Abstract
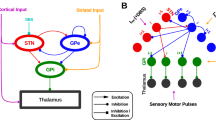
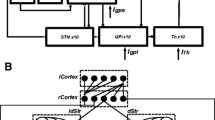
Deep brain stimulation (DBS) of the subthalamic nucleus (STN) or the internal segment of the globus pallidus (GPi) has recently been recognized as an important form of intervention for alleviating motor symptoms associated with Parkinson's disease, but the mechanism underlying its effectiveness remains unknown. Using a computational model, this paper considers the hypothesis that DBS works by replacing pathologically rhythmic basal ganglia output with tonic, high frequency firing. In our simulations of parkinsonian conditions, rhythmic inhibition from GPi to the thalamus compromises the ability of thalamocortical relay (TC) cells to respond to depolarizing inputs, such as sensorimotor signals. High frequency stimulation of STN regularizes GPi firing, and this restores TC responsiveness, despite the increased frequency and amplitude of GPi inhibition to thalamus that result. We provide a mathematical phase plane analysis of the mechanisms that determine TC relay capabilities in normal, parkinsonian, and DBS states in a reduced model. This analysis highlights the differences in deinactivation of the low-threshold calcium T-current that we observe in TC cells in these different conditions. Alternative scenarios involving convergence of thalamic signals in the cortex are also discussed, and predictions associated with these results, including the occurrence of rhythmic rebound bursts in certain TC cells in parkinsonian states and their drastic reduction by DBS, are stated. These results demonstrate how DBS could work by increasing firing rates of target cells, rather than shutting them down.
Similar content being viewed by others
References
Albin R, Young A, Penney J (1989) The functional anatomy of basal ganglia disorders. Trends Neurosci. 12: 366-375.
Anderson M, Postpuna N, Ruffo M (2003) effects of high-frequency stimulation in the internal globus pallidus on the activity of thalamic neurons in the awake monkey. J. Neurophysiol. 89: 1150-1160.
Ashby P (2000) What does stimulation in the brain actually do? Prog. Neurol. Surg. 15: 236-245.
Benabid A, Koudsie A, Benazzouz A, Piallat B, Krack P (2001) Deep brain stimulation for Parkinson's disease. Adv. Neurol. 86: 405-412.
Benabid A, Koudsie A, Benazzouz A, Piallat B, Krack P, Blerkom NV, Fraix V, Pollak P (2001) Subthalamic nucleus deep brain stimulation. In Lozano A. ed., Movement Disorder Surgery. S. Karger AG, Basel.
Benabid A, Koudsie A, Benazzouz A, Piallat B, Krack P, Limousin-Dowsey P, Lebas J, Pollak P (2001) Deep brain stimulation for Parkinson's disease. In: Calne D, Calne SM, eds., Advances in Neurology, Vol 86: Parkinson's Disease. Lippincott Williams & Wilkins, Philadelphia.
Benabid A, Koudsie A, Benazzouz A, Vercueil L, Fraix V, Chabardes S, Lebas J, Pollak P (2001) Deep brain stimulation of the corpus luysi (subthalamic nucleus) and other targets in parkinson's disease. Extension to new indications such as dystonia and epilepsy. J. Neurol. 248 (Supp. 3): S37-S47.
Benazzouz A, Gao D, Ni Z, Piallat B, Bouali-Benazzouz R, Benabid A (2000) Effect of high-frequency stimulation of the subthalamic nucleus on the neuronal activities of the substantia nigra pars reticulate and the ventrolateral nucleus of the thalamus. Neuroscience. 99: 289-295.
Benazzouz A, Hallett M (2000) Mechanisms of action of deep brain stimulation. Neurology, 55 (Supp. 6): S13-S16.
Benazzouz A, Piallat B, Pollack P, Benabid A (1995) Responses of the substantia nigra pars reticulata and globus pallidus complex to high frequency stimulation of the subthalamic nucleus in rats: Electrophysiological data. Neurosci. Lett. 189: 77-80.
Bergman H, Deuschl G (2002) Pathophysiology of Parkinson's disease: From clinical neurology to basic neuroscience and back. Mov. Disord. 17 (Suppl. 3): S28-S40.
Bergman H, Wichmann T, Karmon B, DeLong M(1994) The primate subthalamic nucleus. II. Neuronal activity in the MPTP model of parkinsonism. J. Neurophysiol. 72: 507-520.
Beurrier C, Bioulac B, Audin J, Hammond C (2001) High-frequency stimulation produces a transient blockade of voltage-gated currents in subthalamic neurons. J. Neurophysiol. 85: 1351-1356.
Bevan M, Wilson C (1999) Mechanisms underlying spontaneous oscillation and rhythmic firing in rat subthalamic neurons. J. Neurosci. 19: 7617-7628.
Bevan M, Wilson C, Bolam J, Magill P (2000) Equilibrium potential of GABA-A current and implications for rebound burst firing in rat subthalamic neurons in vitro. J. Neurophysiol. 83: 3169-3172.
Brown P, Marsden C (1999) Bradykinesia and impairment of EEG desynchronization in Parkinson's disease. Mov. Disord. 14: 423-429.
Brown P, Oliviero A, Mazzone P, Insola A, Tonali P, Lazzaro VD (2001) Dopamine dependency of oscillations between subthalamic nucleus and pallidum in Parkinson's disease. J. Neurosci. 21: 1033-1038.
DeLong M (1971) Activity of pallidal neurons during movement. J. Neurophysiol. 34: 414-427.
Deniau J, Chevalier M (1985) Disinhibition as a basic process in the expression of striatal functions. II. The striato-nigral influence on thalamocortical cells of the ventromedial thalamic nucleus. Brain Res. 334: 227-233.
Destexhe A, Contreras D, Steriade M (1998) Mechanisms underlying the synchronizing action of corticothalamic feedback through inhibition of thalamic relay cells. J. Neurophysiol. 79: 999-1016.
Dostrovsky J, Levy R, Wu J, Hutchison W, Tasker R, Lozano A (2000) Microstimulation-induced inhibition of neuronal firing in human globus pallidus. J. Neurophysiol. 84: 570-574.
Dostrovsky J, Lozano A (2002) Mechanisms of deep brain stimulation. Mov. Disord. 17(Supp. 3): S63-S68.
Ermentrout B (2002) Simulating, Analyzing, and Animating Dynamical Systems. Philadelphia: SIAM Press.
Filion M, Tremblay L (1991) Abnormal spontaneous activity of globus pallidus neurons in monkeys with MPTP-induced parkinsonism. Brain Res. 547: 142-151.
Gerfen C, Wilson C (1996) The basal ganglia. In: Swanson L, Björklund A, Hökfelt T, eds. Handbook of Chemical Neuroanatomy, Vol. 12: Integrated Systems of the CNS, Part III. Elsevier Science B.V.
Gross C (2001) Stimulation of the globus pallidus internus. In: Lozano A, ed. Movement Disorder Surgery. S. Karger AG, Basel.
Hashimoto T, Elder C, Okun M, Patrick S, Vitek J (2003) Stimulation of the subthalamic nucleus changes the firing pattern of pallidal neurons. J. Neurosci. 23: 1916-1923.
Hoover J, Strick P (1999) The organization of cerebellar and basal ganglia outputs to primary motor cortex as revealed by retrograde transneuronal transport of herpes simplex virus type 1. J. Neurosci. 19: 1446-1463.
Jech R, Urgosik D, Tintera J, Nebuzelsky A, Krakensy J, Liscak R, Roth J, Ruzicka E (2001) Functional magnetic resonance imaging during deep brain stimulation: A pilot study in four patients with Parkinson's disease. Mov. Disord. 16: 1126-1132.
Levy R, Lang A, Dostrovsky J, Pahapill P, Romas J, Saint-Cyr J, Hutchison W, Lozano A (2001) Lidocaine and muscimol microinjections in subthalamic nucleus reverse Parkinsonian symptoms. Brain 124: 2105-2118.
Limousin P, Greene J, Pollak P, Rothwell J, Benabid A, Frackowiak R (1997) Changes in cerebral activity pattern due to subthalamic nucleus or internal pallidum stimulation in Parkinson's disease. Ann. Neurol. 42: 283-291.
Macchi G, Jones E (1997) Toward an agreement on terminology of nuclear and subnuclear divisions of the motor thalamus. J. Neurosurg. 86: 670-685.
Magnin M, Morel A, Jeanmonod D (2000) Single-unit analysis of the pallidum, thalamus, and subthalamic nucleus in parkinsonian patients. Neuroscience 96: 549-564.
Mason A, Ilinsky I, Maldonado S, Kultas-Ilinsky K (2000) Thalamic terminal fields of individual axons from the ventral part of the dentate nucleus of the cerebellum in Macaca mullata. J. Comp. Neurol. 421: 412-428.
McIntyre C, Grill W (1999) Excitation of central nervous system neurons by nonuniform electric fields. Biophys. J. 76: 878-888.
Middleton F, Strick P (2000) Basal ganglia and cerebellar loops: Motor and cognitive circuits. Brain Res. Rev. 31: 236-250.
Montgomery E Jr, Baker K (2000) Mechanism of deep brain stimulation and future technical developments. Neurol. Res. 22: 259-266.
Nini A, Feingold A, Slovin H, Bergman H (1995) Neurons in the globus pallidus do not show correlated activity in the normal monkey, but phase-locked oscillations appear in the MPTP model of parkinsonism. J. Neurophysiol. 74: 1800-1805.
Nowak L, Bullier J (1998a) Axons, but not cell bodies, are activated by electrical stimulation in cortical gray matter: I. Evidence from chronaxie measurements. Exp. Brain Res. 118: 477-488.
Nowak L, Bullier J (1998b) Axons, but not cell bodies, are activated by electrical stimulation in cortical gray matter: II. Evidence from selective inactivation of cell bodies and axon initial segments. Exp. Brain Res. 118: 489-500.
Obeso J, Rodriguez M, DeLong M (1997) Basal ganglia pathophysiology: A critical review. In: Obeso J, DeLong M, Ohye C, Marsden C, eds. Advances in Neurology. Lippincott-Raven Publishers, Philadelphia. vol. 74, pp. 3-18.
Obeso J, Rodriguez-Oroz M, Rodriguez M, Macias R, Alvarez L, Guridi J, Vitek J, DeLong M (2000) Pathophysiologic basis of surgery for Parkinson's disease. Neurology 55 (Supp. 6): S7-S12.
Ogura M, Kita H (2000) Dynorphin exerts both postsynaptic and presynaptic effects in the globus pallidus of the rat. J. Neurophysiol. 83: 3366-3376.
Olanow W, Brin M (2001) In: Calne D, Calne SM, eds. Advances in Neurology, Vol. 86: Parkinson's Disease. Lippincott Williams & Wilkins, Philadelphia.
Olanow W, Brin M, Obeso J (2000) The role of deep brain stimulation as a surgical treatment for Parkinson's disease. Neurology 55, (Supp. 6): S60-S66.
Paul G, Reum T, Meissner W, Marburger A, Sohr R, Morgenstern R, Kupsch A (2000) High frequency stimulation of the subthalamic nucleus in uences striatal dopaminergic metabolism in naive rats. NeuroReport 11: 441-444.
Plenz D, Kitai S (1999) A basal ganglia pacemaker formed by the subthalamic nucleus and external globus pallidus. Nature 400: 677-682.
Pollak P, Fraix V, Krack P, Moro E, Mendes A, Chabardes S, Koudsie A, Benabid A (2002) Treatment results: Parkinson's disease. Mov. Disord. 17 (Supp. 3): S75-S83.
Raz A, Vaadia E, Bergman H (2000) Firing patterns and correlations of spontaneous discharge of pallidal neurons in the normal and tremulous 1-methyl-4-phenyl-1,2,3,6 tetrahydropyridine vervet model of parkinsonism. J. Neurosci. 20: 8559-8571.
Rinzel J (1985) Excitation dynamics: insights from simplified membrane models. Fed. Proc. 44: 2944-2946.
Rizzone M, Lanotte M, Bergamasco B, Tavella A, Torre E, Faccani G, Melcarne A, Lopiano L (2001) Deep brain stimulation of the subthalamic nucleus in Parkinson's disease: effects of variation in stimulation parameters. J. Neurol. Neurosurg. Psych. 71: 215-219.
Ryan L, Sanders D (1993) Subthalamic nucleus lesion regularizes firing patterns in globus pallidus and substantia nigra pars reticulata. Brain Res. 626: 327-331.
Sakai S, Inase M, Tanji J (2002) The relationship between MI and SMA afferents and cerebellar and pallidal efferents in the macaque monkey. Somatosens. & Mot. Res. 19: 139-148.
Sakai S, Stepniewska I, Qi H, Kaas J (2000) Pallidal and cerebellar afferents to presupplementary motor area thalamocortical neurons in the owlmonkey: a multiple labeling study. J. Comp. Neurol. 417: 164-180.
Sestini S, di Luzio AS, Ammannati F, DeCristofaro M, Passeri A, Martini S, Pupi A (2002) Changes in regional cerebral blood ow caused by deep-brain stimulation of the subthalamic nucleus in Parkinson's disease. J. Nucl. Med. 43: 725-732.
Smith G, Sherman S (2002) Detectability of excitatory versus inhibitory drive in an integrateand-fire-or-burst thalamocortical relay neuron model. J. Neurosci. 22: 10242-10250.
Sohal V, Huguenard J (2002) Reciprocal inhibition controls the oscillatory state in thalamic networks. Neurocomp. 44: 653-659.
Stanford I, Cooper A (1999) Presynaptic µ and δ opioid receptor modulation of GABAa IPSCs in the rat globus pallidus in vitro. J. Neurosci. 19: 4796-4803.
Steriade M, Contreras D, Amzica F (1997) The thalamocortical dialogue duringwake, sleep, and paroxysmal oscillations. In: Steriade M, Jones E, McCormick D, eds. Thalamus, Elsevier, Amsterdam, Vol. II, pp. 213-294.
Tass P (2002) Effective desynchronization with bipolar double-pulse stimulation. Phys. Rev. E 66: 0362261-0362269.
Terman D, Rubin J, Yew A, Wilson C (2001) Synchronous parkinsonian rhythms in a model for the indirect pathway of the basal ganglia. Neurocomp. 39/40: 973-982.
Terman D, Rubin J, Yew A, Wilson C (2002) Activity patterns in a model for the subthalamopallidal network of the basal ganglia. J. Neurosci. 22: 2963-2976.
The Deep Brain Stimulation for Parkinson's Disease Study Group. (2001) Deep-brain stimulation of the subthalamic nucleus or the pars interna of the globus pallidus in Parkinson's disease. New England J. Med. 345: 956-963.
Traub R, Miles R (1991) Neuronal Networks of the Hippocampus. Cambridge, UK: Cambridge University Press.
Vitek J (2002) Mechanisms of deep brain stimulation: excitation or inhibition. Mov. Disord. 17 (Supp. 3): S69-S72.
Vitek J, Giroux M (2000) Physiology of hypokinetic and hyperkinetic movement disorders: Model for dyskinesia. Ann. Neurol. 47 (4 Supp 1): S131-S140.
Wichmann T, Bergman H, Starr P, Subramanian T, Watts R, DeLong M (1999) Comparison of MPTP-induced changes in spontaneous neuronal discharge in the internal pallidal segment and in the substantia nigra pars reticulata in primates. Exp. Brain Res. 125: 397-409.
Windels F, Bruet N, Poupard A, Urbain N, Chouvet G, Feuerstein C, Savasta M(2000) Effects of high frequency stimulation of subthalamic nucleus on extracellular glutamate and GABA in substantia nigra and globus pallidus in the normal rat. Eur. J. Neurosci., 12: 4141-4146.
WuY, Levy R, Ashby P, Tasker R, Dostrovsky J (2001) Does stimulation of the GPi control dyskinesia by activating inhibitory axons? Mov. Disord. 16: 208-216.
Zhan X, Cox C, Rinzel J, Sherman S (1999) Current clamp and modeling studies of lowthreshold calcium spikes in cells of the cat's lateral geniculate nucleus. J. Neurophysiol. 81: 2360-2373.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rubin, J.E., Terman, D. High Frequency Stimulation of the Subthalamic Nucleus Eliminates Pathological Thalamic Rhythmicity in a Computational Model. J Comput Neurosci 16, 211–235 (2004). https://doi.org/10.1023/B:JCNS.0000025686.47117.67
Issue Date:
DOI: https://doi.org/10.1023/B:JCNS.0000025686.47117.67