Abstract
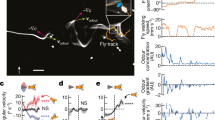
IN studies of moths flying upwind to a pheromone source, attention has focused on the influence on flight orientation of the composition1,2 and concentration3,4 of the chemical message, and of changes in the visual environment5,6 and in wind speeds7–9. The chemical signal must be intermittent for moths to fly upwind10–12, when they usually follow a zigzag track, the evident expression of a self-steered counterturning programme13,14. The integration of counterturning and optomotor anemotaxis allows insects to polarize the zigzags upwind in odour plumes10,15. Not all moths, however, zigzag along a plume16,17. It has been suggested that the propensity to zigzag or to fly straight upwind is related to the frequency at which males encounter pheromone filaments that comprise the plume, as well as the male's latency of response, characteristic for each moth species, to both the onset and loss of contact with filaments18. Here we present evidence that flight manoeuvres are dictated by the interactions of the male with individual odour pulses. We use Cadra cautella, the almond moth, to show how the structure of an odour plume19,20 can greatly modify the flight track. Males following either turbulent or mechanically pulsed plumes fly faster and straighter upwind, and locate sources more frequently than males following continuous narrow plumes. Males also fly straighter upwind to fast-pulsed plumes than to slow-pulsed plumes. The temporally modulated interplay between counterturning and optomotor anemotaxis that is induced by the plume's structure therefore seems to explain the manoeuvres and resultant flight track shapes made by C. cautella males when flying upwind towards a pheromone source.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Linn, C. E., Campbell, M. G. & Roelofs, W. L. Science 237, 650–652 (1987).
Willis, M. A. & Baker, T. C. J. Insect Behav. 1, 357–371 (1988).
Kuenen, L. P. S. & Baker, T. C. Physiol. Ent. 7, 423–434 (1982).
Cardé, R. T. & Hagaman, T. E. Environ. Ent. 8, 475–484 (1979).
Kuenen, L. P. S. & Baker, T. C. Physiol. Ent. 7, 193–202 (1982).
David, C. T. J. comp. Physiol. A 147, 485–493 (1982).
Farkas, S. R. & Shorey, H. H. Science 178, 67–68 (1972).
Willis, M. A. & Cardé, R. T. J. comp. Physiol. A 167, 699–706 (1990).
Willis, M. A. & Arbas, E. A. J. comp. Physiol. A 169, 427–440 (1991).
Kennedy, J. S., Ludlow, A. R. & Sanders, C. J. Nature 295, 475–477 (1980).
Willis, M. A. & Baker, T. C. Physiol. Ent. 9, 341–358 (1983).
Baker, T. C., Willis, M. A., Haynes, K. F. & Phelan, P. L. Physiol. Ent. 10, 257–265 (1985).
Kennedy, J. S. Physiol. Ent. 8, 109–120 (1983).
Kennedy, J. S. in Mechanisms in Insect Olfaction (eds Payne, T. L., Birch, M. C. & Kennedy, C. E. J.) 11–25 (Clarendon, Oxford, 1986).
Baker, T. C., Willis, M. A. & Phelan, P. L. Physiol. Ent. 9, 365–376 (1984).
Haynes, K. F. & Baker, T. C. Physiol. Ent. 14, 279–289 (1989).
Witzgall, P. & Arn, H. Z. Naturforsch 45c, 1067–1069 (1990).
Baker, T. C. Proc. 10th Internat. Symp. on Olfaction and Taste (ed. Døving, K. B.) 18–25 (Oslo, 1990).
Wright, R. H. Can. Ent. 90, 81–89 (1958).
Murlis, J., Elkinton, J. S. & Cardé, R. T. A. Rev. Ent. 37, 505–532 (1992).
Mafra-Neto, A. thesis, Univ. Massachusetts, Amherst (1993).
Kuenen, L. P. S. & Baker, T. C. Physiol. Ent. 7, 423–434 (1982).
Vickers, N. J. & Baker, T. C. J. Insect Behav. 5, 669–687 (1992).
Vickers, N. J. thesis, Univ. California, Riverside (1992).
Charlton, R. E., Kanno, H., Collins, R. D. & Cardé, R. T. Physiol. Ent. 18, 349–362 (1993).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mafra-Neto, A., Cardé, R. Fine-scale structure of pheromone plumes modulates upwind orientation of flying moths. Nature 369, 142–144 (1994). https://doi.org/10.1038/369142a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/369142a0
This article is cited by
-
Sensorimotor transformation underlying odor-modulated locomotion in walking Drosophila
Nature Communications (2023)
-
Disrupting pest reproduction techniques can replace pesticides in vineyards. A review
Agronomy for Sustainable Development (2023)
-
Reduced olfactory acuity in recently flightless insects suggests rapid regressive evolution
BMC Ecology and Evolution (2022)
-
Variation in the ratio of compounds in a plant volatile blend during transmission by wind
Scientific Reports (2022)
-
Odour motion sensing enhances navigation of complex plumes
Nature (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.