Summary
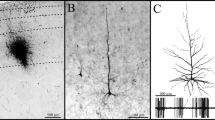
An analysis has been made of the morphology of axons in the geniculocortical pathway of turtles using the anterograde transport of horseradish peroxidase in both in vivo and in vitro preparations. Following injections of HRP into the dorsolateral thalamus, labeled axons could be traced from the dorsal lateral geniculate complex to the telencephalon. They are unbranched and free of varicosities within the diencephalon. They travel in the dorsal peduncle of the lateral forebrain bundle, through the basal telencephalon and dorsally into the pallial thickening. Many axons are situated deep in the pallial thickening and bear numerous varicosities that often appear apposed to the proximal dendrites or somata of neurons retrogradely labeled by thalamic injections of horseradish peroxidase. Individual axons continue from the pallial thickening into the dorsal cortex where they shift dorsally and bear varicosities as they course from lateral to medial in the superficial third of layer 1. These data indicate that the terminal zone of the dorsal lateral geniculate complex within the telencephalon of turtles is more extensive in the mediolateral direction than previously believed. Geniculate axons bear varicosities both within the pallial thickening as well as the dorsal cortex, but have different relationships to potential postsynaptic elements in the two areas. Geniculocortical axons overlie somata and proximal dendrites of neurons in the pallial thickening, but intersect the distal dendrites of neurons in the dorsal cortex.
Similar content being viewed by others
References
Adams JC (1977) Technical considerations in the use of horseradish peroxidase as a neuronal marker. Neurosci 2:141–147
Balaban CD (1978) Structure of the pallial thickening in turtles (Pseudemys scripta elegans). Anat Rec 190:330–331
Balaban CD (1979) Organization of thalamic projections to anterior dorsal ventricular ridge in two species of turtles (Pseudemys scripta elegans and Chrysemys picta belli). PhD dissertation, University of Chicago
Balaban CD, Ulinski PS (1981 a) Organization of thalmic afferents to anterior dorsal ventricular ridge in turtles. I. Projections of thalamic nuclei. J Comp Neurol 200:95–130
Balaban CD, Ulinski PS (1981 b) Organization of thalamic afferents to anterior dorsal ventricular ridge in turtles. II. Properties of the rotundo-dorsal map. J Comp neurol 200:131–150
Bass AH, Andry ML, Northcutt RG (1983) Visual activity in the telencephalon of the painted turtle, Chrysemys picta. Brain Res 263:201–210
Bass AH, Northcutt RG (1981 a) Retinal recipient nuclei in the painted turtle, Chrysemys picta: an autoradiographic and HRP study. J Comp Neurol 199:97–112
Bass AH, Northcutt RG (1981 b) Primary retinal targets in the Atlantic loggerhead sea turtle, Caretta caretta. Cell Tissue Res 218:253–264
Belekhova MG, Kosareva AA, Veselkin NP, Ermakova TV (1979) Telecephalic afferent connections in the turtle Emys orbicularis: a peroxidase study. J Evol Biochem Physiol 15:97–103
Conley M, Fitzpatrick D, Diamond IT (1984) The laminar organization of the lateral geniculate body and the striate cortex in the tree shrew (Tupaia glis). J Neurosci 4:171–197
Davydova TV, Goncharova NV (1979) Comparative characterization of the basic forebrain cortical zones in Emys orbicularis (Linnaeus) and Testudo horsfieldi (Gray). J Hirnforsch 20:245–262
Desan PH (1984) The organization of the cerebral cortex of the pond turtle, Pseudemys scripta elegans. Ph D Dissertation, Harvard University
Ebner FF, Colonnier M (1975) Synaptic patterns in the visual cortex of turtles: an electron microscopic study. J Comp Neurol 160:51–80
Ferster D, LeVay S (1978) The axonal arborizations of lateral geniculate neurons in the striate cortex of the cat. J Comp Neurol 182:923–944
Gaidenko GV (1978) Efferent connections of the dorsal cortex in tortoises. J Evol Biochem Physiol 13:268–270
Guselnikov VI, Morenkov ED, Pivavarov AS (1972) Unit responses of the turtle forebrain to visual stimuli. Neurosci Behav Physiol 5:235–242
Hall JA, Foster RE, Ebner, FF, Hall WC (1977) Visual cortex in a repitle, the turtle (Pseudemys scripta and Chrysemys picta). Brain Res 130:197–216
Hall WC, Ebner FF (1970a) Parallels in the visual afferent projections of the thalamus in the hedgehog (Parechinus hypomelas) and the turtle (Pseudemys scripta). Brain Behav Evol 3:135–154
Hall WC, Ebner FF (1970b) Thalamotelencephalic projections in the turtle (Pseudemys scripta). J Comp Neurol 140:101–122
Humphrey AL, Sur M, Uhlrich DJ, Sherman SM (1985a) Projection patterns of individual X- and Y-cell axons from the lateral geniculate nucleus to cortical areas 17 in the cat. J Comp Neurol 233:159–189
Humphrey AL, Sur M, Uhlrich DJ, Sherman SM (1985b) Termination patterns of individual X- and Y-cell axons in the visual cortex of the cat: Projections to area 18, to the 17/18 border region, and to both areas 17 and 18. J Comp Neurol 233:190–212
Johnston JB (1915) The cell masses in the forebrain of the turtle Cistudo carolina. J Comp Neurol 25:393–468
Kicliter E (1979) Some telencephalic connections in the frog, Rana pipiens. J Comp Neurol 185:75–86
Knapp H, Kang DS (1968a) The retinal projections of the sidenecked turtle (Podocnemis unifilis) with some notes on the possible origin of the pars dorsalis of the lateral geniculate body. Brain Behav Evol 1:369–404
Knapp H, Kang DS (1968 b) The visual pathways of the snapping turtle (Chelydra serpentina). Brain Behav Evol 1:19–42
Kosareva AA (1967) Projection of optic fibers to visual centers in a turtle (Emys orbicularis). J Comp Neurol, 130:263–276
Künzle H, Schnyder H (1983) Do retinal and spinal projections overlap within the turtle thalamus? Neurosci 10:161–168
Mason CA, Gregory E (1984) Postnatal maturation of cerebellar mossy and climbing fibers: transient expression of dual features on single axons. J Neurosci 4:1715–1735
Mazruskaya PZ (1973) Retinal projection in the forebrain of Emys orbicularis. Neurosci Behav Physiol 6:75–82
Mazurskaya PZ (1974) Organization of receptive fields in the forebrain of Emys orbicularis. Neurosci Behav Physiol 7:311–318
Mazurskaya PZ, Davydova TV, Smirnov GD (1967) Functional organization of exteroceptive projections in the forebrain of the turtle. Neurosci Trans 1:109–117
Mori K, Nowycky MC, Shepherd GM (1981) Electrophysiological analysis of mitral cells in the isolated turtle olfactory bulb. J Physiol (London) 314:281–294
Northcutt RG (1970) The telencephalon of the western painted turtle, Chrysemys picta belli. Ill. Biol Mongr No 43, Univ Illinois Press, Urbana
Orrego F (1961) The reptilian forebrain. I. The olfactory pathways and cortical areas in the turtle. Arch Ital Biol 99:425–445
Ouimet CC, Patrick RL, Ebner FF (1981) An ultrastructural and biochemical analysis of norepinephrine-containing varicosities in the cerbral cortex of the turtle Pseudemys. J Comp Neurol 195:289–304
Ourmet CC, Patrick RL, Ebner FF (1985) The projection of three extrathalamic cell groups to the cerebral cortex of the turtle Pseudemys. J Comp Neurol 237:77–84
Parent A, Poitras D (1974). The origin and distribution of catecholaminergic axon terminals in the cerebral cortex of the turtle (Chrysemys picta.) Brain Res 78:345–358
Kainey WT, Jones EG (1983) Spatial distribution of individual medial lemniscal axons in the thalamic ventrobasal complex of the cat. Exp Brain Res
Rainey WT, Ulinski PS (1986) Morphology of neurons in the dorsal lateral geniculate complex in turtles of the genera Pseudemys and Chrysemys. J Comp Neurol in press
Reiner A, Brauth SE, Kitt CA, Karten HJ (1980) Basal ganglionic pathways to the tectum: studies in reptiles. J Comp Neurol 193:565–589
Revzin AM, Karten HJ (1967) Rostral projections of the optic tectum and the nucleus rotundus in the pigeon. Brain Res 3:264–276 J. Comp Neurol 225:193–200
Robson JA, Mason CA (1979) The synaptic organization of terminals traced from individual labeled retino-geniculate axons in the cat. Neurosci 4:99–112
Semba K, Masarachia P, Malamed S, Jacquin M, Harris S, Yang G, Egger MD (1983) An electron microscopic study of primary afferent terminals from slowly adapting type I receptors in the cat. J Comp Neurol 221:446–481
Sjöström A, Ulinski PS (1985) Morphology of retinogeniculate terminals in the turtle, Pseudemys scripta elegans. J Comp Neurol 238:107–120
Smith LM, Ebner FF, Colonnier M (1980) The thalamocortical projection in Pseudemys turtles: a quantiative electron microscope study. J Comp Neurol 190:445–462
Ulinski PS (1981) Thick caliber projections from bainstem to cerebral cortex in the snakes Thamnophis sirtalis and Natrix sipedon. Neuroscience 6:1725–1743
Ulinski PS (1983) Dorsal ventricular ridge: a treatise on forebrain organization in reptiles and birds. John Wiley, New York
Ulinski PS (1987) Ultrastructure of the dorsal lateral geniculate complex in turtles of the genera Pseudemys and Chrysemys. Brain Behav Evol, in press
Ulinski PS, Nautiyal J (1986) Organization of the retinogeniculate projection in turtles of the genera Pseudemys and chrysemys, submitted for publication
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Heller, S.B., Ulinski, P.S. Morphology of geniculocortical axons in turtles of the genera Pseudemys and Chrysemys . Anat Embryol 175, 505–515 (1987). https://doi.org/10.1007/BF00309685
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00309685