Abstract
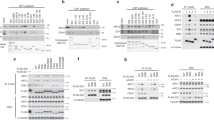
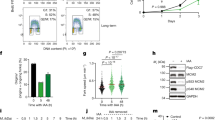
In eukaryotes, the activation of mitotic cyclin-dependent kinases (CDKs) induces mitosis, and their inactivation causes cells to leave mitosis1. In budding yeast, two redundant mechanisms induce the inactivation of mitotic CDKs. In one mechanism, a specialized ubiquitin-dependent proteolytic system (called the APC-dependent proteolysis machinery) degrades the mitotic (Clb) cyclin subunit. In the other, the kinase-inhibitor Sic1 binds to mitotic CDKs and inhibits their kinase activity1,2. The highly conserved protein phosphatase Cdc14 promotes both Clb degradation and Sic1 accumulation. Cdc14 promotes SIC1 transcription and the stabilization of Sic1 protein by dephosphorylating Sic1 and its transcription factor Swi5. Cdc14 activates the degradation of Clb cyclins by dephosphorylating the APC-specificity factor Cdh1 (refs 3, 4). So how is Cdc14 regulated? Here we show that Cdc14 is sequestered in the nucleolus for most of the cell cycle. During nuclear division, Cdc14 is released from the nucleolus, allowing it to reach its targets. A highly conserved signalling cascade, critical for the exit from mitosis, is required for this movement of Cdc14 during anaphase. Furthermore, we have identified a negative regulator of Cdc14, Cfi1, that anchors Cdc14 in the nucleolus.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Morgan, D. O. Cyclin-dependent kinases: engines, clocks and microprocessors. Annu. Rev. Cell Dev. Biol. 13, 261–291 (1997).
King, R. W., Deshaies, R. J., Peters, J.-M. & Kirschner, M. W. How proteolysis drives the cell cycle. Science 274, 1652–1659 (1996).
Visintin, R.et al. The phosphatase Cdc14 triggers mitotic exit by reversal of Cdk-dependent phosphorylation. Mol. Cell 2, 709–718 (1998).
Zachariae, W., Schwab, M., Nasmyth, K. & Seufert, W. Control of cyclin ubiquitination by CDK-regulated binding of Hct1 to the anaphase promoting complex. Science 282, 1721–1724 (1998).
Taylor, G. S., Liu, Y., Baskerville, C. & Charbonneau, H. The activity of Cdc14p, an oligomeric dual specificity protein phosphatase from Saccharomyces cerevisiae is required for cell cycle progression. J. Biol. Chem. 272, 24054–24063 (1997).
Tollervey, D., Lehtonen, H., Carmo-Fonseca, M. & Hurt, E. C. The small nucleolar RNP protein NOP1 (fibrillarin) is required for pre-rRNA processing in yeast. EMBO J. 10, 573–583 (1991).
Smith, J. S., Brachmann, C. B., Pillus, L. & Boeke, J. D. Distribution of a limited Sir2 protein pool regulates the strength of yeast rDNA silencing and is modulated by Sir4p. Genetics 149, 1205–1219 (1998).
Maillet, L.et al. Evidence for silencing compartments within the yeast nucleus: a role for telomere proximity and Sir protein concentration in silencer-mediated repressions. Genes Dev. 10, 1796–1811 (1996).
Bell, S. P. & Stillman, B. ATP-dependent recognition of eukaryotic origins of DNA replication by a multiprotein complex. Nature 357, 128–134 (1992).
Guacci, V., Koshland, D. & Strunnikov, A. Adirect link between sister chromatid cohesion and chromosome condensation revealed through the analysis of MCD1 in S. cerevisiae. Cell 91, 47–57 (1997).
Michaelis, C., Ciosk, R. & Nasmyth, K. Cohesins: Chromosomal proteins that prevent premature separation of sister chromatids. Cell 91, 35–45 (1997).
Surana, U.et al. Destruction of the CDC28/CLB kinase is not required for metaphase/anaphase transition in yeast. EMBO J. 12, 1969–1978 (1993).
Kitada, K., Johnson, L. A., Johnston, L. H. & Sugino, A. Amulticopy suppressor gene of the Saccharomyces cerevisiae G1 cell cycle mutant dbf4 encodes in a protein kinase and is identified as CDC5. Mol. Cell Biol. 13, 4445–4457 (1993).
Toyn, J. H. & Johnston, L. H. The Dbf2 and Dbf20 protein kinases of budding yeast are activated after the metaphase to anaphase cell cycle transition. EMBO J. 13, 1103–1113 (1994).
Shirayama, M., Matsui, Y. & Toh-e, A. The yeast TEM1 gene, which encodes a GTP-binding protein, is involved in termination of M phase. Mol. Cell Biol. 14, 7476–7482 (1994).
Shirayama, M., Matsui, Y. & Toh-e, A. Isolation of a Cdc25 family gene, MSI1/LTE1, as a multicopy suppressor of ira1. Yeast 10, 451–461 (1994).
Ciosk, R.et al. An ESP1/PDS1 complex regulates loss of sister chromatid cohesion at the metaphase to anaphase transition in yeast. Cell 93, 1067–1076 (1998).
McGrew, J. T., Goetsch, L., Byers, B. & Baum, P. Requirement for ESP1 in the nuclear division of Saccharomyces cerevisiae. Mol. Biol Cell 3, 1443–1454 (1992).
Visintin, R., Prinz, S. & Amon, A. CDC20 and CDH1 : a family of substrate-specific activators of APC-dependent proteolysis. Science 278, 460–463 (1997).
Yamamoto, A., Guacci, V. & Koshland, D. Pds1, an inhibitor of anaphase in budding yeast, plays a critical role in the APC and checkpoint pathway(s). J. Cell Biol. 133, 99–110 (1996).
Shirayama, M., Zachariae, W., Ciosk, R. & Nasmyth, K. The Polo-like kinase Cdc5p and the WD-repeat protein Cdc20p/fizzy are regulators and substrates of the anaphase promoting complex in Saccharomyces cerevisiae. EMBO J. 17, 1336–1349 (1998).
James, P., Halladay, J. & Craig, E. A. Genomic libraries and a host strain designed for highly efficient two-hybrid selection in yeast. Genetics 144, 1425–1436 (1996).
Tu, J. & Carlson, M. REG1 binds to protein phosphatase type 1 and regulates glucose repression in Saccharomyces cerevisiae. EMBO J. 14, 5939–5946 (1995).
Amon, A., Irniger, S. & Nasmyth, K. Closing the cell cycle circle in yeast: G2 cyclin proteolysis initiated at mitosis persists until the activation of G1 cyclins in the next cell cycle. Cell 77, 1037–1050 (1994).
Schneider, B. L., Seufert, W., Steiner, B., Yang, Q. H. & Futcher, A. B. Use of polymerase chain reaction epitope tagging for protein tagging in Saccharomyces cerevisiae. Yeast 13, 1265–1274 (1995).
Cohen-Fix, O., Peters, J.-M., Kirschner, M. W. & Koshland, D. Anaphase initiation in Saccharomyces cerevisiae is controlled by the APC-dependent degradation of the anaphase inhibitor Pds1p. Genes Dev. 10, 3081–3093 (1996).
Kilmartin, J. V. & Adams, A. E. Structural rearrangements of tubulin and actin during the cell cycle of the yeast Saccharomyces. J. Cell Biol. 98, 922–933 (1984).
Higgins, D. G., Thompson, J. D. & Gibson, T. J. Using CLUSTAL for multiple sequence alignments. Methods Enzymol. 383–402 (1996).
Acknowledgements
We thank V. Guacci, L. Guarente, L. Pillus, S. Bell and F. Klein for gifts of strains and antibodies; F. Lewitter for help with sequence alignments; and M. Tyers, S. Bell, T. Orr-Weaver, P. Sorger and S. Prinz for critically reading the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Visintin, R., Hwang, E. & Amon, A. Cfi1 prevents premature exit from mitosis by anchoring Cdc14 phosphatase in the nucleolus. Nature 398, 818–823 (1999). https://doi.org/10.1038/19775
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/19775
This article is cited by
-
Analog-sensitive Cdk1 as a tool to study mitotic exit: protein phosphatase 1 is required downstream from Cdk1 inactivation in budding yeast
Chromosome Research (2023)
-
The regional sequestration of heterochromatin structural proteins is critical to form and maintain silent chromatin
Epigenetics & Chromatin (2022)
-
The phosphatase gene MaCdc14 negatively regulates UV-B tolerance by mediating the transcription of melanin synthesis-related genes and contributes to conidiation in Metarhizium acridum
Current Genetics (2020)
-
Cdc14 activation requires coordinated Cdk1-dependent phosphorylation of Net1 and PP2A–Cdc55 at anaphase onset
Cellular and Molecular Life Sciences (2019)
-
Protein phosphatases of Saccharomyces cerevisiae
Current Genetics (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.