Abstract
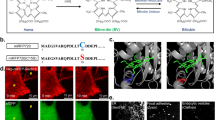
Light is a crucial environmental signal that controls many photomorphogenic and circadian responses in plants1. Perception and transduction of light is achieved by at least two principal groups of photoreceptors, phytochromes and cryptochromes2,3. Phytochromes are red/far-red light-absorbing receptors encoded by a gene family of five members (phyA to phyE)2,4 in Arabidopsis. Cryptochrome 1 (cry1), cryptochrome 2 (cry2) and phototropin are the blue/ultraviolet-A light receptors that have been characterized in Arabidopsis5. Previous studies showed that modulation of many physiological responses in plants is achieved by genetic interactions between different photoreceptors6; however, little is known about the nature of these interactions and their roles in the signal transduction pathway. Here we show the genetic interaction that occurs between the Arabidopsis photoreceptors phyB and cry2 in the control of flowering time, hypocotyl elongation and circadian period by the clock. PhyB interacts directly with cry2 as observed in co-immunoprecipitation experiments with transgenic Arabidopsis plants overexpressing cry2. Using fluorescent resonance energy transfer microscopy, we show that phyB and cry2 interact in nuclear speckles that are formed in a light-dependent fashion.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Kendric, R. E. & Kronenberg, G. H. M. Photomorphogenesis in Plants (Kluwer Academic, Dordrecht, 1994).
Quail, P. H. et al. Phytochromes: photosensory perception and signal transduction. Science 268, 675–680 (1995).
Cashmore, A. R., Jarillo, J. A., Wu, Y. & Liu, D. Cryptochromes: Blue light receptors for plants and animals. Science 284 , 760–765 (1999).
Chory, J. Light modulation of vegetative development. Plant Cell 9, 1225–1234 (1997).
Briggs, W. R. & Huala, E. Blue-light photoreceptors in higher plants. Annu. Rev. Cell Dev. Biol. 15, 33 –62 (1999).
Casal, J. J. Phytochromes, cryptochromes, phototropin: photoreceptor interactions in plants. Photochem. Photobiol. 71, 1– 11 (2000).
Somers, D. E., Devlin, P. F. & Kay, S. A. Phytochromes and cryptochromes in the entrainment of the Arabidopsis circadian clock. Science 282, 1488–1490 (1998).
Millar, A. J., Straume, M., Chory, J., Chua, N. H. & Kay, S. A. The regulation of circadian period by phototransduction pathways in Arabidopsis. Science 267, 1163–1166 (1995).
Whitelam, G. C. & Devlin, P. F. Roles of different phytochromes in Arabidopsis photomorphogenesis. Plant Cell Environ. 20, 752–758 ( 1998).
Guo, H., Yang, H., Mockler, T. C. & Lin, C. Regulation of flowering time by Arabidopsis photoreceptors. Science 279, 1360–1363 (1998).
Mockler, T. C., Guo, H., Yang, H., Duong, H. & Lin, C. Antagonistic actions of Arabidopsis cryptochromes and phytochrome B in the regulation of floral induction. Development 126, 2073–2082 (1999).
Reed, J. W., Nagpal, P., Poole, D. S., Furuya, M. & Chory, J. Mutations in the gene for the red/far red light receptor phytochrome B alter cell elongation and physiological responses throughout Arabidopsis development. Plant Cell 5, 147–157 (1993).
Lin, C. et al. Enhancement of blue-light sensitivity of Arabidopsis seedlings by blue light receptor cryptochrome 2. Proc. Natl Acad. Sci. USA 95, 2686–2690 ( 1998).
Guo, H., Duong, H., Ma, N. & Lin, C. The Arabidopsis blue light receptor cryptochrome 2 is a nuclear protein regulated by a blue light-dependent post-transcriptional mechanism. Plant J. 19, 279–287 ( 1999).
Kleiner, O., Kircher, S., Harter, K. & Batschauer, A. Nuclear localization of the Arabidopsis blue light receptor cryptochrome 2. Plant J. 19, 289–296 ( 1999).
Yamaguchi, R., Nakamura, M., Mochizuki, N., Kay, S. A. & Nagatani, A. Light-dependent translocation of a phytochrome B-GFP fusion protein to the nucleus in transgenic Arabidopsis . J. Cell Biol. 145, 437– 445 (1999).
Kircher, S. et al. Light quality-dependent nuclear import of the plant photoreceptors phytochrome A and B. Plant Cell 11, 1445 –1456 (1999).
Wu, P. & Brand, L. Resonance energy transfer: methods and applications. Anal. Biochem. 218, 1– 13 (1994).
Gadella, T. W. J., van der Krogt, G. N. M. & Bisseling, T. GFP-based FRET microscopy in living plant cells. Trends Plant Sci. 4, 287– 291 (1999).
Bastiaens, P. I. H. & Jovin, T. M. in Cell Biology: A Laboratory Handbook (ed. Celis, J. E.) 136– 146 (Academic, New York, 1998).
Yeh, K. C. & Lagarias, J. C. Eukaryotic phytochromes: light-regulated serine/threonine protein kinases with histidine kinase ancestry. Proc. Natl Acad. Sci. USA 95, 13976– 13981 (1998).
Ni, M., Tepperman, J. M. & Quail, P. H. PIF3, a phytochrome-interacting factor necessary for normal photoinduced signal transduction, is a novel basic helix–loop–helix protein. Cell 95, 657–667 (1998).
Fankhauser, C. et al. PKS1, a substrate phosphorylated by phytochrome that modulates light signaling in Arabidopsis. Science 284, 1539–1541 (1999).
Choi, G. et al. Phytochrome signalling is mediated through nucleoside diphosphate kinase 2. Nature 401, 610– 613 (1999).
Ahmad, M., Jarrillo, J. A., Smirnova, O. & Cashmore, A. R. The CRY1 blue light photoreceptor of Arabidopsis interacts with phytochrome in vitro. Mol. Cell 1, 939– 948 (1998).
Lamond, A. I. & Earnshaw, W. C. Structure and function in the nucleus. Science 280, 547– 553 (1998).
Somers, D. E., Schultz, T. F., Milnamow, M. & Kay, S. A. ZEITLUPE encodes a novel clock-associated PAS protein from Arabidopsis . Cell 101, 319–329 (2000).
Nelson, D. C., Lasswell, J., Rogg, L. E., Cohen, M. A. & Bartel, B. FKF1, a clock-controlled gene that regulates the transition to flowering in Arabidopsis. Cell 101, 331–340 ( 2000).
Carrington, J. C., Freed, D. D. & Leinicke, A. J. Bipartite signal sequence mediates nuclear translocation of the plant potyviral NIa protein. Plant Cell 3, 953–962 (1991).
Más, P. & Beachy, R. N. Replication of tobacco mosaic virus on endoplasmic reticulum and role of the cytoskeleton and virus movement protein in intracellular distribution of viral RNA. J. Cell Biol. 147, 945–958 (1999).
Acknowledgements
We thank S. L. Harmer, T. F. Schultz and M. J. Yanovsky for critical comments of the manuscript. We are grateful to C. Lin for providing cry2 cDNA, the cry2 overexpression line and cry2 antibody. We also thank A. Nagatani for the phyB–GFP construct and anti-phyB antibody. We thank G. Patterson and D. Piston for GFP spectra data, and Clontech for DsRFP spectra data. We are grateful to D. Millar and T. K. Nomanbhoy for helpful discussion on FRET analysis. Research support came from the NIH. P.M. was supported by a Novartis Agricultural Discovery Institute and P.F.D. was supported by an European Molecular Biology Organization long-term fellowship and the NSF. S.P. was supported by TSRI graduate program.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Más, P., Devlin, P., Panda, S. et al. Functional interaction of phytochrome B and cryptochrome 2. Nature 408, 207–211 (2000). https://doi.org/10.1038/35041583
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35041583
This article is cited by
-
Efficient transformation of the isolated microspores of Chinese cabbage (Brassica rapa L. ssp. pekinensis) by particle bombardment
Plant Methods (2024)
-
Diversity of gene expression responses to light quality in barley
Scientific Reports (2023)
-
CRY2 gene of rice (Oryza sativa subsp. indica) encodes a blue light sensory receptor involved in regulating flowering, plant height and partial photomorphogenesis in dark
Plant Cell Reports (2023)
-
Arabidopsis cryptochrome 2 forms photobodies with TCP22 under blue light and regulates the circadian clock
Nature Communications (2022)
-
Rapid and reversible optogenetic silencing of synaptic transmission by clustering of synaptic vesicles
Nature Communications (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.