Abstract
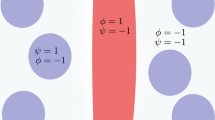
Fluid shear stress has been implicated as a regulator of sprouting angiogenesis. However, whether endothelial cells within capillary sprouts in vivo experience physiologically relevant shear stresses remains unclear. The objective of our study is to estimate the shear stress distribution along the length of a capillary sprout through computational modeling of blood flow in a blind-ended channel branching off a host vessel. In this model, we use sprout geometries typical for the rat mesenteric microvasculature and consider three types of boundary conditions: (1) a non-permeable vessel wall, (2) a uniformly permeable vessel wall, and (3) a non-permeable vessel wall with open slots (representative of endothelial clefts). Our numerical simulation predicts that for each boundary condition a local maximum shear stress (13.9, 8.9, and 13.3 dyne cm−2, respectively) occurs at the entrance of a 50 μm long, 6 μm diameter sprout branching at 90° off of a 11 μm diameter host vessel. The shear stress dropped below 0.2 dyne cm−2, a threshold for endothelial cell activation, within 4.1 μm of the entrance for the non-permeable wall case and 4.2 μm for the uniformly permeable wall case. Shear stress magnitudes within the sprout were above 0.2 dyne cm−2 for longer sprout scenarios and peaked at 5.9 dyne cm−2 at endothelial cell clefts. These results provide a first estimate of relative fluid shear stress magnitudes along a capillary sprout and highlight the importance of investigating endothelial cell responses to flow conditions during angiogenesis in tumors and other altered microenvironments.










Similar content being viewed by others
References
Anderson, C. R., A. M. Ponce, and R. J. Price. Immunohistochemical identification of an extracellular matrix scaffold that microguides capillary sprouting in vivo. J. Histochem. Cytochem. 52:1063–1072, 2004.
Barber, J. O., J. P. Alberding, J. M. Restrepo, and T. W. Secomb. Simulated two-dimensional red blood cell motion, deformation, and partitioning in microvessel bifurcations. Ann. Biomed. Eng. 36:1690–1698, 2008.
Bates, D. O. The chronic effect of vascular endothelial growth factor on individually perfused frog mesenteric microvessels. J. Physiol. 513(Pt 1):225–233, 1998.
Chen, B. P., Y. S. Li, Y. Zhao, K. D. Chen, S. Li, J. Lao, S. Yuan, J. Y. Shyy, and S. Chien. DNA microarray analysis of gene expression in endothelial cells in response to 24-h shear stress. Physiol. Genomics 7:55–63, 2001.
Cooke, J. P., E. Rossitch, Jr., N. A. Andon, J. Loscalzo, and V. J. Dzau. Flow activates an endothelial potassium channel to release an endogenous nitrovasodilator. J. Clin. Invest. 88:1663–1671, 1991.
Dai, G., M. R. Kaazempur-Mofrad, S. Natarajan, Y. Zhang, S. Vaughn, B. R. Blackman, R. D. Kamm, G. Garcia-Cardena, and M. A. Gimbrone, Jr. Distinct endothelial phenotypes evoked by arterial waveforms derived from atherosclerosis-susceptible and -resistant regions of human vasculature. Proc. Natl Acad. Sci. USA 101:14871–14876, 2004.
Davies, P. F. Flow-mediated endothelial mechanotransduction. Physiol. Rev. 75:519–560, 1995.
Davies, P. F., A. Remuzzi, E. J. Gordon, C. F. Dewey, Jr., and M. A. Gimbrone, Jr. Turbulent fluid shear stress induces vascular endothelial cell turnover in vitro. Proc. Natl Acad. Sci. USA 83:2114–2117, 1986.
Davies, P. F., A. Robotewskyj, and M. L. Griem. Quantitative studies of endothelial cell adhesion. Directional remodeling of focal adhesion sites in response to flow forces. J. Clin. Invest. 93:2031–2038, 1994.
DePaola, N., M. A. Gimbrone, Jr., P. F. Davies, and C. F. Dewey, Jr. Vascular endothelium responds to fluid shear stress gradients. Arterioscler. Thromb. 12:1254–1257, 1992.
Dewey, Jr., C. F., S. R. Bussolari, M. A. Gimbrone, Jr., and P. F. Davies. The dynamic response of vascular endothelial cells to fluid shear stress. J. Biomech. Eng. 103:177–185, 1981.
Evans, E., and Y. C. Fung. Improved measurements of the erythrocyte geometry. Microvasc. Res. 4:335–347, 1972.
Fraser, P. A., L. H. Smaje, and A. Verrinder. Microvascular pressures and filtration coefficients in the cat mesentery. J. Physiol. 283:439–456, 1978.
Fukumura, D., D. G. Duda, L. L. Munn, and R. K. Jain. Tumor microvasculature and microenvironment: novel insights through intravital imaging in pre-clinical models. Microcirculation 17:206–225, 2010.
Fung, Y. C. Mechanics of erythrocytes, leukocytes, and other cells. In: Biomechanics Mechanical Properties of Living Tissues Anonymous New York. New York: Springer Science + Business Media, LLC, 1993, pp. 109–164.
Galbraith, C. G., R. Skalak, and S. Chien. Shear stress induces spatial reorganization of the endothelial cell cytoskeleton. Cell Motil. Cytoskeleton 40:317–330, 1998.
Gerhardt, H. VEGF and endothelial guidance in angiogenic sprouting. Organogenesis 4:241–246, 2008.
Gerhardt, H., M. Golding, M. Fruttiger, C. Ruhrberg, A. Lundkvist, A. Abramsson, M. Jeltsch, C. Mitchell, K. Alitalo, D. Shima, and C. Betsholtz. VEGF guides angiogenic sprouting utilizing endothelial tip cell filopodia. J. Cell Biol. 161:1163–1177, 2003.
Guerreiro-Lucas, L. A., S. R. Pop, M. J. Machado, Y. L. Ma, S. L. Waters, G. Richardson, K. Saetzler, O. E. Jensen, and C. A. Mitchell. Experimental and theoretical modelling of blind-ended vessels within a developing angiogenic plexus. Microvasc. Res. 76:161–168, 2008.
Hahn, C., and M. A. Schwartz. Mechanotransduction in vascular physiology and atherogenesis. Nat. Rev. Mol. Cell Biol. 10:53–62, 2009.
le Noble, F., D. Moyon, L. Pardanaud, L. Yuan, V. Djonov, R. Matthijsen, C. Breant, V. Fleury, and A. Eichmann. Flow regulates arterial-venous differentiation in the chick embryo yolk sac. Development 131:361–375, 2004.
Lin, K., P. P. Hsu, B. P. Chen, S. Yuan, S. Usami, J. Y. Shyy, Y. S. Li, and S. Chien. Molecular mechanism of endothelial growth arrest by laminar shear stress. Proc. Natl Acad. Sci. USA 97:9385–9389, 2000.
Milkiewicz, M., M. D. Brown, S. Egginton, and O. Hudlicka. Association between shear stress, angiogenesis, and VEGF in skeletal muscles in vivo. Microcirculation 8:229–241, 2001.
Mitsumata, M., R. S. Fishel, R. M. Nerem, R. W. Alexander, and B. C. Berk. Fluid shear stress stimulates platelet-derived growth factor expression in endothelial cells. Am. J. Physiol. 265:H3–H8, 1993.
Murfee, W. L., M. R. Rehorn, S. M. Peirce, and T. C. Skalak. Perivascular cells along venules upregulate NG2 expression during microvascular remodeling. Microcirculation 13:261–273, 2006.
Neal, C. R., and C. C. Michel. Differing effects of Vascular Endothelial Growth Factor (VEGF) on the ultrastructure of mesenteric microvessels of frog and rat. J. Physiol. 506:24P, 1998.
Pries, A. R., T. W. Secomb, and P. Gaehtgens. Relationship between structural and hemodynamic heterogeneity in microvascular networks. Am. J. Physiol. 270:H545–H553, 1996.
Rhodin, J. A., and H. Fujita. Capillary growth in the mesentery of normal young rats. Intravital video and electron microscope analyses. J. Submicrosc. Cytol. Pathol. 21:1–34, 1989.
Schmid-Schonbein, G. W., R. Skalak, S. Usami, and S. Chien. Cell distribution in capillary networks. Microvasc. Res. 19:18–44, 1980.
Secomb, T. W., R. Skalak, N. Ozkaya, and J. F. Gross. Flow of axisymmetric red blood cells in narrow capillaries. J. Fluid Mech. 163:405–423, 1986.
Shankar, P. N., and M. D. Deshpande. Fluid mechanics in the driven cavity. Annu. Rev. Fluid Mech. 32:93–136, 2000.
Shevkoplyas, S. S., T. Yoshida, S. C. Gifford, and M. W. Bitensky. Direct measurement of the impact of impaired erythrocyte deformability on microvascular network perfusion in a microfluidic device. Lab. Chip. 6:914–920, 2006.
Skalak, T. C., and R. J. Price. The role of mechanical stresses in microvascular remodeling. Microcirculation 3:143–165, 1996.
Sugihara-Seki, M., and R. Skalak. Numerical study of asymmetric flows of red blood cells in capillaries. Microvasc. Res. 36:64–74, 1988.
Tarbell, J. M., L. Demaio, and M. M. Zaw. Effect of pressure on hydraulic conductivity of endothelial monolayers: role of endothelial cleft shear stress. J. Appl. Physiol. 87:261–268, 1999.
Tardy, Y., N. Resnick, T. Nagel, M. A. Gimbrone, Jr., and C. F. Dewey, Jr. Shear stress gradients remodel endothelial monolayers in vitro via a cell proliferation-migration-loss cycle. Arterioscler. Thromb. Vasc. Biol. 17:3102–3106, 1997.
Tözeren, H., and R. Skalak. The steady flow of closely fitting incompressible elastic spheres in a tube. J. Fluid Mech. 87:1–16, 1978.
Wang, H., and R. Skalak. Viscous flow in a cylindrical tube containing a line of spherical particles. J. Fluid Mech. 38:75–96, 1969.
Xiong, W., and J. Zhang. Shear stress variation induced by red blood cell motion in microvessel. Ann. Biomed. Eng. 38:2649–2659, 2010.
Fung, Y. C. Microcirculation. In: Biomechanics: Circulation Anonymous New York. New York: Springer Science + Business Media, LLC, 1997, pp. 266–332.
Acknowledgments
This work was supported by Louisiana Board of Regents grants LEQSF(2007-12)-ENH-PKSFI-PRS-01 (D. Khismatullin) and LEQSF(2009-12)-RD-A-19 (W. Murfee).
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Laura Suggs oversaw the review of this article.
P. C. Stapor and W. Wang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Stapor, P.C., Wang, W., Murfee, W.L. et al. The Distribution of Fluid Shear Stresses in Capillary Sprouts. Cardiovasc Eng Tech 2, 124–136 (2011). https://doi.org/10.1007/s13239-011-0041-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13239-011-0041-y