Abstract
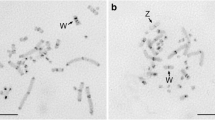
A number of repetitive DNA clones were generated from PCR amplifications of Indian muntjac genomic DNA using primer sequences derived from a white tailed deer satellite II DNA sequence. One clone (Mmv-0.7) was characterized and shown to be a cervid satellite II DNA clone. Multiple colored FISH studies with cervid satellite I (C5) and this satellite II clone (Mmv-0.7) to Chinese muntjac metaphase chromosomes localized both satellite DNAs at the pericentromeric regions of all chromosomes except for chromosome 3 and the Y chromosome, whereas chromosome 3 exhibited pericentromeric satellite II DNA only. Where distinguishable, the pericentromeric satellite II signals appeared terminally oriented with respect to satellite I. Six pairs of Chinese muntjac autosomes had interstitial satellite I sites with four of these autosomal pairs (chromosomes 1, 2 and two other smaller autosomal pairs) also exhibiting interstitial satellite II signals. An interstitial site on the X chromosome was found to have satellite II signals. For the Indian muntjac chromosomes, FISH studies revealed a pericentromeric hybridization for satellites I and II as well as 27 distinct interstitial hybridization sites, each having at least one of the satellite DNAs. These data were used to more precisely define the chromosome fusion-associated breakpoints that presumably led to the formation of the present-day Indian muntjac karyotype. It further hints at the possibility that the Indian muntjac karyotype may have evolved directly from a 2n=70 ancestral karyotype rather than from an intermediate 2n=46 Chinese muntjac-like karyotype.
Similar content being viewed by others
References
Bogenberger JM, Neumaier PS, Fittler F (1985) The Muntjak satellite 1A sequence is composed of 31–bp-pair internal repeats that are highly homologous to the 31–base-pair subrepeats of the bovine satellite 1.715. Eur J Biochem 148: 55–59.
Csink AK, Henikoff S (1998) Something from nothing: the evolution and utility of satellite repeats. Trends Genet 14: 200–204.
D'Aiuto L, Barsanti P, Mauro S, Cserpan I, Lanave C, Ciccarese S (1997) Physical relationship between satellite I and II DNA in centromeric regions of sheep chromosomes. Chromosome Res 5: 375–381.
Fontana F, Rubini M (1990) Chromosomal evolution in Cervidae. BioSystems 24: 157–174.
Frönicke L, Scherthan H (1997) Zoo-fluorescence in situ hybridization analysis of human and Indian muntjac karyotypes (Muntiacus muntjak vaginalis) reveals satellite DNA clusters at the margins of conserved syntenic segments. Chromosome Res 5: 254–261.
Hsu TC, Pathak S, Chen TR (1975) The possibility of latent centromeres and a proposed nomenclature for total chromosome and whole arm translocations. Cytogenet Cell Genet 15: 41–49.
Johnston FP, Church RB, Lin CC (1982) Chromosome rearrangement between the Indian muntjac and Chinese muntjac is accompanied by a deletion of middle repetitive DNA. Can J Biochem 60: 497–506.
Lan H, Wang W, Shi LM (1995) Phylogeny of Muntiacus (Cervidae) based on mitochondrial DNA restriction maps. Biochem Genet 33: 377–388.
Lee C, Lin CC (1996) Conservation of a 31–bp bovine subrepeat in centromeric satellite DNA monomers of Cervus elaphus and other cervid species. Chromosome Res 4: 428–436.
Lee C, Sasi R, Lin CC (1993) Interstitial localization of telomeric DNA sequences in the Indian muntjac chromosomes: further evidence for tandem chromosome fusions in the karyotypic evolution of the Asian muntjacs. Cytogenet Cell Genet 63: 156–159.
Lee C, Ritchie DBC, Lin CC (1994) A tandemly repetitive, centromeric DNA sequence from the Canadian woodland caribou (Rangifer tarandus caribou): its conservation and evolution in several deer species. Chromosome Res 2: 293–306.
Lee C, Court DR, Cho C, Haslett J, Lin CC (1997) Higher-order organization of subrepeats and the evolution of cervid satellite I DNA. J Mol Evol 44: 327–335.
Lee C, Griffin DK, O'Brien PC, Yang F, Lin CC, Ferguson-Smith MA (1998) Defining the anatomy of the Rangifer tarandus sex chromosomes. Chromosoma 107: 61–69.
Lee C, Stanyon R, Lin CC, Ferguson-Smith MA (1999) Conservation of human gamma-X centromeric satellite DNA among primates with an autosomal localization in certain Old World monkeys. Chromosome Res 7: 43–47.
Lima-de-Faria A (1980) How to produce a human with 3 chromosomes and 1000 primary genes. Hereditas 93: 47–73.
Lin CC, Sasi R, Fan Y-S, Chen Z-Q (1991) New evidence for tandem chromosome fusions in the karyotypic evolution of Asian muntjacs. Chromosoma 102: 333–339.
Maniatis T, Fritsch EF, Sambrook J (1982) Molecular Cloning: a Laboratory Manual. New York: Cold Spring Harbor Laboratory.
Murphy TD, Karpen GH (1998) Centromeres take flight: alpha satellite and the quest for the human centromere. Cell 93: 317–320.
Neitzel H (1987) Chromosome evolution of cervidae: Karyotypic and molecular aspects. In: Obe G, Basler A, eds. Cytogenetics. Berlin: Springer-Verlag, pp 90–112.
Ohno S (1967) Sex Chromosomes and Sex-linked Genes. New York: Springer-Verlag.
Plucienniczak A, Skowronski J, Jaworski J (1982) Nucleotide sequence of bovine 1.715 satellite DNA and its relation to other bovine satellite sequences. J Mol Biol 158: 293–304.
Qureshi SA, Blake RD (1995) Sequence characteristics of a cervid DNA repeat family. J Mol Evol 40: 400–404.
Scherthan H (1995) Chromosome evolution in muntjac revealed by centromere, telomere and whole chromosome paint probes. Kew Chromosome Conference IV: 267–280.
Shi L, Ye Y, Duan X (1980) Comparative cytogenetic studies on the red muntjac. Chinese muntjac and their F1 hybrids. Cytogenet Cell Genet 26: 22–27.
Vafa O, Shelby RD, Sullivan KF (1999) CENP-A associated complex satellite DNA in the kinetochore of the Indian muntjac. Chromosoma 108: 367–374.
Whitehead GK (1993) The Whitehead Encyclopedia of Deer. Shrewsbury, England: Swan Hill Press, pp 238–242, 313–314.
Wurster DH, Benirschke K (1967) Chromosome studies in some deer, the springbox, and the pronghorn, with notes on placentation in deer. Cytologia 32: 273–285.
Wurster DH, Benirschke K (1970) Indian muntjac, Muntiacus muntjak: a deer with a low diploid chromosome number. Science 168: 1364–1366.
Yang F, Carter NP, Shi L, Ferguson-Smith MA (1995) A comparative study of karyotypes of muntjacs by chromosome painting. Chromosoma 103: 642–652.
Yang F, O'Brien PCM, Wienberg J, Neitzel H, Lin CC, Ferguson-Smith MA (1997a) Chromosomal evolution of the Chinese muntjac (Muntiacus reevesi). Chromosoma 106: 37–43.
Yang F, O'Brien PCM, Wienberg J, Ferguson-Smith MA (1997b) A reappraisal of the tandem fusion theory of karyotype evolution in the Indian muntjac using chromo-some painting. Chromosome Res 5: 109–117.
Yu LC, Lowensteiner D, Wong EFK, Sawada I, Mazrimas J, Schmid C (1986) Localization and characterization of recombinant DNA clones derived from the highly repetitive DNA sequences in the Indian muntjac cells: Their presence in the Chinese muntjac. Chromosoma 93: 521–528.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Li, YC., Lee, C., Sanoudou, D. et al. Interstitial colocalization of two cervid satellite DNAs involved in the genesis of the Indian muntjac karyotype. Chromosome Res 8, 363–373 (2000). https://doi.org/10.1023/A:1009203518144
Issue Date:
DOI: https://doi.org/10.1023/A:1009203518144