Abstract
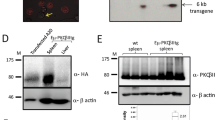
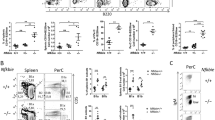
Protein kinase C (PKC), which comprises 11 closely related isoforms, has been implicated in a wide variety of cellular processes, such as growth, differentiation, secretion, apoptosis and tumour development1,2,3,4. Among the PKC isotypes, PKC-δ is unique in that its overexpression results in inhibition of cell growth5,6,7,8,9,10,11. Here we show that mice that lack PKC-δ exhibit expansion of the B-lymphocyte population with the formation of numerous germinal centres in the absence of stimulation. The rate of proliferation in response to stimulation was greater for B cells from PKC-δ-deficient mice than for those from wild-type mice. Adoptive transfer experiments suggested that the hyperproliferation phenotype is B-cell autonomous. Production of interleukin-6 was markedly increased in B cells of PKC-δ-null mice as a result of an increase in the DNA-binding activity of NF-IL6. Furthermore, the PKC-δ-deficient mice contain circulating autoreactive antibodies and display immune-complex-type glomerulonephritis, as well as lymphocyte infiltration in many organs. These results suggest that PKC-δ has an indispensable function in negative regulation of B-cell proliferation, and is particularly important for the establishment of B-cell tolerance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Nishizuka, Y. Protein kinase C and lipid signaling for sustained cellular responses. FASEB J. 9, 484–496 (1995)
Mellor, H. & Parker, P. J. The extended protein kinase C superfamily. Biochem. J. 332, 281–292 (1998)
Livneh, E. & Fishman, D. D. Linking protein kinase C to cell-cycle control. Eur. J. Biochem. 248, 1–9 (1997)
Weiss, A. & Littman, D. R. Signal transduction by lymphocyte antigen receptors. Cell 76, 263–274 (1994)
Ohno, S., Akita, Y., Konno, Y., Imajoh, S. & Suzuki, K. A novel phorbol ester receptor/protein kinase, nPKC, distantly related to the protein kinase C family. Cell 53, 731–741 (1988)
Gschwendt, M. Protein kinase Cδ. Eur. J. Biochem. 259, 555–564 (1999)
Watanabe, T. et al. Cell division arrest induced by phorbol ester in CHO cells overexpressing protein kinase C-δ subspecies. Proc. Natl Acad. Sci. USA 89, 10159–10163 (1992)
Mischak, H. et al. Overexpression of protein kinase C-δ and -ε in NIH 3T3 cells induces opposite effects on growth, morphology, anchorage dependence, and tumorigenicity. J. Biol. Chem. 268, 6090–6096 (1993)
Fukumoto, S. et al. Protein kinase Cδ inhibits the proliferation of vascular smooth muscle cells by suppressing G1 cyclin expression. J. Biol. Chem. 272, 13816–13822 (1997)
Mishima, K., Ohno, S., Shitara, N., Yamaoka, K. & Suzuki, K. Opposite effects of the overexpression of protein kinase Cγ and δ on the growth properties of human glioma cell line U251 MG. Biochem. Biophys. Res. Commun. 201, 363–372 (1994)
Harrington, E. O. et al. Enhancement of migration by protein kinase Cα and inhibition of proliferation and cell cycle progression by protein kinase Cδ in capillary endothelial cells. J. Biol. Chem. 272, 7390–7397 (1997)
Pessino, A. et al. Antisense oligodeoxynucleotide inhibition of δ protein kinase C expression accelerates induced differentiation of murine erythroleukaemia cells. Biochem. J. 312, 549–554 (1995)
Li, W. & Pierce, J. H. Protein kinase C-δ, an important signaling molecule in the platelet-derived growth factor β receptor pathway. Curr. Topics Microbiol. Immunol. 211, 55–65 (1996)
Mischak, H. et al. Phorbol ester-induced myeloid differentiation is mediated by protein kinase C-α and -δ and not by protein kinase C-β II, -ε, -ζ, and -η. J. Biol. Chem. 268, 20110–20115 (1993)
Emoto, Y., Kisaki, H., Manome, Y., Kharbanda, S. & Kufe, D. Activation of protein kinase Cδ in human myeloid leukemia cells treated with 1-β-d-arabinofuranosylcytosine. Blood 87, 1990–1996 (1996)
Emoto, Y. et al. Proteolytic activation of protein kinase Cδ by an ICE-like protease in apoptotic cells. EMBO J. 14, 6148–6156 (1995)
Ghayur, T. et al. Proteolytic activation of protein kinase Cδ by an ICE/CED 3-like protease induces characteristics of apoptosis. J. Exp. Med. 184, 2399–2404 (1996)
Li, W. et al. Expression of an ATP binding mutant of PKC-δ inhibits Sis-induced transformation of NIH3T3 cells. Oncogene 13, 731–737 (1996)
La Porta, C. A. & Comolli, R. Over-expression of protein kinase Cδ is associated with a delay in preneoplastic lesion development in diethylnitrosamine-induced rat hepatocarcinogenesis. Carcinogenesis 16, 1233–1238 (1995)
Geiges, D., Marks, F. & Gschwendt, M. Loss of protein kinase Cδ from human HaCaT keratinocytes upon ras transfection is mediated by TGF α. Exp. Cell Res. 219, 299–303 (1995)
Lu, Z. et al. Tumor promotion by depleting cells of protein kinase Cδ. Mol. Cell. Biol. 17, 3418–3428 (1997)
Reddig, P. J. et al. Transgenic mice overexpressing protein kinase Cδ in the epidermis are resistant to skin tumor promotion by 12-O-tetradecanoylphorbol-13-acetate. Cancer Res. 59, 5710–5718 (1999)
Akira, S. & Kishimoto, T. IL-6 and NF-IF6 in acute-phase response and viral infection. Immunol. Rev. 127, 25–50 (1992)
Trautwein, C., van der Geer, P., Karin, M., Hunter, T. & Chojkier, M. Protein kinase A and C site-specific phosphorylations of LAP (NF-IL6) modulate its binding affinity to DNA recognition elements. J. Clin. Invest. 93, 2554–2561 (1994)
Leitges, M. et al. Immunodeficiency in protein kinase Cβ-deficient mice. Science 273, 788–791 (1996)
Sun, Z. et al. PKC-θ is required for TCR-induced NF-κB activation in mature but not immature T lymphocytes. Nature 404, 402–407 (2000)
Suematsu, S. et al. IgG1 plasmacytosis in interleukin 6 transgenic mice. Proc. Natl Acad. Sci. USA 86, 7547–7551 (1989)
Nakayama, K. et al. Mice lacking p27Kip1 display increased body size, multiple organ hyperplasia, retinal dysplasia, and pituitary tumors. Cell 85, 707–720 (1996)
Tsukiyama, T. et al. Down-regulation of p27Kip1 expression is required for development and function of T cells. J. Immunol. 166, 304–312 (2001)
Burlingame, R. W. & Rubin, R. L. Subnucleosome structures as substrates in enzyme-linked immunosorbent assays. J. Immunol. Methods 134, 187–199 (1990)
Acknowledgements
We thank S. Akira and K. Hoshino for the plasmids; N. Motoyama, A. Yoshimura and U. Kikkawa for discussions; Y. Yamada, K. Shimoharada and N. Ohtsuji for technical assistance; and M. Kimura and C. Sugita for help in preparation of the manuscript. This work was supported in part by a grant from the Ministry of Education, Science, Sports and Culture of Japan, by Nissan Science Foundation, and by a research grant from the Human Frontier Science Program.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests
Rights and permissions
About this article
Cite this article
Miyamoto, A., Nakayama, K., Imaki, H. et al. Increased proliferation of B cells and auto-immunity in mice lacking protein kinase Cδ. Nature 416, 865–869 (2002). https://doi.org/10.1038/416865a
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/416865a
This article is cited by
-
Up-regulation of PKCα and δ during beating cardiomyocyte differentiation of P19CL6 cells with suppressed apoptotic cell populations
Molecular & Cellular Toxicology (2024)
-
Genetic association of PRKCD and CARD9 polymorphisms with Vogt–Koyanagi–Harada disease in the Chinese Han population
Human Genomics (2023)
-
PKCδ regulates the vascular biology in diabetic atherosclerosis
Cell Communication and Signaling (2023)
-
Cellular and molecular mechanisms breaking immune tolerance in inborn errors of immunity
Cellular & Molecular Immunology (2021)
-
Equivocal, explicit and emergent actions of PKC isoforms in cancer
Nature Reviews Cancer (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.