is the use of insulin analogues cost-effective?. Adv Therapy 25, 285–299 (2008). https://doi.org/10.1007/s12325-008-0043-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-008-0043-9