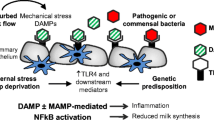
Abstract
The primary phagocytic cells of the bovine mammary gland, polymorphonuclear neutrophil leukocytes (PMN), and macrophages, comprise the first line of defense against invading pathogens. In the normal healthy mammary gland, macrophages predominate and act as sentinels to invading mastitis-causing pathogens. Once invaders are detected, macrophages, and possibly mammary epithelial cells, release chemoattractants that direct migration of PMN into the area. In the mammary gland, protection is only effective if rapid influx of PMN from the circulation and subsequent phagocytosis and killing of bacteria occur. The second line of defense against infection consists of a network of memory cells and immunoglobulins that interact with the first line of defense. To minimize mammary tissue damage caused by bacterial toxins and oxidative products released by PMN, elimination of invading bacteria must proceed quickly. Therefore, the inflammatory response needs to be regulated. Hormones, metabolites, and acute phase proteins act to influence the outcome of mastitis, especially around parturition. The number of circulating PMN in cows during early lactation is highly heritable and closely related to susceptibility to clinical mastitis at this time. Advances in molecular biology are making available the tools, techniques, and products to study and modulate host–pathogen interactions. For example, the cloning and expression of proteins such as recombinant bovine soluble (rbos) CD (cluster of differentiation) 14 antigens, may provide ways of minimizing damaging effects of endotoxin during acute coliform mastitis. Soluble CD14 binds and neutralizes lipopolysacharide (LPS) and causes local recruitment of PMN after binding of CD14-LPS complexes to mammary epithelial cells. Development of transgenic animals that express rbosCD14 in their milk could prevent infection by Gram-negative pathogens.
Similar content being viewed by others
REFERENCES
M. J. Paape and W. P. Wergin (1977). Scanning and transmission electron microscopy of polymorphonuclear leukocytes isolated from milk. Fed.Proc. 36: 1201–1205.
M. J. Paape and W. P. Wergin (1977). The leukocyte as a defense mechanism. J.Am.Vet.Med.Assoc. 170: 1214–1223.
M. J. Paape, W. P. Wergin, A. J. Guidry, and R. E. Pearson (1979). Leukocytes--the second line of defense against invad-ing mastitis pathogens. J.Dairy Sci. 62: 135–153.
G. A. Zimmerman (1992). Endothelial cell interaction with granulocyte: Tethering and signaling molecules. Immunol. Today 13: 93–112.
T. K. Kishimoto, M. A. Jutila, E. L. Berg, and E. C. Butcher (1989). Neutrophil Mac-1 and MEL-14 adhesion proteins in-versely regulated by chemotactic factors. Science 245: 1238–1241.
M. J. Paape, A. J. Guidry, N. C. Jain, and R. H. Miller (1991). Leukocytic defense mechanisms in the udder. Flemish Vet.J. 1:(Suppl.)95–109.
J. Savill (1997). Recognition and phagocytosis of cells undergo-ing apoptosis. Br.Med.Bull. 53: 491–508.
R. B. Gennaro, B. Dewald, U. Horisberger, H. U. Gubler, and M. A. Baggiolini (1983). A novel type of cytoplasmic granule in bovine neutrophils. J.Cell Biol. 96: 1651–1661.
S. J. Klebanoff (1970). Myeloperoxidase--mediated antimicro-bial systems and their role in leukocyte function. In Biochemistry of the Phagocytic Process, North-Holland Publishing, London, pp. 89–114.
M. J. Paape, H. D. Hafs, and W. W. Snyder (1963). Variation of estimated numbers of milk somatic cells stained with Wright's stain or pyronin-Y-methyl green stain. J.Dairy Sci. 46:1211–1216.
K. Östensson, M. Hageltorn, and G. Aström (1988). Differen-tial cell counting in fraction-collected milk from dairy cows. Acta Vet.Scand. 29: 493–500.
W. Suriyasathaporn, Y. H. Schukken, M. Nielen, and A. Brand (2000). Low somatic cell count: A risk factor for subsequent clinical mastitis in a dairy herd. J.Dairy Sci. 83: 1248–1255.
M. J. Paape, G. Ziv, R. H. Miller, and W. D. Schultze (1986). Update on the use of intramammary devices in the control of mastitis. In Proceedings of the 25th Annual Meeting of the National Mastitis Council, Inc., Columbus, Ohio, pp. 87–103.
G. Vandeputte-Van Messom, C. Burvenich, E. Roets, L. Devriese, and F. Haesebrouck (1995). Effects of Staphylococ-cus aureus mastitis after endotoxin application on milk yield and composition during subsequent lactation of Guinea-pigs. J.Vet.Med. 2: 118–126.
C. Burvenich, M. J. Paape, A. W. Hill, A. J. Guidry, R. H. Miller, R. Heyneman, W. D. J. Kremer, and A. Brand (1994). Role of the neutrophil leukocyte in the local and systemic reactions dur-ing experimentally induced E.coli mastitis in cows immediately after calving. Vet.Q. 16: 45–50.
S. C. Nickerson and C. W. Heald (1981). Histopathologic response of the bovine mammary gland to experimentally induced Staphylococcus aureus infection. Am.J.Vet.Res. 42: 1351–1354.
A. V. Capuco, M. J. Paape, and S. C. Nickerson (1986). In vitro study of polymorphonuclear leukocyte damage to mammary tissues of lactating cows. Am.J.Vet.Res. 47: 663–668.
R. M. Akers and W. Thompson (1987). Effect of induced leuko-cyte migration on mammary cell morphology and milk compo-nent biosynthesis. J.Dairy Sci. 70: 1685–1695.
J. K. Miller, E. Brzezinska-Slebodzinska, and F. C. Madsen (1993). Oxidative stress, antioxidants and animal function. J. Dairy Sci. 76: 2812–2823.
C. Riollet, P. Rainard, and B. Poutrel (2000). Differential in-duction of complement fragment C5a and inflammatory cy-tokines during intramammary infections with Escherichia coli and Staphylococcus aureus. Clin.Diagn.Lab.Immunol. 7: 161–167.
D. J. Granville, C. M. Carthy, D. W. C. hunt, and B. M. McManus (1998). Apoptosis: Molecular aspects of cell death and disease. Lab.Invest. 78: 893–913.
K. Van Oostveldt, G. M. Tomita, M. J. Paape, and C. Burvenich (2002). Apoptosis of neutrophils during Eschericha coli and endotoxin mastitis. Am.J.Vet.Res. 63: 448–453.
D. F. Bainton, J. L. Ullyot, and M. G. Farquahar (1971). The development of neutrophilic polymorphonuclear leukocytes in human bone marrow. J.Exp.Med. 134: 907–934.
G. P. Carlson and J. J. Kaneko (1975). Intravascular granulocyte kinetics in developing calves. Am.J.Vet Res. April: 421–425.
N. Manlongat, T. J. Yang, L. S. Hinckley, R. B. Bendel, and H. M. Krider (1998). Physiologic-chemoattractant-induced migration of polymorphonuclear leukocytes in milk. Clin.Diagn.Lab. Immunol. 5: 375–381.
S. Struyf, P. Proost, J. J. Lenaerts, G. Stoops, A. Wuyts, and J. V. Damme (2001). Identification of a blood-derived chemoattac-tant for neutrophils and lymphocytes as a novel CCchemokine, Regakine-1. Blood 97: 2197–2204.
D. D. Taubb and J. J. Oppenheim (1993). Review of the chemokine meeting: Third International Symposium of Chemotactic Cytokines. Cytokine 5: 175–179.
G. D. Gray, K. A. Knight, R. D. Nelson, and M. J. Herron (1982). Chemotactic requirements of bovine leukocytes. Am.J.Vet. Res. 43: 757–759.
J. Lee and X. Zhao (2000). Recombinant human interleukin-8, but not human interleukin-1¯, induces bovine neutrophil migration in an in vitro co-culture system. Cell Biol.Int. 24: 889–895.
M. J. Daley, P. A. Coyle, T. J. Williams, G. Furda, R. Dougherty, and P. W. Hayes (1991). Staphylococcus aureus mastitis: Pathogenesis and treatment with bovine interleukin 1B and interleukin–2. J.Dairy Sci. 74: 4413–4424.
C. Burvenich, M. J. Paape, D. Hoeben, H. Dosogne, A. M. Massart-Le¨ en, and J. Blum (1999). Modulation of the inflam-matory reaction and neutrophil defense of the bovine lac-tating mammary gland by growth hormone. Domest. Anim. Endocrinol. 17: 149–159.
I. Politis, X. Zhao, B. W. McBride, and J. H. Burton (1992). Function of bovine mammary macrophages as antigen present-ing cells. Vet.Immunol.Immunopath. 30: 399–410.
I. Politis, B. W. McBride, J. H. Burton, X. Zhao, and J. D. Turner (1991). Limited secretion of interleukin-1 by bovine mammary gland macrophages. Am.J.Vet.Res. 52: 858–862.
H. Okada, H. Ohtsuka, S. Kon-Nai, R. Kirsawa, Y. Yokomizo, T. Yoshino, and T. Rosol (1999). Effects of lipopolysaccha-ride on production of interleukin-1 and interleukin-6 by bovine mammary epithelial cells in vitro. J.Vet.Med.Sci. 61: 33–35.
N. Boudjellab, H. S. Chan-Tang, and X. Zhao (2000). Bovine interleukin-1 expression by cultured mammary epithelial cells (MAC-T) and its involvement in the release of MAC-T derived interleukin-8. Comp.Physiol.Biochem. 127: 191–199.
N. Boudjellab, H. S. Chan-Tang, X. Li, and X. Zhao (1998). Bovine mammary epithelial cells secrete interleukin-8 in re-sponse to lipopolysaccharide stimulation. Am.J.Vet.Res. 59: 1563–1567.
M. R. Barber, A. G. Pantschenko, L. S. Hinckley, and T. J. Yang (1999). Inducible and constitutive in vitro neutrophil chemokine expression by mammary epithelial and myoepithe-lial cells. Clin.Diagn.Lab.Immunol. 6: 791–798.
M. J. Paape and A. J. Guidry (1969). Effect of milking on leuko-cytes in the subcutaneous abdominal vein of the cow. J.Dairy Sci. 52: 998–1002.
M. J. Paape, W. D. Schultze, A. V. Capuco, and G. As-trom (1985). Trafficking of leukocytes in the lactating bovine. mammary gland. Keiler Milchwirtschaftliche Forschungs-berichte 37: 622–623.
O. W. Schalm and J. Lasmanis (1976). Cytological features of bone marrow in normal and mastitic cows. J.Am.Vet.Med. Assoc. 37: 359–363.
M. J. Paape, A. J. Guidry, S. T. Kirk, and D. J. Bolt (1975). Measurement of phagocytosis of 32 P-labeled Staphylococcus aureus by bovine leukocytes: Lysostaphin digestion and in-hibitory effect of cream. Am.J.Vet. Res. 36: 1737–1743.
M. J. Paape and A. J. Guidry (1977). Effect of fat and casein on intracellular killing of Staphylococcus aureus by milk leuko-cytes. Proc.Soc.Exp.Bio.Med. 155: 588–593.
S. P. Targowski and M. Niemialtowski (1986). Appearance of Fc receptors on polymorphonuclear leukocytes after migration and their role in phagocytosis. Infect.Immunol. 52: 798–802.
M. Worku, M. J. Paape, R. Filep, and R. H. Miller (1994). Ef-fect of in vitro and in vivo migration of bovine neutrophils on binding and expression of Fc receptors for IgG2 and IgM. Am. J.Vet Res. 55: 221–226.
L. M. Berning, M. J. Paape, and R. R. Peters (1993). Functional variation in endogenous and exogenous immunoglobulin bind-ing to bovine neutrophils relative to parturition. Am.J.Vet.Res. 54: 1145–1153.
A. L. DiCarlo, M. J. Paape, and R. H. Miller (1996). Reactivity of purified complement component 3b with bovine neutrophils and modulation of complement receptor 1. Am.J.Vet.Res. 57: 151–156.
L. Leino and M. J. Paape (1993). Comparison of the chemilu-minescence responses of bovine neutrophils to differently op-sonized zymosan particles. Am.J.Vet.Res. 54: 1055–1059.
C. Burvenich, A. J. Guidry, A. V. Capuco, and M. J. Paape (1996). The influence of neutrophils and systemic diseases on bovine mastitis during early lactation. In J. W. Blum and R. M. Bruckmaier (eds), Milk Synthesis, Secretion and Removal in Ruminants. University of Berne, Switzerland, pp. 123–130.
H. Dosogne, C. Burvenich, and M. J. Paape (1998). Effect of extracellular ionic calcium and magnesium on opsonic and non-opsonic phagocytosis of Escherichia coli by bovine blood poly-morphonuclear leucocytes. Comp.Haematol.Int. 8: 82–86.
M. J. Paape, E. M. Lillius, P. A. Wiitanen, and M. P. Kontio (1996). Intramammary defense against infections induced by Escherichia coli in cows. Am.J.Vet.Res. 57: 477–482.
L. J. Old (1988). Tumor necrosis factor. Sci.Am. 258: 59–75.
A. Haziot, E. Ferrero, M. G. Low, R. Selber, and S. M. Goyert (1988). The monocyte diffferentiation antigen, CD14, is an-chored to the cell membrane by a phosphatidylinositol linkage. J.Immunol. 141: 547–552.
V. Bazil, V. Horejsi, M. Baudys, H. Kristofova, J. L. Strominger, E. Kostka, and I. Hilgert (1986). Biochemical characterization of a soluble form of the 53-kDa monocyte surface antigen. Eur. J.Immunol. 16: 1583–1589.
D. Filipp, K. Alizadeh-khiavi, C. Richardson, A. Palma, N. Paredes, O. Takeuchi, S. Akira, and M. Julius (2001). Soluble CD14 enriched in colostrum and milk induces B cell growth and differentiation. Proc.Natl. Acad.Sci. 98: 603–608.
Y. Wang, D. S. Zarlenga, M. J. Paape, and G. E. Dahl (2002). Re-combinant bovine soluble CD14 sensitizes the mammary gland to lipopolysaccharide. Vet.Immunol.Immunopathol. 86: 115–124.
R. J. Ulevitch and P. S. Tobias (1999). Recognition of Gram-negative bacteria and endotoxin by the innate immune system. Curr.Opin.Immunol. 11: 19–22.
M. E. Kehrli, B. J. Nonnecke, and J. A. Roth (1989). Alterations in bovine neutrophil function during the periparturient period. Am.J.Vet.Res. 50: 207–214.
D. Hoeben, E. Monfardini, G. Opsomer, H. Dosogne, A. De Kruif, J. F. Beckers, and C. Burvenich (2000). Chemilumi-nescence of bovine polymorphonuclear leukocytes during the periparturient period and relation with metabolic parameters and bovine pregnancy-associated glycoprotein. J.Dairy Res. 67: 249–259.
D. E. Shuster, E. K. Lee, and M. E. Kehrli (1996). Bacterial growth, inflammatory cytokine production, and neutrophil re-cruitment during coliform mastitis in cows within ten days after calving compared with cows at mid lactation. Am.J.Vet.Res. 57: 1569–1576.
H. Dosogne, F. Vangroenweghe, B. Barrio, P. Rainard, and C. Burvenich (2001). Decreased number and bactericidal activity against Staphylococcus aureus of milk-resident cells of dairy cows during early lactation. J.Dairy Res. 68: 539–549.
J. Mehrzad, H. Dosogne, E. Meyer, R. Heyneman, and C. Burvenich (2001). Respiratory burst activity of blood and milk neutrophils in dairy cows during different stages of lacta-tion. J.Dairy Res. 68: 399–415.
E. K. Lee and M. Kehrli (1998). Expression of adhesion molecules on neutrophils of periparturient cows and neonatal calves. Am.J.Vet.Res. 59: 37–43.
E. Monfardini, M. J. Paape, Y. Wang, A. V. Capuco, M. Husheem, L. Wood, and C. Burvenich (2002). Evaluation of L-selectin expression and assessment of protein tyrosine phos-phorylation in bovine polymorphonuclear neutrophil leuko-cytes around parturition. Vet.Res. 33: 1–5.
J. C. Detilleux, K. J. Koehler, A. E. F. Freeman, M. E. Kehrli, and D. H. Kelley (1994). Immunological parameters of peripar-turient Holstein cattle: Genetic variation. J.Dairy Sci. 77: 2640–2645.
W. Suriyasathaporn, Y. H. Schukken, M. Nielen, and A. Brand (2000). Low somatic cell count: A risk factor for subse-quent clinical mastitis in a dairy herd. J.Dairy Sci. 83: 1248–1255.
A. M. Dulin, M. J. Paape, and S. C. Nickerson (1988). Com-parison of phagocytosis and chemiluminescence by blood and mammary gland neutrophils from multiparous and nulliparous cows. Am.J.Vet.Res. 49: 172–177.
J. Mehrzad, H. Dosogne, E. Meyer, and C. Burvenich (2001). Local and systemic effects of endotoxin mastitis on the chemi-luminescence of milk and blood neutrophils in dairy cows. Vet. Res. 32: 131–144.
J. Mehrzad, H. Dosogne, F. Vangroenweghe, and C. Burvenich (2001). A comparative study of bovine blood and milk neu-trophils functions with luminol dependent chemiluminescence. Luminescence 16: 343–356.
L. Weber, E. Peterhans, and R. Wyler (1983). The chemilu-minescent response of bovine polymorphonuclear leukocytes isolated from milk and blood. Vet.Immunol.Immunopathol 4: 397–412.
R. C. Allen, R. L. Stjernholm, and R. H. Steele (1972). Evi-dence for the generation of an electronic excitation state(s) in human polymorphonuclear leukocytes and its participation in bactericidal activity. Biochem.Biophys.Res.Commun. 47: 679–684.
L. S. Webb, B. B. Keele, and R. B. Johnston Jr (1974). Inhibition of phagocytosis-associated chemiluminescence by superoxide dismutase. Infect.Immun. 9: 1051–1056.
C. Hallèn Sandgren, K. Nordling, and I. Björk (1991). Isolation and phagocytotic properties of neutrophils and other phago-cytes from nonmastitic bovine milk. J.Dairy Sci. 74: 2965–2975.
E. Smits, C. Burvenich, A. J. Guidry, R. Heyneman, and A. Massart-Leen (1999). Diapedesis across mammary epithelium reduces phagocytotic and oxidative burst of bovine neutrophils. Vet.Immunol.Immunopathol. 68: 169–176.
A. Karlsson, J. B. Nixon, and L. C. Mcphail (2000). Phorbol myristate acetate induces neutrophils NADPH-oxidase activity by two separate signal transduction pathways: Dependent or independent of phosphatidylinositol 3-kinase. J.Leukoc.Biol. 67: 396–404.
K. Van Oostveldt, F. Vangroenweghe, H. Dosogne, and C. Burvenich (2001). Appoptosis and necrosis of blood and milk polymophonuclear leukocytes in early and mid lactating healthy cows. Vet.Res. 32: 617–622.
A. Jankowski, C. C. Scott, and S. Grinstein (2002). Deter-minants of phagosomal pH in neutrophils. J.Biol.Chem. 277: 6059–6066.
S. J. Mayer, P. M. Keen, N. Craven, and F. J. Bourn (1989). Regulation of phagolysosome pH in bovine and human neu-trophils: The role of NADPH oxidase activity and an NaC/HC antiporter. J.Leukoc.Biol. 45: 239–224.
D. M. Reinitz, M. J. Paape, and I. H. Mather (1982). Effect of phagocytosed fat and casien on the intraphagosomal pH in bovine polymorphonuclear leukocytes. Soc.Exp.Biol.Med. 170: 281–285.
G. Biozzi, O. G. Ribeiro, A. Saran, M. L. Araujo, D. A. Maria, M. De Franco, W. K. Cabrera, O. A. Santanna, S. Massa, V. Covelli, D. Mouton, T. Neveu, M. Siqueira, and O. M. Ibanez (1998). Effect of genetic modification of acute inflammatory responsiveness on tumorigenesis in the mouse. Carcinogenesis 19: 337–346.
B. Wilkie and B. Mallard (1999). Selection for high immune response: An alternative approach to animal health mainte-nance? Vet.Immunol.Immunopath. 72: 231–235.
Bovine genome database of the national animal genome re-search programme (http://bos.cvm.tamu.edu/bovgbase.html) and Bovmap database (http://locus.jouy.inra.fr/cgi-bin/bovmap/intro.pl)
N. C. Franc, K. White, and R. A. B. Ezekpwitz (1999). Phago-cytosis and development: Back to the future. Curr.Opin. Immunol. 11: 47–52.
S. C. Kelm, J. C. Detilleux, A. E. Freeman, M. E. Kehrli, A. B. Dietz, L. K. Fox, J. E. Butler, I. Kasckovics, and D. H. Kelley (1997). Genetic association between parameters of innate im-munity and measures of mastitis in periparturient holstein cat-tle. J.Dairy Sci. 80: 1767–1775.
L. C. Wagter, B. A. Mallard, B. N. Wilkie, K. E. Leslie, P. J. Boettcher, and J. C. M. Dekkers (2000). A quantitative ap-proach to classifying Holstein cows based on antibody respon-siveness and its relationship to peripartum mastitis occurrence. J.Dairy Sci. 83: 488–498.
J. M. Wanner, G. W. Rogers, M. E. Kehrli, and J. B. Cooper (1999). Clinical mastitis in primiparous Holsteins: Compar-isons of bovine leukocyte adhesion deficiency carriers and non-carriers. J.Dairy Sci. 82: 2517–2525.
S. T. Qureshi, P. Gros, and D. Malo (1999). Host resistance to infection: Genetic control of lipopolysaccharide responsiveness by Toll-like receptor genes. Trends Genetics 15: 291–294.
F. D. Bastida-Corcuera, J. E. Butler, S. Yahiro, and L. B. Corbeil (1999). Differential complement activation by bovine IgG2 al-lotypes. Vet.Immunol.Immunopath. 71: 115–123.
H. Dosogne, C. Burvenich, T. van Werven, E. Roets, E. N. Noordhuizen-Stassen, and B. Goddeeris (1997). Increased sur-face expression of CD11b receptors on polymorphonuclear leukocytes is not sufficient to sustain phagocytosis during Escherichia coli mastitis in early postpartum dairy cows. Vet. Immunol.Immunopathol. 12: 47–59.
A. Belaaouaj, R. McCarthy, M. Baumann, Z. Gao, T. J. Ley, S. N. Abraham, and S. D. Shapiro (1998). Mice lacking neutrophil elastase reveal impaired host defense against gram negative bacterial sepsis. Nat.Med. 4: 615–619.
B. A. Mallard, J. C. Dekkers, M. J. Ireland, K. E. Leslie, S. Sharif, C. L. Vankampen, L. Wagter, and B. N. Wilkie (1998). Alteration in immune responsiveness during the peripartum period and its ramification on dairy cow and calf health. J.Dairy Sci. 81: 585–594.
C. Burvenich, H. Dosogne, J. DeTilleux, and T. Van Werven (1999). Is it possible to predict the severity of acute masti-tis during early lactation through measurement of the activity of circulating polymorphonuclear cells? In Journ´ ees Nationales GTV-INRA, 26–28 Mai 1999, Nantes, pp. 43–52.
B. Wilkie and B. Mallard (1999). Selection for high immune response: An alternative approach to animal health mainte-nance? Vet.Immunol.Immunopath. 72: 231–235.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Paape, M., Mehrzad, J., Zhao, X. et al. Defense of the Bovine Mammary Gland by Polymorphonuclear Neutrophil Leukocytes. J Mammary Gland Biol Neoplasia 7, 109–121 (2002). https://doi.org/10.1023/A:1020343717817
Issue Date:
DOI: https://doi.org/10.1023/A:1020343717817