Abstract
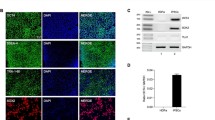
Human immunodeficiency virus (HIV) primarily infects glial cells in the central nervous system (CNS). Recent evidence suggests that HIV-infected individuals who abuse drugs such as methamphetamine (METH) have higher viral loads and experience more severe neurological complications than HIV-infected individuals who do not abuse drugs. The aim of this study was to determine the effect of METH on HIV expression from the HIV long terminal repeat (LTR) promoter and on an HIV integrated provirus in microglial cells, the primary host cells for HIV in the CNS. Primary human microglial cells immortalized with SV40 T antigen (CHME-5 cells) were cotransfected with an HIV LTR reporter and the HIV Tat gene, a key regulator of viral replication and gene expression, and exposed to METH. Our results demonstrate that METH treatment induced LTR activation, an effect potentiated in the presence of Tat. We also found that METH increased the nuclear translocation of the nuclear factor kappa B (NF-κB), a key cellular transcriptional regulator of the LTR promoter, and the activity of an NF-κB-specific reporter plasmid in CHME-5 cells. The presence of a dominant-negative regulator of NF-κB blocked METH-related activation of the HIV LTR. Furthermore, treatment of HIV-latently infected CHME-5 (CHME-5/HIV) cells with METH induced HIV expression and nuclear translocation of the p65 subunit of NF-κB. These results suggest that METH can stimulate HIV gene expression in microglia cells through activation of the NF-κB signaling pathway. This mechanism may outline the initial biochemical events leading to the observed increased neurodegeneration in HIV-positive individuals who use METH.





Similar content being viewed by others
References
Asanuma M, Cadet J (1998) Methamphetamine-induced increase in striatal NF-kappaB DNA-binding activity is attenuated in superoxide dismutase transgenic mice. Brain Res Mol Brain Res 60:305–9
Buchacz K, McFarland W, Kellogg TA, Loeb L, Holmberg SD, Dilley J, Klausner JD (2005) Amphetamine use is associated with increased HIV incidence among men who have sex with men in San Francisco. AIDS 19:1423–4
Cadet JL, Krasnova IN (2007) Interactions of HIV and methamphetamine: cellular and molecular mechanisms of toxicity potentiation. Neurotox Res 12:181–204
Cass W, Harned M, Peters L, Nath A, Maragos W (2003) HIV-1 protein Tat potentiation of methamphetamine-induced decreases in evoked overflow of dopamine in the striatum of the rat. Brain Res 984:133–42
Chang L, Ernst T, Speck O, Grob CS (2005) Additive effects of HIV and chronic methamphetamine use on brain metabolite abnormalities. Am J Psychiatry 162:361–9
Copeland K (2005) Modulation of HIV-1 transcription by cytokines and chemokines. Mini Rev Med Chem 5:1093–101
Cox C, Cao S, Lu Y (2009) Enhanced detection and study of murine norovirus-1 using a more efficient microglial cell line. Virol J 6:196
Dull T, Zufferey R, Kelly M, Mandel RJ, Nguyen M, Trono D, Naldini L (1998) A third‐generation lentivirus vector with a conditional packaging system. J Virol 72:8463–71
Ellis R, Childers M, Cherner M, Lazzaretto D, Letendre S, Grant I, Group HNRC (2003) Increased human immunodeficiency virus loads in active methamphetamine users are explained by reduced effectiveness of antiretroviral therapy. J Infect Dis 188:1820–6
Freeman P, Walker BC, Harris DR, Garofalo R, Willard N, Ellen JM, Team ftATNfHAIb (2011) Methamphetamine use and risk for HIV among young men who have sex with men in 8 US cities. Arch Pediatr Adolesc Med 165:736–740
Gavrilin M, Mathes L, Podell M (2002) Methamphetamine enhances cell-associated feline immunodeficiency virus replication in astrocytes. J Neurovirol 8:240–9
Ghosh S, Karin M (2002) Missing pieces in the NF-kappaB puzzle. Cell 109(Suppl):S81–96
Guilarte TR, Nihei MK, McGlothan JL, Howard AS (2003) Methamphetamine-induced deficits of brain monoaminergic neuronal markers: distal axotomy or neuronal plasticity. Neuroscience 122:499–513
Janabi N, Peudenier S, Héron B, Ng KH, Tardieu M (1995) Establishment of human microglial cell lines after transfection of primary cultures of embryonic microglial cells with the SV40 large T antigen. Neurosci Lett 195:105–8
Jayanthi S, Deng X, Ladenheim B, McCoy M, Cluster A, Cai N, Cadet J (2005) Calcineurin/NFAT-induced up-regulation of the Fas ligand/Fas death pathway is involved in methamphetamine-induced neuronal apoptosis. Proc Natl Acad Sci U S A 102:868–73
Kaul M, Lipton SA (2006) Mechanisms of neuronal injury and death in HIV-1 associated dementia. Curr HIV Res 4:307–18
Kaul M, Garden GA, Lipton SA (2001) Pathways to neuronal injury and apoptosis in HIV-associated dementia. Nature 410:988–94
Kim YK, Bourgeois CF, Pearson R, Tyagi M, West MJ, Wong J, Wu SY, Chiang CM, Karn J (2006) Recruitment of TFIIH to the HIV LTR is a rate-limiting step in the emergence of HIV from latency. EMBO J 25:3596–604
King JE, Eugenin EA, Buckner CM, Berman JW (2006) HIV tat and neurotoxicity. Microbes Infect 8:1347–57
Langford D, Adame A, Grigorian A, Grant I, McCutchan JA, Ellis RJ, Marcotte TD, Masliah E, Group HNRC (2003) Patterns of selective neuronal damage in methamphetamine-user AIDS patients. J Acquir Immune Defic Syndr 34:467–74
LaVoie M, Card J, Hastings T (2004) Microglial activation precedes dopamine terminal pathology in methamphetamine-induced neurotoxicity. Exp Neurol 187:47–57
Lee SC, Hatch WC, Liu W, Brosnan CF, Dickson DW (1993a) Productive infection of human fetal microglia in vitro by HIV-1. Ann N Y Acad Sci 693:314–6
Lee SC, Hatch WC, Liu W, Kress Y, Lyman WD, Dickson DW (1993b) Productive infection of human fetal microglia by HIV-1. Am J Pathol 143:1032–9
Mallardo M, Dragonetti E, Baldassarre F, Ambrosino C, Scala G, Quinto I (1996) An NF-kappaB site in the 5′-untranslated leader region of the human immunodeficiency virus type 1 enhances the viral expression in response to NF-kappaB-activating stimuli. J Biol Chem 271:20820–7
Maragos WF, Young KL, Turchan JT, Guseva M, Pauly JR, Nath A, Cass WA (2002) Human immunodeficiency virus-1 Tat protein and methamphetamine interact synergistically to impair striatal dopaminergic function. J Neurochem 83:955–63
Mattson MP (2005) NF-kappaB in the survival and plasticity of neurons. Neurochem Res 30:883–93
Mocchetti I, Nosheny RL, Tanda G, Ren K, Meyer EM (2007) Brain-derived neurotrophic factor prevents human immunodeficiency virus type 1 protein gp120 neurotoxicity in the rat nigrostriatal system. Ann N Y Acad Sci 1122:144–54
Nath A, Maragos W, Avison M, Schmitt F, Berger J (2001) Acceleration of HIV dementia with methamphetamine and cocaine. J Neurovirol 7:66–71
Pearson R, Kim YK, Hokello J, Lassen K, Friedman J, Tyagi M, Karn J (2008) Epigenetic silencing of human immunodeficiency virus (HIV) transcription by formation of restrictive chromatin structures at the viral long terminal repeat drives the progressive entry of HIV into latency. J Virol 82:12291–303
Pereira L, Bentley K, Peeters A, Churchill M, Deacon N (2000) A compilation of cellular transcription factor interactions with the HIV-1 LTR promoter. Nucleic Acids Res 28:663–8
Ravi D, Mitra D (2007) HIV-1 long terminal repeat promoter regulated dual reporter: potential use in screening of transcription modulators. Anal Biochem 360:315–7
Rippeth JD, Heaton RK, Carey CL, Marcotte TD, Moore DJ, Gonzalez R, Wolfson T, Grant I, Group H (2004) Methamphetamine dependence increases risk of neuropsychological impairment in HIV infected persons. J Int Neuropsychol Soc 10:1–14
Sanchez JF, Sniderhan LF, Williamson AL, Fan S, Chakraborty-Sett S, Maggirwar SB (2003) Glycogen synthase kinase 3beta-mediated apoptosis of primary cortical astrocytes involves inhibition of nuclear factor kappaB signaling. Mol Cell Biol 23:4649–62
Sekine Y, Ouchi Y, Sugihara G, Takei N, Yoshikawa E, Nakamura K, Iwata Y, Tsuchiya K, Suda S, Suzuki K, Kawai M, Takebayashi K, Yamamoto S, Matsuzaki H, Ueki T, Mori N, Gold M, Cadet J (2008) Methamphetamine causes microglial activation in the brains of human abusers. J Neurosci 28:5756–61
Sheng P, Ladenheim B, Moran T, Wang X, Cadet J (1996) Methamphetamine-induced neurotoxicity is associated with increased striatal AP-1 DNA-binding activity in mice. Brain Res Mol Brain Res 42:171–4
Tallóczy Z, Martinez J, Joset D, Ray Y, Gácser A, Toussi S, Mizushima N, Nosanchuk J, Nosanchuk J, Goldstein H, Loike J, Sulzer D, Santambrogio L (2008) Methamphetamine inhibits antigen processing, presentation, and phagocytosis. PLoS Pathog 4:e28
Tambuyzer BR, Ponsaerts P, Nouwen EJ (2009) Microglia: gatekeepers of central nervous system immunology. J Leukoc Biol 85:352–70
Tang H, Kuhen K, Wong-Staal F (1999) Lentivirus replication and regulation. Annu Rev Genet 33:133–70
Theodore S, Cass W, Maragos W (2006a) Methamphetamine and human immunodeficiency virus protein Tat synergize to destroy dopaminergic terminals in the rat striatum. Neuroscience 137:925–35
Theodore S, Cass WA, Nath A, Steiner J, Young K, Maragos WF (2006b) Inhibition of tumor necrosis factor-alpha signaling prevents human immunodeficiency virus-1 protein Tat and methamphetamine interaction. Neurobiol Dis 23:663–8
Thomas D, Kuhn D (2005) MK-801 and dextromethorphan block microglial activation and protect against methamphetamine-induced neurotoxicity. Brain Res 1050:190–8
Thomas D, Walker P, Benjamins J, Geddes T, Kuhn D (2004) Methamphetamine neurotoxicity in dopamine nerve endings of the striatum is associated with microglial activation. J Pharmacol Exp Ther 311:1–7
Toussi S, Joseph A, Zheng J, Dutta M, Santambrogio L, Goldstein H (2009) Short communication: methamphetamine treatment increases in vitro and in vivo HIV replication. AIDS Res Hum Retrovir 25:1117–21
Volkow ND, Fowler JS, Wang GJ, Shumay E, Telang F, Thanos PK, Alexoff D (2010) Distribution and pharmacokinetics of methamphetamine in the human body: clinical implications. PLoS One 5:e15269
Wang X, Watanabe Y, Osugi T, Ikemoto M, Hirata M, Miki N (1992) In situ DNA-protein binding: a novel method for detecting DNA-binding activity of transcription factor in brain. Neurosci Lett 146:25–8
Acknowledgments
The following reagents were obtained through the AIDS Research and Reference Program, Division of AIDS, NIAID, NIH: pCTatBL43.CS and pETatBL43.CS from Dr. Udaylumar Ranga. This work was supported by the Intramural Research Program at the National Institute on Drug Abuse and DP1 DA028869 to JK.
Author information
Authors and Affiliations
Corresponding author
Additional information
Emily S. Wires and David Alvarez contributed equally to this manuscript.
Rights and permissions
About this article
Cite this article
Wires, E.S., Alvarez, D., Dobrowolski, C. et al. Methamphetamine activates nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) and induces human immunodeficiency virus (HIV) transcription in human microglial cells. J. Neurovirol. 18, 400–410 (2012). https://doi.org/10.1007/s13365-012-0103-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13365-012-0103-4